Abstract
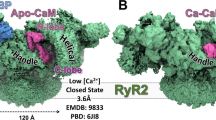
In muscle, excitation-contraction coupling is defined as the process linking depolarization of the surface membrane with Ca2+ release from cytoplasmic stores, which activates contraction of striated muscle. This process is primarily controlled by interplay between two Ca2+ channels—the voltage-gated L-type Ca2+ channel (dihydropyridine receptor, DHPR) localized in the t-tubule membrane and the Ca2+-release channel (ryanodine receptor, RyR) of the sarcoplasmic reticulum membrane. The structures of both channels have been extensively studied by several groups using electron cryomicroscopy and single particle reconstruction techniques. The structures of RyR, determined at resolutions of 22–30 Å, reveal a characteristic mushroom shape with a bulky cytoplasmic region and the membrane-spanning stem. While the cytoplasmic region exhibits a complex structure comprising a multitude of distinctive domains with numerous intervening cavities, at this resolution no definitive statement can be made about the location of the actual pore within the transmembrane region. Conformational changes associated with functional transitions of the Ca2+ release channel from closed to open states have been characterized. Further experiments determined localization of binding sites for various channel ligands. The structural studies of the DHPR are less developed. Although four 3D maps of the DHPR were reported recently at 24–30 Å resolution from studies of frozen-hydrated and negatively stained receptors, there are some discrepancies between reported structures with respect to the overall appearance and dimensions of the channel structure. Future structural studies at higher resolution are needed to refine the structures of both channels and to substantiate a proposed molecular model for their interaction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
E. Rios G. Brum (1987) Nature 325 717–720 Occurrence Handle2434854 Occurrence Handle1:CAS:528:DyaL2sXkt1Wht78%3D
M. Nabauer G. Callewaert L. Cleemann M. Morad (1989) Science 244 800–803 Occurrence Handle2543067 Occurrence Handle1:STN:280:DyaL1M3ms1yrtg%3D%3D
T. Tanabe K. G. Beam J. A. Powell S. Numa (1988) Nature 336 134–139 Occurrence Handle2903448 Occurrence Handle1:CAS:528:DyaL1MXltlGl
M. F. Schneider W. K. Chandler (1973) Nature 242 244–246 Occurrence Handle4540479 Occurrence Handle1:STN:280:CSyC2Mzltlw%3D
G. Pizarro L. Csernoch I. Uribe M. Rodriguez E. Rios (1991) J. Gen. Physiol. 97 913–947 Occurrence Handle1650812 Occurrence Handle1:STN:280:By6A38vosVU%3D
W. Chiu K. H. Downing J. Dubochet R. M. Glaeser H. G. Heide E. Knapek D. A. Kopf M. K. Lamvik J. Lepault J. D. Robertson E. Zeitler F. Zemlin (1986) J. Microsc. 141 385–391
J. Dubochet M. Adrian J. J. Chang J. C. Homo J. Lepault A. W. McDowall P. Schultz (1988) Q. Rev. Biophys. 21 129–228 Occurrence Handle3043536 Occurrence Handle1:STN:280:BieA3cvkvVI%3D Occurrence Handle10.1017/S0033583500004297
