Abstract
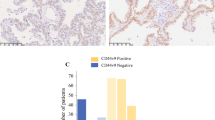
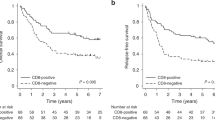
CD44 and CD44 variant isoforms have been reported as contributing factors to cancer progression. In this study, we aimed to assess whether CD44 and its variant isoforms were correlated with the prognostic factors for distant metastasis in stage I lung adenocarcinomas using tissue microarray and immunohistochemistry. In this single-center retrospective study, we analyzed the data of 490 patients with stage I lung adenocarcinoma resected between 1999 and 2016. We constructed tissue microarrays and performed immunohistochemistry for CD44s, CD44v6, and CD44v9. The risk of disease recurrence and its associations with clinicopathological risk factors were assessed. CD44v6 expression was significantly associated with recurrence. Patients with CD44v6-negative tumors had a significantly increased risk of developing distant recurrence than patients with CD44v6-positive tumors (5-year cumulative incidence of recurrence (CIR), 10.7% vs. 4.6%; P = 0.009). However, CD44v6-negative tumors were not associated with an increased risk of locoregional recurrence compared to CD44v6-positive tumors (5-year CIR, 6.0% vs. 4.0%; P = 0.39). The overall survival (OS) of patients with CD44v6-negative tumors was significantly lower than that of patients with CD44v6-positive tumors (5-year OS: 87% vs. 94%, P = 0.016). CD44v6-negative tumors were also associated with invasive tumor size and lymphovascular invasion. Even in stage I disease, tumors with negative-CD44v6 expression had more distant recurrences than those with positive-CD44v6 expression and were associated with poor prognosis in resected stage I lung adenocarcinomas. Thus, CD44v6 downregulation may be a prognostic factor for distant metastasis in stage I lung adenocarcinomas.




Similar content being viewed by others
Data availability
We confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr, Kinzler KW. Cancer genome landscapes. Science. 2013;339:1546–58. https://doi.org/10.1126/science.1235122.
Sasaki H, Suzuki A, Tatematsu T, et al. Prognosis of recurrent non-small cell lung cancer following complete resection. Oncol Lett. 2014;7:1300–4. https://doi.org/10.3892/ol.2014.1861.
Ekeke CN, Mitchell C, Schuchert M, Dhupar R, Luketich JD, Okusanya OT. Early distant recurrence in patients with resected stage I lung cancer: A case series of “Blast Metastasis.” Clin Lung Cancer. 2021;22:e132–5. https://doi.org/10.1016/j.cllc.2020.09.002.
Wang Z, Zhao K, Hackert T, Zöller M. CD44/CD44v6 a reliable companion in cancer-initiating cell maintenance and tumor progression. Front Cell Dev Biol. 2018;6:97. https://doi.org/10.3389/fcell.2018.00097.
Tjhay F, Motohara T, Tayama S, et al. CD44 variant 6 is correlated with peritoneal dissemination and poor prognosis in patients with advanced epithelial ovarian cancer. Cancer Sci. 2015;106:1421–8. https://doi.org/10.1111/cas.12765.
Yu P, Zhou L, Ke W, Li K. Clinical significance of pAKT and CD44v6 overexpression with breast cancer. J Cancer Res Clin Oncol. 2010;136:1283–92. https://doi.org/10.1007/s00432-010-0779-x.
Coppola D, Hyacinthe M, Fu L, et al. CD44V6 expression in human colorectal carcinoma. Hum Pathol. 1998;29:627–35. https://doi.org/10.1016/s0046-8177(98)80014-2.
Avoranta ST, Korkeila EA, Syrjänen KJ, Pyrhönen SO, Sundström JT. Lack of CD44 variant 6 expression in rectal cancer invasive front associates with early recurrence. World J Gastroenterol. 2012;18:4549–56. https://doi.org/10.3748/wjg.v18.i33.4549.
Afify A, Durbin-Johnson B, Virdi A, Jess H. The expression of CD44v6 in colon: from normal to malignant. Ann Diagn Pathol. 2016;20:19–23. https://doi.org/10.1016/j.anndiagpath.2015.10.010.
Nitadori J, Bograd AJ, Kadota K, et al. Impact of micropapillary histologic subtype in selecting limited resection vs lobectomy for lung adenocarcinoma of 2 cm or smaller. J Natl Cancer Inst. 2013;105:1212–20. https://doi.org/10.1093/jnci/djt166.
Donington J, Ferguson M, Mazzone P, et al. American college of chest physicians and society of thoracic surgeons consensus statement for evaluation and management for high-risk patients with stage I non-small cell lung cancer. Chest. 2012;142:1620–35. https://doi.org/10.1378/chest.12-0790.
Amin MB, Edge S, Greene F, et al. AJCC cancer staging manual. 8th ed. New Yok: Springer; 2017. p. 431–56.
Nicholson AG, Tsao MS, Beasley MB, et al. The 2021 WHO classification of lung tumors: impact of advances since 2015. J Thorac Oncol. 2022;17:362–87. https://doi.org/10.1016/j.jtho.2021.11.003.
Chappell R. Competing risk analyses: how are they different and why should you care? Clin Cancer Res. 2012;18:2127–9. https://doi.org/10.1158/1078-0432.CCR-12-0455.
Dignam JJ, Zhang Q, Kocherginsky M. The use and interpretation of competing risks regression models. Clin Cancer Res. 2012;18:2301–8. https://doi.org/10.1158/1078-0432.CCR-11-2097.
Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1998;16:1141–54.
Naor D, Sionov RV, Ish-Shalom D. CD44: structure, function, and association with the malignant process. Adv Cancer Res. 1997;71:241–319. https://doi.org/10.1016/s0065-230x(08)60101-3.
