Abstract
In recent years, many researchers have focused on the development of comorbidities associated with axial spondyloarthritis. Hypertension, smoking, dyslipidemia, diabetes mellitus, obesity, and the presence of inflammation, which is a well-known risk factor, increase cardiovascular comorbidities. Previous studies reported that HLA-B27 might be a genetic risk factor for various cardiac conditions. In the treatment and follow-up, raising the awareness of cardiovascular risk and guiding clinical practice and future research in rheumatic and musculoskeletal diseases should be aimed. There is consensus that cardiovascular risks should be evaluated periodically in these patients, particularly during the course of treatment. A decision regarding the selection and follow-up protocols of the drugs should be made considering all the risks and benefits in spondyloarthropathies. In this review, we discussed the recommendations and practices for the management of cardiovascular comorbidities in axial spondyloarthritis.
Key Points • The risk of cardiovascular morbidity and mortality is increased in axial spondyloarthritis. • Higher disease activity increases cardiovascular risk factors. • Axial spondyloarthritis patients should be carefully monitored for signs of cardiovascular events as this is necessary to ensure that patients are treated appropriately and awareness should be raised in all patients. • Reducing the activity of the underlying disease with an appropriate and early treatment will also reduce the risk of cardiovascular events. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spondyloarthritis (SpA), with human leukocyte antigen-B27 (HLA-B27) as the major contributing gene, is a rheumatic disease characterized by inflammatory changes in the axial skeletal system, peripheral joints, and the areas of the enthesis. Acute anterior uveitis, psoriasis, and inflammatory bowel disease are the typical extra-articular manifestations that coexisted with SpA. There are several interrelated subgroups of SpA according to traditional categories which are ankylosing spondylitis (AS), reactive arthritis, psoriatic arthritis (PsA), inflammatory bowel disease-related arthritis, and undifferentiated SpA (uSpA). Nevertheless, the current approach uses two major subclasses, predominantly axial SpA (axSpA) and peripheral SpA. Non-radiographic axial SpA (nr-axSpA) and radiographic axial SpA (r-axSpA) have been regarded under the covering term of axSpA.
SpA prevalence, including all subgroups, ranges from 0.3 to 1.9% worldwide. Although AS is the largest subgroup, the SpA prevalence is closely associated with the frequency of HLA-B27 in general population [1]. AxSpA, precisely AS, has been accepted as the most popular prototype and frequent SpA. In patients with SpA, it is not surprising to see several extra-musculoskeletal comorbidities. Several large-scale meta-analyses reported that hypertension, diabetes mellitus, cardiac diseases, depression, and fibromyalgia are the most common comorbidities in AS patients. The frequency and risk factors for cardiovascular (CV) comorbidities have been assessed in various studies [2, 3]. In a cross-sectional study comparing comorbidity rates in AS and nr-axSpA patients, the overall burden and frequency of such comorbidities were similar. However, the patients with nr-axSpA were significantly younger compared to AS [4].
There have been significant improvements in the early diagnosis, and early and active treatment modalities of axSpA recently. Nevertheless, short life expectancy and increased mortality rates have been observed due to rheumatic diseases. Data on mortality mainly originated from the studies including AS patients, contrary to the limited data about the mortality rates of the nr-axSpA. It is generally known that the survival in patients with AS is shorter than in the overall population. Based on the previously published studies, the standardized mortality ratio ranged from 1.6 to 1.9. Duration of the disease and its severity, increased acute phase reactants, delayed diagnosis, and comorbidities have been reported as mortality predictors in patients with AS [5].
The Assessment in SpondyloArthritis International Society (ASAS) COMOrbidities in SPondyloArthritis (COMOSPA) has been initiated to evaluate the possible risk factors, prevalence rates, and management strategies for comorbidities in this patient group. The first study published in 2016 showed a high prevalence rate of comorbidities in axSpA patients; osteoporosis (13%) and gastroduodenal ulcer (11%) were the most frequent comorbidities. It has been reported that hypertension, smoking, and hypercholesterolemia were the most frequent risk factors detected in 34%, 29%, and 27% of the patients. As shown in the ASAS-COMOSPA study, CV comorbidities, like myocardial infarction (MI) (2.7%) and stroke (1.3%), were frequently observed as well as the traditional risk factors [6].