H. Takeshima S. Nishimura T. Matsumoto H. Ishida K. Kangawa N. Minamino H. Matsuo M. Ueda M. Hanaoka T. Hirose et al. (1989) Nature 339 439–445 Occurrence Handle2725677 Occurrence Handle1:CAS:528:DyaK3cXhsFertb8%3D
F. Zorzato J. Fujii K. Otsu M. Phillips N. M. Green F. A. Lai G. Meissner D. H. MacLennan (1990) J. Biol. Chem. 265 2244–2256 Occurrence Handle2298749 Occurrence Handle1:CAS:528:DyaK3cXksFegu74%3D
Y. Hakamata J. Nakai H. Takeshima K. Imoto (1992) FEBS Lett. 312 229–235 Occurrence Handle1330694 Occurrence Handle1:CAS:528:DyaK3sXltFWmsL0%3D
R. E. Tunwell C. Wickenden B. M. Bertrand V. I. Shevchenko M. B. Walsh P. D. Allen F. A. Lai (1996) Biochem. J. 318 477–487 Occurrence Handle8809036 Occurrence Handle1:CAS:528:DyaK28XlvFSmtbk%3D
J. L. Sutko J. A. Airey (1996) Physiol. Rev. 76 1027–1071 Occurrence Handle8874493 Occurrence Handle1:CAS:528:DyaK28XntVSjtL8%3D
V. Sorrentino P. Volpe (1993) Trends Pharmacol. Sci. 14 98–103 Occurrence Handle8387707 Occurrence Handle1:CAS:528:DyaK3sXkt1Gjtbc%3D
V. Sorrentino C. Reggiani (1999) Trends Cardiovasc. Med. 9 54–61 Occurrence Handle10189968 Occurrence Handle1:CAS:528:DyaK1MXitFGksbw%3D
F. A. Lai M. Misra L. Xu H. A. Smith G. Meissner (1989) J. Biol. Chem. 264 16776–16785 Occurrence Handle2550460 Occurrence Handle1:CAS:528:DyaL1MXlvV2rt7k%3D
M. Radermacher V. Rao R. Grassucci J. Frank A. P. Timerman S. Fleischer T. Wagenknecht (1994) J. Cell. Biol. 127 411–423 Occurrence Handle7929585 Occurrence Handle1:CAS:528:DyaK2cXmt1Clsr8%3D
T. Wagenknecht M. Radermacher (1995) FEBS Lett. 369 43–46 Occurrence Handle7641882 Occurrence Handle1:CAS:528:DyaK2MXntl2gsL0%3D
I. I. Serysheva E. V. Orlova W. Chiu M. B. Sherman S. L. Hamilton M. Heel Particlevan (1995) Nat. Struct. Biol. 2 18–24 Occurrence Handle7719847 Occurrence Handle1:CAS:528:DyaK2MXivF2ls7s%3D
E. V. Orlova I. I. Serysheva M. Heel Particlevan S. L. Hamilton W. Chiu (1996) Nat. Struct. Biol. 3 547–552 Occurrence Handle8646541 Occurrence Handle1:CAS:528:DyaK28Xjtlylsr8%3D
C. C. Yin F. A. Lai (2000) Nat. Cell. Biol. 2 669–671 Occurrence Handle10980710 Occurrence Handle1:CAS:528:DC%2BD3cXmslGks7Y%3D
M. L. Baker I. I. Serysheva S. Sencer Y. Wu S. J. Ludtke W. Jiang S. L. Hamilton W. Chiu (2002) Proc. Natl. Acad. Sci. USA 99 12155–12160 Occurrence Handle12218169 Occurrence Handle1:CAS:528:DC%2BD38XntlClu7k%3D
M. R. Sharma P. Penczek R. Grassucci H. B. Xin S. Fleischer T. Wagenknecht (1998) J. Biol. Chem. 273 18429–18434 Occurrence Handle9660811 Occurrence Handle1:CAS:528:DyaK1cXkvVCgsb4%3D
L. H. Jeyakumar J. A. Copello A. M. O’Malley G. M. Wu R. Grassucci T. Wagenknecht S. Fleischer (1998) J. Biol. Chem. 273 16011–16020 Occurrence Handle9632651 Occurrence Handle1:CAS:528:DyaK1cXkt1Giu70%3D
B. A. Block T. Imagawa K. P. Campbell C. Franzini-Armstrong (1988) J. Cell. Biol. 107 2587–2600 Occurrence Handle2849609 Occurrence Handle1:STN:280:DyaL1M%2FnvFOltA%3D%3D
T. Wagenknecht M. Radermacher R. Grassucci J. Berkowitz H. B. Xin S. Fleischer (1997) J. Biol. Chem. 272 32463–32471 Occurrence Handle9405457 Occurrence Handle1:CAS:528:DyaK1cXislWktg%3D%3D
