Abstract
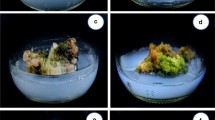
Protocols for in vitro plant multiplication from somatic tissues and production of artificial seeds through encapsulation of nodes were developed for Aristolochia tagala Cham., a rare and valuable medicinal plant, as a measure of conservation and as a prerequisite for genetic transformation procedure. A maximum number of adventitious shoots were regenerated from leaf-derived callus on Murashige and Skoog (MS) medium containing 6-benzylaminopurine (BAP; 2 μM), α-naphthaleneacetic acid (NAA; 0.5 μM), and phloroglucinol (PG; 10μM). Nodes collected from in vitro established shoot cultures were encapsulated in 3 % (m/v) sodium alginate and 1 % (m/v) calcium chloride. Multiple shoots were successfully regenerated from the encapsulated nodes cultured on MS medium supplemented with 3 μM BAP and 0.5 μM kinetin (KIN). Regenerated shoots from callus and artificial seeds were successfully rooted and acclimated to greenhouse conditions. Since roots of A. tagala are primarily used in traditional medicine, a protocol for regenerating roots directly from the leaf derived callus was also developed. Maximum root length was obtained when the callus was cultured in MS medium supplemented with KIN (1 μM), indole acetic acid (IAA; 0.5 μM), NAA (0.1 μM), and PG (10 μM). Biochemical parameters were studied in calli grown with and without PG in the medium to establish a correlation between these parameters and shoot morphogenesis. An increment of antioxidant enzymes (peroxidase and catalase) and metabolites (sugars and proteins), and a decrease in the amount of polyphenol oxidase was observed in the calli which were grown in the presence of PG.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AC:
-
activated charcoal
- BAP:
-
6-benzylaminopurine
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- IAA:
-
indole acetic acid
- IBA:
-
indolebutyric acid
- KIN:
-
kinetin
- MS:
-
Murashige and Skoog
- NAA:
-
α-naphthaleneacetic acid
- PG:
-
phloroglucinol
References
Abdelmajid, K., Annie, J.: Somatic embryogenesis, rhizogenesis, and morphinan alkaloids production in two species of opium poppy. — J. Biomed. Biotechnol. 1: 70–78, 2001.
Ahmad, N., Anis, M.: Direct plant regeneration from encapsulated nodal segments of Vitex negundo. — Biol. Plant. 54: 748–752, 2010.
Ali, A., Gull, I., Majid, A., Saleem, A., Naz, S., Naveed, N.H.: In vitro conservation and production of vigorous and desiccation tolerant synthetic seeds in Stevia rebaudiana. — J. med. Plants Res. 6: 1327–1333, 2012.
Antonietta, G.M., Emanuele, P., Alvaro, S.: Effects of encapsulation on Citrus reticulata Blanco. somatic embryo conversion. — Plant Cell Tissue Organ Cult. 55: 235–237, 1999.
Apurva, P., Thakur, P.C.: Somatic embryogenesis and root proliferation from internode of Anthocephalus cadamba in vitro. — Asian J. exp. Sci. 23: 99–102, 2009.
Ara, H., Jaiswal, U., Jaiswal, V.S.: Germination and plantlet regeneration from encapsulated somatic embryos of mango (Mangifera indica L.). — Plant Cell Rep. 19: 166–170, 1999.
Ataei-Azimi, A., Hashemloian, B.D., Ebrahimzadeh, H., Majd, A.: High in vitro production of anti-canceric indole alkaloids from periwinkle (Catharanthus roseus) tissue culture. — African J. Biotechnol. 7: 2834–2839, 2008.
Beers, R.F., Jr., Sizer, I.W.: A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. — J. biol. Chem. 195: 133–140, 1952.
Biswas, A., Bari, M.A., Mohashweta, R., Bhadra, S.K.: In vitro regeneration of Aristolochia tagala Champ. a rare medicinal plant of Chittagong hill tracts. — J. biol. Sci. 15: 63–67, 2007.
Bliss, B.J., Landherr, L., DePamphilis, C.W., Ma, H., Hu, Y., Maximova, S.N.: Regeneration and plantlet development from somatic tissues of Aristolochia fimbriata. — Plant Cell Tissue Organ Cult. 98: 105–114, 2009.
Bradford, M.: A rapid and sensitive method for the quantization of microgram quantities of protein utilizing the principle of protein-dye binding. — Anal. Biochem. 72: 248–254, 1976.
Castillo, J.A., Jordan, M.: In vitro regeneration of Minthostachya andina (Brett) Epling — a Bolivian native species with aromatic and medicinal properties. — Plant Cell Tissue Organ Cult. 49: 157–160, 1997.