In another retrospective registry-based study in which all-cause and cause-specific mortality rates were performed, the all-cause mortality rates were 1.45 and 1.06 for rheumatoid arthritis (RA) and PsA. Besides, the hazard ratio (HR) of all-cause mortality was 1.38. Circulatory diseases, malignancies, and respiratory problems were the three most evident causes of mortality. Others reported that the HRs due to circulatory diseases were 1.41 (RA), 1.13 (PsA), and 1.4 (axSpA) [7].
In light of these data, The European Alliance of Associations for Rheumatology (EULAR) published recommendations for the CV risk management of patients with rheumatoid arthritis (RA) and other inflammatory joint disorders. The guidelines, which have been updated recently, aimed to increase awareness of CV risk in these patients’ treatment and follow-up and guide clinical practice and future research in rheumatic and musculoskeletal diseases [8, 9].
The importance of comorbidities in the management of axSpA has been emphasized in the ASAS EULAR Recommendations. The first recommendation was related to the individualized treatment modalities that should be tailored to the current signs and symptoms of the disease, including the axial, peripheral, and extra-articular manifestations and the clinical characteristics, such as comorbidities and psychosocial factors [10].
The aim of this narrative review is to summarize cardiovascular comorbidity and risk factors in axSpA patients and to analyze the relationship of cardiovascular events with HLA-B27, disease activity, and medications used in these patients.
Statement of literature search
PubMed, EMBASE, and Scopus for English-language sources published in the last 10 years were searched. Key words and search terms used to compose this narrative review were “ankylosing spondylitis or axial spondyloarthritis or spondyloarthritis or inflammatory joint diseases or inflammatory rheumatic diseases” and “cardiovascular diseases or cardiovascular risk or cardiovascular morbidity or cardiovascular event or cardiac function or hypertension or acute coronary syndrome or atherosclerotic disease or comorbidities”. This review is based on previously completed studies, and results specified in preferred articles have been summarized.
Cardiovascular morbidity and related risk factors
It has been shown that patients with SpA had significantly higher MI and stroke risk ratios (RR) than the general population in a recent meta-analysis investigating CV morbidity and mortality of AS, PsA, and uSpA in a large-scale population which were 1.52 and 1.21 respectively. In this meta-analysis, the all-cause mortality RR was 1.23. Therefore, the increased risk associated with MI and stroke was insignificant considering the RR of all-cause mortality. This controversy indicates that there should be a need for CV risk factor correction and disease management in patients with SpA [11].
There are various methodological limitations of most epidemiological studies focusing on the cardiovascular disease (CVD) risk in patients with chronic inflammatory rheumatic diseases. The retrospective nature of these studies is the main limiting factor for the widespread acceptance of this evidence. To overcome such methodological problems, the CARdiovascular in rheuMAtology (CARMA) project performed in Spain evaluated CV risk in this patient group prospectively. In this study, 89.1% of the patients (n = 2595) completed a follow-up time of 2.5 years. The cumulative incidence of cardiovascular events (CVE) in patients with the chronic inflammatory rheumatic disease was 15.30 cases per 1000 patients. The incidence was 22.03 cases per 1000 patients with AS, which was higher than the overall patient group. They found that AS patients had the highest risk for the first attack of CVE, with an HR of 4.11 during the 2.5-year follow-up. Besides AS (HR: 4.11), older age (HR: 1.09), systolic hypertension (HR: 1.02), and long duration of the disease (HR: 1.07) were the other independent risk factors for the development of the first CVE during their follow-up time. The female gender was a protective factor with an HR:0.43 compared to male gender [12]. In the 5-year follow-up of the CARMA project, CV mortality and CVE have been evaluated according to different subgroups of chronic inflammatory rheumatic diseases. A total of 2382 patients completed the 5-year follow-up time, and the highest incidence of CVE was detected in PsA patients, with an incidence rate of 7.39 cases per 1000 person-years. The adjustment for age, sex, and CV risk factors revealed that AS was associated more frequently with CVE (HR:4.60 at 5 years) during the 5-year follow-up compared to the patients without chronic inflammatory rheumatic diseases. [13].
In another prospective, population-based cohort study, the first-time risk for acute coronary syndrome (ACS), stroke, and venous thromboembolism (VTE) in patients with AS (n = 6448), PsA (n = 16,063), and uSpA (n = 5190) was evaluated and compared with general population (GP, n = 266,435). They reported that the age-and sex-adjusted HRs for ACS significantly increased in patients with AS (HR:1.54), PsA (HR:1.76), and uSpA (HR:1.36) than in the GP. Based on the findings of this nationwide, prospective population-based cohort study, the authors concluded that there was a significantly increased risk, ranging from 36 to 76% for ACS and 50% for VTE in all subtypes of SpA patients compared to the GP. So, the CV risk factors for AS, PsA, and uSpA patients should be described in detail, considering their higher risks for the development of ACS, stroke, and VTE. Besides, the importance of the CV risk management algorithms should be stressed [14]. These findings are also in accordance with the new EULAR recommendations for CVD risk management which recommend that CVD risk assessment should be performed in AS and PsA patients at least once every 5 years [8].