M. Samso T. Wagenknecht (2002) J. Biol. Chem. 277 1349–1353 Occurrence Handle11694536 Occurrence Handle1:CAS:528:DC%2BD38XnvFOrtA%3D%3D
G. G. Du B. Sandhu V. K. Khanna X. H. Guo D. H. MacLennan (2002) Proc. Natl. Acad. Sci. USA 99 16725–16730 Occurrence Handle12486242 Occurrence Handle1:CAS:528:DC%2BD3sXhvV2itw%3D%3D
R. Coronado J. Morrissette M. Sukhareva D. M. Vaughan (1994) Am. J. Physiol. 266 C1485–C1504 Occurrence Handle8023884 Occurrence Handle1:CAS:528:DyaK2cXlsFeqsbs%3D
G. Meissner (1994) Annu. Rev. Physiol. 56 485–508 Occurrence Handle7516645 Occurrence Handle1:CAS:528:DyaK2cXktVSjsL0%3D
Y. Ogawa (1994) Crit. Rev. Biochem. Mol. Biol. 29 229–274 Occurrence Handle8001396 Occurrence Handle1:CAS:528:DyaK2MXhvVWhsL0%3D
I. I. Serysheva M. Schatz M. Heel Particlevan W. Chiu S. L. Hamilton (1999) Biophys. J. 77 1936–1944 Occurrence Handle10512814 Occurrence Handle1:CAS:528:DyaK1MXmtl2msLY%3D
M. R. Sharma L. H. Jeyakumar S. Fleischer T. Wagenknecht (2000) J. Biol. Chem. 275 9485–9491 Occurrence Handle10734096 Occurrence Handle1:CAS:528:DC%2BD3cXitlCru70%3D
N. Ikemoto B. Antoniu L. G. Meszaros (1985) J. Biol. Chem. 260 14096–14100 Occurrence Handle2414290 Occurrence Handle1:CAS:528:DyaL2MXlsFGqsLo%3D
T. V. McCarthy K. A. Quane P. J. Lynch (2000) Hum. Mutat. 15 410–417 Occurrence Handle10790202 Occurrence Handle1:CAS:528:DC%2BD3cXjtlylsb4%3D
W. A. Catterall M. J. Seagar M. Takahashi (1988) J. Biol. Chem. 263 3535–3538 Occurrence Handle2450086 Occurrence Handle1:CAS:528:DyaL1cXhs1egsbk%3D
S. B. Ellis M. E. Williams N. R. Ways R. Brenner A. H. Sharp A. T. Leung K. P. Campbell E. McKenna W. J. Koch A. Hui et al. (1988) Science 241 1661–1664 Occurrence Handle2458626 Occurrence Handle1:CAS:528:DyaL1MXhvVOjsb4%3D
H. Glossmann J. Striessnig (1988) Vitam. Horm. 44 155–328 Occurrence Handle10.1016/S0083-6729(08)60695-0 Occurrence Handle2853487 Occurrence Handle1:CAS:528:DyaL1MXltlShug%3D%3D
S. D. Jay S. B. Ellis A. F. McCue M. E. Williams T. S. Vedvick M. M. Harpold K. P. Campbell (1990) Science 248 490–492 Occurrence Handle2158672 Occurrence Handle1:CAS:528:DyaK3cXltFWjurg%3D
W. A. Catterall (1995) Annu. Rev. Biochem. 64 493–531 Occurrence Handle7574491 Occurrence Handle1:CAS:528:DyaK2MXmsl2qsbc%3D
L. L. Isom K. S. Jongh ParticleDe W. A. Catterall (1994) Neuron 12 1183–1194 Occurrence Handle7516685 Occurrence Handle1:CAS:528:DyaK2MXitV2quw%3D%3D
W. Melzer A. Herrmann-Frank H. C. Luttgau (1995) Biochim. Biophys. Acta 1241 59–116 Occurrence Handle10.1016/0304-4157(94)00014-5 Occurrence Handle1:CAS:528:DyaK2MXmsVahs7w%3D Occurrence Handle7742348
W. A. Catterall (2000) Annu. Rev. Cell. Dev. Biol. 16 521–555 Occurrence Handle11031246 Occurrence Handle1:CAS:528:DC%2BD3MXpvFyr
T. F. McDonald S. Pelzer W. Trautwein D. J. Pelzer (1994) Physiol. Rev. 74 365–507 Occurrence Handle8171118 Occurrence Handle1:CAS:528:DyaK2cXksl2jtLY%3D
A. T. Leung T. Imagawa B. Block C. Franzini-Armstrong K. P. Campbell (1988) J. Biol. Chem. 263 994–1001 Occurrence Handle2826471 Occurrence Handle1:CAS:528:DyaL1cXltFGksQ%3D%3D