Dubois, M.K.A., Gilles, J.K., Hamilton, P.K., Repers, F.: Colorimetric method for determination of sugars and related substances. — Anal. Chem. 28: 350–356, 1956.
Ganapathi, T.R., Suprasanna, P., Bapat, V.A., Rao, P.S.: Propagation of banana through encapsulated shoot tips. — Plant Cell Rep. 11: 571–575, 1992.
Gozukirmizi, Z.I.N.: Direct somatic embryogenesis and synthetic seed production from Paulownia elongata. — Plant Cell Rep. 22: 16–24, 2003.
Irwani, N., Solouki, M., Omidi, M., Zare, A.R., Shahnazi, S.: Callus induction and plant regeneration in Dorema ammoniacum, an endangered medicinal plant. — Plant Cell Tissue Organ Cult. 100: 293–299, 2010.
Jana, S., Shekhawat, G.S.: In vitro regeneration of Anethum graveolens, antioxidative enzymes during organogenesis and RAPD analysis for clonal fidelity. — Biol. Plant. 56: 9–14, 2011.
Johansen, D.A. (ed.): Plant Microtechnique. — McGraw-Hill, New York 1940.
Kim, K.M., Kim, M.Y., Yun, P.Y., Chandrasekhar, T., Lee, H.Y., Song, P.S.: Production of multiple shoots and plant regeneration from leaf segments of fig tree (Ficus carica L.). — J. Plant Biol. 50: 440–446, 2007.
Macháčková, I., Zažímalová, E., George, E.F.: Plant growth regulators I: Introduction; auxins, their analogues and inhibitors. — In: George, E.F., Hall, M.A., De Klerk, G.-J. (ed.): Plant Propagation by Tissue Culture. Vol. 1. The Background. Pp.175–204. Springer, Dordrecht 2008.
Malik, C.P., Singh, M.B. (ed.): Plant Enzymology and Histoenzymology. — Kalyani Publisher, New Delhi 1980.
Manjula, S., Thomas, A., Daniel, B., Nair, G.M.: In vitro plant regeneration of Aristolochia indica through axillary shoot multiplication and organogenesis. — Plant Cell Tissue Organ Cult. 51:145–148, 1997.
Mitrović, A., Bogdanović, J.: Activities of antioxidative enzymes during Chenopodium rubrum L. ontogenesis in vitro. — Arch. biol. Sci. 60: 223–231, 2008.
Mitrović, A., Janošević, D., Budimir, S., Bogdanović Pristov, J.: Changes in antioxidative enzymes activities during Tacitus bellus direct shoot organogenesis. — Biol. Plant. 56: 357–361, 2012.
Mohanraj, R., Ananthan, R., Bai, V.N.: Production and storage of Coelogyne breviscapa Lindl. — Asian J. Biotech. 1: 124–128, 2009.
Murashige, T., Skoog, F.: A revised medium for rapid growth and bioassays with tobacco tissue culture. — Physiol Plant. 15: 473–497, 1962.
Murugan, R., Shivanna, K.R., Rao, R.R.: Pollination biology of Aristolochia tagala, a rare species of medicinal importance. — Curr. Sci. 91: 795–798, 2006.
Nelson, N.: A photometric adaptation of the Somogyi method for the determination of glucose. — J. biol. Chem. 153: 375–380, 1944.
Németh, G.: Induction of rooting. — In: Bajaj, Y.P.S. (ed.): Biotechnology in Agriculture and Forestry. Vol. 1. Pp. 49–64. Springer-Verlag, Berlin 1986.
Novak, F.J.: Musa (bananas and plantains). — In: Hammerschlag, F.A., Litz, R.E. (ed.). Biotechnology of Perennial Fruit Crops. Pp. 449–488. CAB International, Wallingford 1992.
Patil, C.S., Gangawane, A.K., Anjali, R., Naik, G.R.: Organic constituents in the different stages of regenerated callus of sugarcane Var Co-740. — Int. J. mol. Biol. 1: 15–20, 2010.
Pospíšilová, J., Wilhelmová, N., Synková, H., Čatský, J., Krebs, D., Tichá, I., Hanáčková, B., Snopek, J.: Acclimation of tobacco plantlets to ex vitro conditions as affected by application of abscisic acid. — J. exp. Bot. 49: 863–869, 1998.
Rai, M.K., Jaiswal, V.S., Jaiswal, U.: Encapsulation of shoot tips of guava (Psidium guajava L.) for short-term storage and germplasm exchange. — Sci. Hort. 118: 33–38, 2008.
Rajeshwari, V., Paliwal, K.: Peroxidase and catalase changes during in vitro adventitious shoot organogenesis from hypocotyls of Albizia odoratissima L.f. (Benth). — Acta Physiol. Plant. 30: 825–832, 2008.