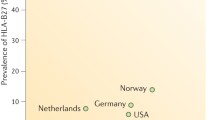
As a part of the ASAS-COMOSPA project, the researchers found that smoking was the most frequent CV risk factor seen in 31.2% of the cohort originating from countries in northern Europe and the Mediterranean region. In the same study, ischemic heart disease (3.2%) was the most common CVD, with a prevalence rate of 3.2%. Hypertension was seen in 19.2%, 33.8%, and 26.6% of the patients with axial, peripheral, and mixed phenotypes of SpA, respectively. There were significant incidence rates for type 2 diabetes mellitus (DM) (4.3%, 8.5%, and 7.4%), dyslipidemia (13.9%, 28.4%, and 15.2%), and ischemic heart disease (2.4%, 7.0%, and 3.2%) in patients with axial, peripheral, and mixed phenotypes of SpA, respectively. The patients from the North European countries had significantly higher frequencies of hypertension (34.7% vs. 19.4%), dyslipidemia (19.3% vs. 14.4%), obesity (29.3% vs. 20.7%), and ischemic heart disease (6.2% vs. 1.8%) than those from the Mediterranean region. The ASAS-COMOSPA cohort findings suggest that differences in the SpA phenotype and geographical area are associated with the higher prevalence rates of CV risk factors and the CV risk itself [15].
The National Biologic and targeted Synthetic anti-rheumatic drugs Registry (BioStaR) revealed that the hypertension and DM prevalence rates were 13.4% and 6.7% (n = 1242). They also reported that smoking and body mass index higher than 25 kg/m2 were observed in 52.2% and 66.9% of the patients [16].
Relationship between cardiovascular risk and disease activity
In CVD, besides the non-modifiable risk factors such as age, sex, and family history, there are five modifiable classical risk factors: hypertension, smoking, dyslipidemia, diabetes mellitus, and obesity. Inflammation is a well-known risk factor for the increased CV risk in rheumatic disorders. It also accelerates atherosclerosis. Co-occurrence of the traditional risk factors and inflammation is directly bound with an underlying common inflammatory mechanism. In a cross-sectional study, 804 patients with axSpA were evaluated for the presence of five traditional CV risk factors, and the impact of the number of CV risk factors on the disease severity was analyzed. The multivariate analysis showed that the patients with one (beta coefficient: 0.3) and two or more (beta coefficient: 0.5) CV risk factors had significantly higher Ankylosing Spondylitis Disease Activity Score-C reactive protein (ASDAS-CRP) activity scores than those without CV risk factors. Besides, as the number of CV risk factors increased, the disease activity scores also increased. A significant association between the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and the number of CV risk factors was found. Based on these findings, the authors concluded that the disease activity is affected by the number of CV risk factors, and the effect of two or more CV risk factors over one CV risk factor has been directly related to the higher beta coefficients and odds ratios. Previous studies have shown a significant difference in ASDAS-CRP scores between patients with one or two CV risk factors and those without CV risk factors. The disease activity scores were significantly higher in patients with CV risk factors. Similarly, higher odds ratios in patients with one, two, or more risk factors were associated with higher disease activity than in patients without CV risk. It has also been shown that higher BASDAI scores were significantly associated with a higher number of CV risk factors. In that way, any reduction in the rates of CVD and CVE and improvement in the disease activity scores can be succeeded. However, the reciprocal relationships between CV risk factors and inflammation warrant further research [17].
In a large multicenter retrospective cohort study, the authors investigated the predictive power of increased CRP values and higher disease activity scores on the development of CVD. They analyzed the impact of persisting increased CRP levels, ASDAS score of > 2.1, and BASDAI score > 4 on the occurrence of fatal and non-fatal CVE the during the 10-year follow-up period. After adjustment for age, sex, and diabetes mellitus, a strong association was found between CVE and the persistency of increased CRP levels (HR:1.03), ASDAS score of > 2.1 (HR: 1.01), BASDAI score of > 4 (HR:1.019). Based on these findings, the authors suggested that persistency of increased CRP levels and high disease activity may be considered biomarkers in identifying the axSpA patients at higher risk of CVD. However, future studies are needed to develop the innovative axSpA-specific CV risk score, including CRP, ASDAS, and BASDAI [18].