K. Murata N. Odahara A. Kuniyasu Y. Sato H. Nakayama K. Nagayama (2001) Biochem. Biophys. Res. Commun. 282 284–291 Occurrence Handle11264005 Occurrence Handle1:CAS:528:DC%2BD3MXitFeisLc%3D
I. I. Serysheva S. J. Ludtke M. R. Baker W. Chiu S. L. Hamilton (2002) Proc. Natl. Acad. Sci. USA 99 10370–10375 Occurrence Handle12149473 Occurrence Handle1:CAS:528:DC%2BD38Xmt1CiurY%3D
M. C. Wang G. Velarde R. C. Ford N. S. Berrow A. C. Dolphin A. Kitmitto (2002) J. Mol. Biol. 323 85–98 Occurrence Handle12368101 Occurrence Handle1:CAS:528:DC%2BD38XnsVantrg%3D
M. Wolf A. Eberhart H. Glossmann J. Striessnig N. Grigorieff (2003) J. Mol. Biol. 332 171–182 Occurrence Handle12946355 Occurrence Handle1:CAS:528:DC%2BD3sXms1Oqt7c%3D
M. C. Wang R. F. Collins R. C. Ford N. S. Berrow A. C. Dolphin A. Kitmitto (2004) J. Biol. Chem. 279 7159–7168 Occurrence Handle14634003 Occurrence Handle1:CAS:528:DC%2BD2cXht1Cksb4%3D
M. Pragnell J. Sakamoto S. D. Jay K. P. Campbell (1991) FEBS Lett. 291 253–258 Occurrence Handle1657644 Occurrence Handle1:CAS:528:DyaK3sXhvVWnuw%3D%3D
M. Pragnell M. Waard Particlede Y. Mori T. Tanabe T. P. Snutch K. P. Campbell (1994) Nature 368 67–70 Occurrence Handle10.1038/368067a0 Occurrence Handle1:CAS:528:DyaK2cXis1eqsbY%3D Occurrence Handle7509046
D. Walker D. Bichet K. P. Campbell M. Waard Particlede (1998) J. Biol. Chem. 273 2361–2367 Occurrence Handle9442082 Occurrence Handle1:CAS:528:DyaK1cXnsVKguw%3D%3D
Y. Opatowsky O. Chomsky-Hecht M. G. Kang K. P. Campbell J. A. Hirsch (2003) J. Biol. Chem. 278 52323–52332 Occurrence Handle14559910 Occurrence Handle1:CAS:528:DC%2BD3sXpvFCqu78%3D
Y. Opatowsky C. C. Chen K. P. Campbell J. A. Hirsch (2004) Neuron 42 387–399 Occurrence Handle15134636 Occurrence Handle1:CAS:528:DC%2BD2cXks1Clsbk%3D
F. Petegem ParticleVan K. A. Clark F. C. Chatelain D. L. Minor SuffixJr. (2004) Nature 429 671–675 Occurrence Handle15141227
Y. H. Chen M. H. Li Y. Zhang L. L. He Y. Yamada A. Fitzmaurice Y. Shen H. Zhang L. Tong J. Yang (2004) Nature 429 675–680 Occurrence Handle15170217 Occurrence Handle1:CAS:528:DC%2BD2cXks1KlsLY%3D
S. J. Ludtke P. R. Baldwin W. Chiu (1999) J. Struct. Biol. 128 82–97 Occurrence Handle10600563 Occurrence Handle1:CAS:528:DyaK1MXnvFejsrw%3D
D. G. Ferguson H. W. Schwartz C. Franzini-Armstrong (1984) J. Cell. Biol. 99 1735–1742 Occurrence Handle6386826 Occurrence Handle1:STN:280:DyaL2M%2FjtVGrsg%3D%3D
C. Franzini-Armstrong J. W. Kish (1995) J. Muscle Res. Cell. Motil. 16 319–324 Occurrence Handle7560004 Occurrence Handle1:STN:280:BymD38vhtVc%3D
F. Protasi C. Franzini-Armstrong B. E. Flucher (1997) J. Cell. Biol. 137 859–870 Occurrence Handle9151688 Occurrence Handle1:CAS:528:DyaK2sXjtlemtr0%3D
I. Marty M. Robert M. Villaz K. S. DeJohngh Y. Lai W. A. Catterall M. Ronjat (1994) Proc. Natl. Acad. Sci. USA 91 2270–2274 Occurrence Handle8134386 Occurrence Handle1:CAS:528:DyaK2cXitlGntrk%3D