Ravikumar, K., Ved, D.K. (ed.): 100 Red Listed Medicinal Plants of Conservation Concern in Southern India. — Foundation for Revitalization of Local Health Traditions, Bangalore 2000.
Redenbaugh, K., Slade, D., Viss, P., Fujii, J.A.: Encapsulation of somatic embryos in synthetic seed coats. — Hort. Sci. 22: 803–809, 1987.
Remeshree, A.B., Hariharan, M., Unnikrishnan, K.: In vitro organogenesis in Aristolochia indica. — Phytomorphology 47: 161–165, 1997.
Remeshree, A.B., Hariharan, M., Unnikrishanan, K.: Micropropagation and callus induction of Aristolochia bracteolata Lam. a medicinal plant. — Phytomorphology 44: 247–252, 1994.
Růžička, K., Simaskova, M., Duclercq, J., Petrasek, J., Zazimalova, E., Simon, S., Friml, J., Van Montagu M.C.E., Benková, E.: Cytokinin regulates root meristem activity via modulation of the polar auxin transport. — Proc. nat. Acad. Sci. USA 106: 4284–4289, 2009.
Sáez, P.L., Bravo, L.A., Sáez, K.L., Sánchez-Olate, M., Latsague, M.I., Ríos, D.G.: Photosynthetic and leaf anatomical characteristics of Castanea sativa: a comparison between in vitro and nursery plants. — Biol. Plant. 56: 15–24, 2012.
Sankar, A., Libin, M.S., Vijayakumar, A., Karthi Rani, R., Raja Selvam, J., Kohila, R., Liby, I., Vadivukarasi, S., Ganesh, D.: Phloroglucinol enhances shoot proliferation in nodal explants of Vanilla planifoloia Andr. — J. Plantation Crops 36: 127–129, 2008.
Sarkar, D., Naik, P.S.: Phloroglucinol enhances growth and rate of axillary shoot proliferation in potato shoot tip cultures in vitro. — Plant Cell Tissue Organ Cult. 60: 139–149, 2000.
Sarvesh, A., Reddy, T.P.: Peroxidase, polyphenol oxidase, acid phosphatase and alkaline inorganic pyrophosphatase activities during leaf senescence in varieties of castor (Ricinus communis L.). — Indian J. exp. Biol. 26: 133–136, 1988.
Shasthree, T., Imran, M.A., Mallaiah, B.: In vitro rooting from callus cultures derived from seedling explants of Erythrina variegate L. — Curr. Trends Biotechnol. Pharmacol. 3: 447–452, 2009.
Singh, S.K., Manoj, K., Pooja A., Sahoo, L.: Alginateencapsulation of nodal segments for propagation, short-term conservation and germplasm exchange and distribution of Eclipta alba (L.). — Acta Physiol. Plant. 32: 607–610, 2010.
Soneji, J.R., Rao, P.S., Mhatre, M.: Germination of synthetic seeds of pineapple (Ananas comosus L. Merr.). — Plant Cell Rep. 20: 891–894, 2002.
Soniya, E.V., Sujitha, M.: An efficient in vitro propagation of Aristolochia indica. — Biol. Plant. 50: 272–274, 2006.
Steephen, M., Nagarajan, S., Ganesh, D.: Phloroglucinol and silver nitrate enhances axillary shoot proliferation in nodal explants of Vitex negundo L. — an aromatic medicinal plant. — Iranian J. Biotechnol. 8: 82–89, 2010.
Su, Y.H., Liu, Y.B., Zhang, X.S.: Auxin-cytokinin interaction regulates meristem development. — Mol. Plant. 4: 1–11, 2011.
Tang, W., Newton, R.J.: Regulated gene expression by glucocorticoids in cultured Virginia pine (Pinus virginiana Mill.) cells. — J. exp. Bot. 55: 1499–1508, 2004.
Tang, W., Newton, R.J.: Peroxidase and catalase activities are involved in direct adventitious shoot formation induced by thidiazuron in eastern white pine (Pinus strobus L.) zygotic embryos. — Plant Physiol. Biochem. 43: 760–769, 2005.
Te-Chato, S., Lim, M.: Plant regeneration of mangosteen via nodular callus formation. — Plant Cell Tissue Organ Cult. 59: 89–93, 1999.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: The authors express their sincere thanks to Dr. R. Gopalan, Botanical Survey of India, Coimbatore, Tamil Nadu and Dr. Venkata Subramaniam, Institute Of Forest Genetics and Tree Breeding, Coimbatore, Tamil Nadu, for their valuable assistance in collection and identification of the plant.
Rights and permissions
About this article
Cite this article
Remya, M., Narmatha Bai, V. & Mutharaian, V.N. In vitro regeneration of Aristolochia tagala and production of artificial seeds. Biol Plant 57, 210–218 (2013). https://doi.org/10.1007/s10535-012-0280-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-012-0280-2