Inflammatory cardiac disease associated with axSpA
Previous studies suggest a solid immuno-genetic link between spondyloarthropathies, especially AS, and HLA-B27. Besides, the associated inflammatory processes might be a causative factor for rheumatic diseases and may also target cardiac function. Various factors are involved in the increased risk of CVD in AS: inflammation that accelerates atherosclerosis and cardiac inflammation in the form of aortitis and conduction anomalies. Aortic insufficiency and aortitis, conduction abnormalities, and myocardial involvement affecting the left ventricular ejection fraction are the three types of inflammatory cardiac diseases associated with AS. Its pathophysiological mechanism includes the obliterative (occlusive) endarteritis of the small blood vessels that supply the atrioventricular (AV) node and the aortic root. These changes lead to the development of AV block, arrhythmias, and aortic valve insufficiency. An aortic regurgitation murmur is an outcome in these patients. There is a similarity between the involvement of the affected joints and the development of AV block and aortic valve insufficiency in AS patients. In AS patients with severe AV block or aortic valve insufficiency, pacemaker placement for rhythm control or repair or replacement of the valve may be needed [19].
It has been reported that electrocardiographic abnormalities are common findings in patients with AS. In a cross-sectional study, the prevalence rates of cardiac conduction system abnormalities showed variations depending on the criteria that the authors used. In applying the conservative criteria, the rate was 10%, whereas the less conservative criteria led to a higher prevalence rate of 33%. First-degree AV block and prolonged QRS duration were the most frequent arrythmias; however, age, male gender, and body weight were significantly associated with these cardiac conduction abnormalities. There was no significant relationship between the measures of disease activity, functional limitations, and conductive cardiac problems [20].
Previous studies compared the rates of atrial fibrillation, pacemaker implantation, and aortic regurgitation between patients with spondyloarthritis and the general population. A prospective nationwide study analyzed the HRs of AV block, atrial fibrillation, implanted pacemaker, and aortic regurgitation in patients with different rheumatic diseases; the number of participants was 266,435 and AS (n = 6448), PsA (n = 16,063), and uSpA (n = 5,190). They showed that the HRs for AV block, atrial fibrillation, implanted pacemaker, and aortic regurgitation were significantly increased in patients with AS (HRs 2.3, 1.3, 2.1, and 1.9, respectively), uSpA (HRs 2.9, 1.3, 1.9, and 2.0, respectively), and PsA (HRs 1.5, 1.5, 1.6, and 1.8, respectively) than those with the GP. Male uSpA (HR: 4.2) and AS patients (HR:2.5) had the highest HRs for AV block compared to the GP. They also reported that the patients with SpA are at increased risk for aortic regurgitation, cardiac rhythm disturbances, and, most probably, pacemaker implantation as a probable final consequence. Although increased caution is warranted, particularly for atrial fibrillation as the most common arrhythmia, AV block should be investigated, especially in male patients with AS or uSpA [21].
It has been known that AS has been associated with cardiac conduction disturbances for a long time. Nevertheless, contemporary longitudinal studies are scarce on this subject. A longitudinal cohort study with the baseline and 5-year follow-up electrocardiographic examinations showed that at least one type of cardiac conduction abnormality was seen in 18% of the patients, and the first-degree AV block was the most frequent abnormality. Previous analyses indicated that baseline male sex, history of anterior uveitis (OR: 4.4), higher ASDAS-CRP score (OR: 2.3), greater waist circumference (OR: 1.3), and medication with antiplatelets (OR: 7.0) and beta-blockers (OR: 3.4) were significantly associated with the development of cardiac conduction disease during the follow-up. There was a higher correlation between older age and longer symptom duration. Besides, these two variables were associated with newly developed cardiac conduction disturbances during the follow-up period [22]. In a recent meta-analysis, the authors evaluated the systematic prevalence rates of left ventricular (LV) systolic and diastolic dysfunction and valvular heart disease (VHD) in axSpA patients without a history of CVD. They performed transthoracic echocardiographic examinations of the heart valves and cardiac functions. Their findings revealed that the patients with axSpA have LV ejection fraction alterations without clinically relevant significance. Besides, the prevalence rates of VHD were similar in axSpA patients and healthy individuals. Although the LV diastolic transthoracic echocardiographic parameters were altered in axSpA patients, most studies fail to combine the appropriate echocardiographic parameters to recognize diastolic dysfunction. We believe that the clinical relevance of diastolic dysfunction detected during transthoracic echocardiographic examinations remains to be determined in future longitudinal studies [23].