Brown RL, Reinke LM, Damerow MS, et al. CD44 splice isoform switching in human and mouse epithelium is essential for epithelial-mesenchymal transition and breast cancer progression. J Clin Invest. 2011;121:1064–74. https://doi.org/10.1172/JCI44540.
Negi LM, Talegaonkar S, Jaggi M, Ahmad FJ, Iqbal Z, Khar RK. Role of CD44 in tumour progression and strategies for targeting. J Drug Target. 2012;20:561–73. https://doi.org/10.3109/1061186X.2012.702767.
Sacks JD, Barbolina MV. Expression and function of CD44 in epithelial ovarian carcinoma. Biomolecules. 2015;5:3051–66. https://doi.org/10.3390/biom5043051.
Xu H, Niu M, Yuan X, Wu K, Liu A. CD44 as a tumor biomarker and therapeutic target. Exp Hematol Oncol. 2020;9:36. https://doi.org/10.1186/s40164-020-00192-0.
Zhao S, Chen C, Chang K, et al. CD44 expression level and isoform contributes to pancreatic cancer cell plasticity, invasiveness, and response to therapy. Clin Cancer Res. 2016;22:5592–604. https://doi.org/10.1158/1078-0432.CCR-15-3115.
Saito S, Okabe H, Watanabe M, et al. CD44v6 expression is related to mesenchymal phenotype and poor prognosis in patients with colorectal cancer. Oncol Rep. 2013;29:1570–8. https://doi.org/10.3892/or.2013.2273.
Tuncay Cagatay S, Cimen I, Savas B, Banerjee S. MTA-1 expression is associated with metastasis and epithelial to mesenchymal transition in colorectal cancer cells. Tumour Biol. 2013;34:1189–204. https://doi.org/10.1007/s13277-013-0662-x.
Chen C, Zhao S, Karnad A, Freeman JW. The biology and role of CD44 in cancer progression: therapeutic implications. J Hematol Oncol. 2018;11:64. https://doi.org/10.1186/s13045-018-0605-5.
Todaro M, Gaggianesi M, Catalano V, et al. CD44v6 is a marker of constitutive and reprogrammed cancer stem cells driving colon cancer metastasis. Cell Stem Cell. 2014;14:342–56. https://doi.org/10.1016/j.stem.2014.01.009.
Zhao LH, Lin QL, Wei J, Huai YL, Wang KJ, Yan HY. CD44v6 expression in patients with stage II or stage III sporadic colorectal cancer is superior to CD44 expression for predicting progression. Int J Clin Exp Pathol. 2015;8:692–701.
Wang L, Liu Q, Lin D, Lai M. CD44v6 down-regulation is an independent prognostic factor for poor outcome of colorectal carcinoma. Int J Clin Exp Pathol. 2015;8:14283–93.
Zlobec I, Günthert U, Tornillo L, et al. Systematic assessment of the prognostic impact of membranous CD44v6 protein expression in colorectal cancer. Histopathology. 2009;55:564–75. https://doi.org/10.1111/j.1365-2559.2009.03421.x.
Kataki A, Giannakoulis VG, Derventzi A, Papiris K, Koniaris E, Konstadoulakis M. Membranous CD44v6 is upregulated as an early event in colorectal cancer: downregulation is associated with circulating tumor cells and poor prognosis. Oncol Lett. 2021;22:820. https://doi.org/10.3892/ol.2021.13081.
Jones DR, Daniel TM, Denlinger CE, Rundall BK, Smolkin ME, Wick MR. Stage IB nonsmall cell lung cancers: are they all the same? Ann Thorac Surg. 2006;81:1958–62. https://doi.org/10.1016/j.athoracsur.2005.12.054.
Hung JJ, Jeng WJ, Hsu WH, et al. Prognostic factors of postrecurrence survival in completely resected stage I non-small cell lung cancer with distant metastasis. Thorax. 2010;65:241–5. https://doi.org/10.1136/thx.2008.110825.
Hung JJ, Jeng WJ, Hsu WH, Chou TY, Huang BS, Wu YC. Predictors of death, local recurrence, and distant metastasis in completely resected pathological stage-I non-small-cell lung cancer. J Thorac Oncol. 2012;7:1115–23. https://doi.org/10.1097/JTO.0b013e31824cbad8.
Ginsberg RJ, Rubinstein LV. Randomized trial of lobectomy versus limited resection for T1 N0 non-small cell lung cancer. Lung cancer study group. Ann Thorac Surg. 1995;60:615–22. https://doi.org/10.1016/0003-4975(95)00537-u.
Wang C, Wu Y, Shao J, Liu D, Li W. Clinicopathological variables influencing overall survival, recurrence and post-recurrence survival in resected stage I non-small-cell lung cancer. BMC Cancer. 2020;20:150. https://doi.org/10.1186/s12885-020-6621-1.
Funding
This work was supported, in part, by JSPS KAKENHI (G. No. JP20K07392).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. C.Y. was involved in conceptualization, formal analysis, investigation, visualization, writing—original draft preparation, and editing. K.K. helped in funding acquisition, methodology, and writing—reviewing. K.Y. contributed to data curation. S.F. was involved in data curation. E.I. and R.I. helped in resources. R.H. contributed to project administration. T.Y. helped in supervision.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was approved by the Institutional Review Board of Kagawa University in Japan.
Consent to participate
The requirement for informed consent was waived owing to the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshida, C., Kadota, K., Yamada, K. et al. CD44v6 downregulation as a prognostic factor for distant recurrence in resected stage I lung adenocarcinomas. Clin Exp Med 23, 5191–5200 (2023). https://doi.org/10.1007/s10238-023-01185-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-01185-z