X. Lu L. Xu G. Meissner (1994) J. Biol. Chem. 269 6511–6516 Occurrence Handle8120002 Occurrence Handle1:CAS:528:DyaK2cXitlyns7w%3D
R. El-Hayek M. Yano B. Antoniu J. R. Mickelson C. F. Louis N. Ikemoto (1995) Am. J. Physiol. 268 C1381–C1386 Occurrence Handle7611356 Occurrence Handle1:CAS:528:DyaK2MXmvVGlur8%3D
T. Tanabe A. Mikami S. Numa K. G. Beam (1990) Nature 344 451–453 Occurrence Handle2157159 Occurrence Handle1:CAS:528:DyaK3cXhvFGqs7c%3D
M. Grabner R. T. Dirksen N. Suda K. G. Beam (1999) J. Biol. Chem. 274 21913–21919 Occurrence Handle10419512 Occurrence Handle1:CAS:528:DyaK1MXltVymt7c%3D
P. Leong D. H. MacLennan (1998) J. Biol. Chem. 273 29958–29964 Occurrence Handle9792715 Occurrence Handle1:CAS:528:DyaK1cXntlOjsrc%3D
K. C. Kim A. H. Caswell J. A. Talvenheimo N. R. Brandt (1990) Biochemistry 29 9281–9289 Occurrence Handle2176846 Occurrence Handle1:CAS:528:DyaK3cXlsVOntLw%3D
A. M. Corbett A. H. Caswell N. R. Brandt J. P. Brunschwig (1985) J. Membr. Biol. 86 267–276 Occurrence Handle4046012 Occurrence Handle1:CAS:528:DyaL2MXlvFWksr8%3D
J. Nakai R. T. Dirksen H. T. Nguyen I. N. Pessah K. G. Beam P. D. Allen (1996) Nature 380 72–75 Occurrence Handle8598910 Occurrence Handle1:CAS:528:DyaK28XhsFeisb8%3D
G. Avila R. T. Dirksen (2000) J. Gen. Physiol. 115 467–480 Occurrence Handle10736313 Occurrence Handle1:CAS:528:DC%2BD3cXis1emurw%3D
T. Wagenknecht C. E. Hsieh B. K. Rath S. Fleischer M. Marko (2002) Biophys. J. 83 2491–2501 Occurrence Handle12414683 Occurrence Handle1:CAS:528:DC%2BD38Xos1CrsLo%3D Occurrence Handle10.1016/S0006-3495(02)75260-0
J. Zhang Z. Liu H. Masumiya R. Wang D. Jiang F. Li T. Wagenknecht S. R. Chen (2003) J. Biol. Chem. 278 14211–14218 Occurrence Handle12576471 Occurrence Handle1:CAS:528:DC%2BD3sXjtVShsrY%3D
Z. Liu J. Zhang R. Wang S. R. Wayne Chen T. Wagenknecht (2004) J. Mol. Biol. 338 533–545 Occurrence Handle15081811 Occurrence Handle1:CAS:528:DC%2BD2cXjtVegtbo%3D
I. I. Serysheva S. L. Hamilton S. J. Ludtke W. Chiu (2004) Biophys. J. 86 242
R. Matadeen A. Patwardhan B. Gowen E. V. Orlova T. Pape M. Cuff F. Mueller R. Brimacombe M. Heel Particlevan (1999) Structure Fold. Des. 7 1575–1583 Occurrence Handle10647188 Occurrence Handle1:CAS:528:DC%2BD3cXkt1ymtQ%3D%3D
I. S. Gabashvili R. K. Agrawal C. M. Sahn R. A. Grassucci D. I. Svergun J. Frank P. Penczek (2000) Cell 100 537–549 Occurrence Handle10721991 Occurrence Handle1:CAS:528:DC%2BD3cXhvVequrw%3D
S. J. Ludtke D. H. Chen J. L. Song D. T. Chuang W. Chiu (2004) Structure (Camb.) 12 1129–1136 Occurrence Handle1:CAS:528:DC%2BD2cXlsFyrs74%3D
Author information
Authors and Affiliations
Additional information
Translated from Biokhimiya, Vol. 69, No. 11, 2004, pp. 1506–1514.
Original Russian Text Copyright © 2004 by Serysheva.
Rights and permissions
About this article
Cite this article
Serysheva, I.I. Structural insights into excitation—contraction coupling by electron cryomicroscopy. Biochemistry (Moscow) 69, 1226–1232 (2004). https://doi.org/10.1007/s10541-005-0068-5
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10541-005-0068-5