Hypertension
The relationship between CV comorbidities and disease duration was evaluated in 3923 subjects using ASAS-COMOSPA project data. Hypertension (22.4%), ischemic heart disease (2.6%), stroke (1.3%), and DM (5.5%) were the most frequent main CV-related comorbidities. They also showed a significant association between hypertension and disease duration of SpA. The subgroup analysis revealed that the most decisive impact of hypertension on the disease duration was seen in patients with the axial-only SpA phenotype. There was no significant association between hypertension and disease duration for the peripheral-only SpA. Based on the findings of this study, the authors did not find any association between SpA disease duration and other CV-related comorbidities, including ischemic heart disease, stroke, and diabetes mellitus. It is known that long-lasting hypertension is a significant risk factor for CV morbidities. So, blood pressure measurements in routine clinical practice should be performed regularly in patients with SpA. The management of hypertension, particularly in those with longer disease duration, seems to be an essential preventive practice for future CV morbidities [24].
The analysis of 1242 patients recorded in the Biologic and targeted Synthetic anti-rheumatic drugs Registry (BioStaR) revealed that hypertension and DM were the most frequent two comorbidities in 13.4% and 6.7% of the cases, respectively. The incidences of hypertension (55.9% vs. 10.0%) and DM (28.0% vs. 5.0%) were significantly higher in patients over 60 years of age. In this study, the authors regarded the patients whose body mass index was higher than 25 kg/m2 were overweight. Such discrimination showed a similar pattern to those defined for age. Patients with a body mass index over 25 kg/m2 had a significantly higher incidence of hypertension and diabetes mellitus [16].
Ischemic heart disease
Although several studies showed an increased risk for ischemic heart disease and acute MI in patients with AS, these results might be contradictory considering the study designs and the heterogeneities in the patient groups.
The incidence of the acute coronary syndrome (ACS) has been retrospectively evaluated in patients with (n = 6262) and without AS (n = 25,048) in a study. They found that the overall incidence of ACS was higher in patients with AS than in those without AS (4.4 vs. 2.9 per 1000 person-year) (adjusted HR was 1.36). In this study, AS patients with hypertension, DM, and cancer had a higher risk for the development of ACS with an HR of 7.74 compared to patients without these comorbidities [25].
Uveitis has been another risk factor for ACS based on a 15-year retrospective study. The patients with AS and uveitis had a significantly higher risk of ACS than those without uveitis. After adjustment for age, sex, and comorbidities, the authors found that uveitis in patients with AS had an adjusted HR of 1.675 for ACS. The common underlying mechanism for the association between uveitis and ACS may be inflammatory.The authors reported a higher incidence of ACS in patients with AS and uveitis compared those without uveitis (adjusted HR: 1.675). Based on the findings of this study, uveitis has been proposed as a new potential risk factor for ACS in patients with AS [26].
In another study comparing atherosclerosis disease burden and defining CV risk between AS and nr-axSpA patients, the authors evaluated 639 patients with AS and 167 patients with nr-axSpA in patients with no CVE history. They performed a carotid ultrasound to determine the carotid intima-media thickness and to detect the carotid plaques. The European Cardiovascular Disease Risk Assessment Model and the Systematic COronary Risk Evaluation (SCORE) were used, and the authors reclassified the patients from the low and moderate CV risk categories to the very high-risk category based on the presence of carotid plaques. The percentages of the risk categories were comparable in AS and nr-axSpA patients (10.7% vs. 10.1% and 40.5% vs. 45.5%, respectively). They concluded that there was a similar atherogenic burden in AS and nr-axSpA. A substantial proportion of the patients with moderate CV risk were reclassified using the SCORE system and regarded as the very high-risk category because of having carotid plaques. So, carotid ultrasound evaluation should be performed in all patients with axSpA with moderate risk which may avoid unwanted consequences of the carotid plaques [27].
In a cross-sectional observational study investigating atherosclerosis-related factors in AS and nr-axSpA, 639 AS and 167 nr-axSpA patients were compared. When classical CV risk factors were excluded from the analysis, the factors associated with atherosclerotic disease were inflammatory response and disease severity. They also found no difference between AS and nr-axSpA [28].
