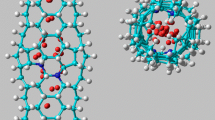
Abstract
Recently there has been lot of interest in the development of hydrogen storage in various systems for the large-scale application of fuel cells, mobiles and for automotive uses. Hectic materials research is going on throughout the world with various adsorption mechanisms to increase the storage capacity. It was observed that physisorption proves to be an effective way for this purpose. Some of the materials in this race include graphite, zeolite, carbon fibers and nanotubes. Among all these, the versatile material carbon nanotube (CNT) has a number of favorable points like porous nature, high surface area, hollowness, high stability and light weight, which facilitate the hydrogen adsorption in both outer and inner portions. In this work we have considered armchair (5,5), zig zag (10,0) and chiral tubes (8,2) and (6,4) with and without structural defects to study the physisorption of hydrogen on the surface of carbon nanotubes using DFT calculations. For two different H2 configurations, adsorption binding energies are estimated both for defect free and defected carbon nanotubes. We could observe larger adsorption energies for the configuration in which the hydrogen molecular axis perpendicular to the hexagonal carbon ring than for parallel to C–C bond configuration corresponding to the defect free nanotubes. For defected tubes the adsorption energies are calculated for various configurations such as molecular axis perpendicular to a defect site octagon and parallel to C–C bond of octagon and another case where the axis perpendicular to hexagon in defected tube. The adsorption binding energy values are compared with defect free case. The results are discussed in detail for hydrogen storage applications.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CNT:
-
Carbon Nanotube
- DFT:
-
Density Functional Theory
- SWCNTs:
-
Single Walled Carbon Nanotubes
- H2 :
-
Hydrogen molecule
- H:
-
Hydrogen atom
- E ads :
-
Adsorption binding energy
- 1 Å:
-
=10−10 m
- 1 eV:
-
=1.602×10−19 J
- O:
-
Octagon
- P:
-
Pentagon
References
Alonso, J.A., et al.: Interaction of molecular and atomic hydrogen with single-wall carbon nanotubes. IEEE Trans. Nanotechnol. 3, 304–310 (2004)
Arellano, J.S., et al.: Interaction of molecular and atomic hydrogen with (5,5) and (6,6) single-wall carbon nanotubes. J. Chem. Phys. 117, 2281–2288 (2002)
Cracknell, R.F.: Simulation of hydrogen adsorption in carbon nanotubes. Mol. Phys. 100, 2079–2086 (2002)
Dillon, A.C., et al.: Storage of hydrogen in single-walled carbon nanotubes. Nature 386, 377–379 (1997)
Gulseren, O., et al.: Tunable adsorption on carbon nanotubes. Phys. Rev. Lett. 87, 116802 (2001)
Jhi, S.-H., Kwon, Y.-K.: Hydrogen adsorption on boron nitride nanotubes: a path to room-temperature hydrogen storage. Phys. Rev. B 69, 245407 (2004)
Krasheninnikov, A.V., et al.: Adsorption and migration of carbon adatoms on zigzag carbon nanotubes. Carbon 42, 1021–1025 (2004)
Pradhan, B.K., et al.: Experimental probes of the molecular hydrogen-carbon nanotube interaction. Phys. B 323, 115–121 (2002a)
Pradhan, B.K., et al.: Large cryogenic storage of hydrogen in carbon nanotubes at low pressures. J. Mater. Res. 17, 2209–2216 (2002b)
Poirier, E., et al.: Storage of hydrogen on single-walled carbon nanotubes and other carbon structures. Appl. Phys. A Mater. Sci. Process. 78, 961–967 (2004)
Sinnot, S.B., et al.: Carbon nanotubes synthesis, properties and applications. In: Critical Review in Solid State and Materials Science, p. 26 (2001)
Tada, K., et al.: Ab initio study of hydrogen adsorption to single-walled carbon nanotubes. Phys. Rev. B 63, 155405 (2001)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gayathri, V., Geetha, R. Hydrogen adsorption in defected carbon nanotubes. Adsorption 13, 53–59 (2007). https://doi.org/10.1007/s10450-007-9002-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-007-9002-z