In an ultrasound-based study on SpA patients with no known CV risk factors or a history of CVD, it was found that the carotid intima-media thickness and endothelial dysfunction were significantly increased in patients with SpA than the healthy controls. These findings confirm that accelerated subclinical atherosclerosis can be seen in young patients with SpA (< 50 years old). The traditional CV risk factors, such as creatinine, total cholesterol, and age, were also the predictive clinical markers for subclinical atherosclerosis. These data highlight the need for tight CV risk control and early diagnosis of atherosclerosis in patients with SpA [29].
Effect of HLA-B27
Previous studies reported that HLA-B27 might be a genetic risk factor for various cardiac conditions [2]. In HLA-B27 transgenic rats, spontaneous inflammation developed in the hearts of the animals, besides the joints, gastrointestinal system, eyes, and skin [30]. In a study including 2370 axSpA patients with an HLA-B27 positivity rate of 78.4%, the authors found that male sex, longer disease duration, definite AS, higher CRP, tobacco consumption, and excessive alcohol intake were significantly associated with the HLA-B27 positivity in axSpA patients [31].
However, there was no significant difference in the HLA-B27 status of patients with and without a history of cardiac conduction disorders, according to the 5-year follow-up cohort study findings. The authors tried to explain this controversy as the high prevalence of HLA-B27 [22]. In a recent study using cardiac ultrasound to evaluate the heart chambers of 124 patients, the prevalence rate of VHD was 24.2%. The subgroup analysis based on the status of HLA-B27 revealed that patients with positive HLA-B27 presented more frequently with symptomatic VHD (27.4%). The difference between the HLA-B27 positive and negative patients was significant [32].
Impact of treatment on cardiovascular risk
Nonsteroidal anti-inflammatory drugs (NSAIDs)
The NSAIDs have been investigated regarding their safety profile of cardiovascular, gastrointestinal, and other systems for a long-time. However, there are still conflicting data about the use of NSAIDs in patients with axSpA. Several papers reported that cardiovascular side effects had been observed among patients using such medications. At the same time, others proposed that even a decrease in CV complications and mortality is possible in these patients. A reduction in the inflammation and better mobility of the patients due to NSAID use has been speculated as the underlying pathophysiological mechanisms associated with the cardiac protective effects. Furthermore, axSpA patients are younger than those with RA and osteoarthritis. So, it is possible to see a relatively minor cardiovascular and gastrointestinal risk in these patients. More studies are needed to conclude the potential risk of NSAID in treating axSpA [33].
Among the different classes of NSAIDs, the highly selective cyclooxygenase-2 (COX-2) inhibitors and diclofenac were significantly associated with a higher risk of major vascular events. This risk was less reported with naproxen usage. According to most of the recommendations, NSAIDs are the drugs that are used as the first-line therapy in axSpA. However, the increased NSAID-induced CV risk in patients with axSpA has been controversial. In two different mortality studies performed among AS patients, the authors reported that the lack of or insufficient NSAID intake was significantly associated with increased CV mortality. It has been suggested that NSAIDs can partially control inflammation, and thus atherosclerosis may be accelerated in patients who do not use NSAIDs. However, these results may reflect a selection bias that NSAIDs may be recommended only to patients without CV risk factors [5].
In a study investigating the risk factors for major adverse cardiovascular events (MACE) in AS patients receiving medical treatment, the highly selective COX-2 inhibitors, especially at higher doses and corticosteroid use, were found to be the significant risk factors. The authors reported no significant impact of the traditional NSAIDs, the highly selective COX-2 inhibitors, biological modifying drugs, methotrexate, and sulfasalazine on developing MACE risk. The major limitation of this study was the lack of information about the patient’s lifestyle patterns and disease activities [34].
Biologic disease modifying anti-rheumatic drugs (bDMARDs)
Tumor necrosis factor (TNF) inhibitors
It is expected to see a reduction in cardiovascular risk of the axSpA patients with the use of TNF inhibitors considering the role of TNF-α in inflammation and atherosclerosis. It is known that there is a significant relationship between inflammation and increased CV risk. Controlling the inflammatory status via appropriate medications decreases the CV risk, and an improvement in myocardial changes can be observed. Previous studies have shown that anti-TNF drugs improve sub-clinical atherosclerosis and the disturbed lipid profile. Although there may be some controversial results, it is generally accepted that TNF inhibitors positively impact the development of atherosclerosis [5, 35].
A retrospective analysis with a total of 5046 patients 2616 SpA patients and 2430 patients with non-specific back pain showed that the overall incidence rates of MACE and stroke were significantly higher in SpA patients. The HRs for MACE and cerebrovascular events were 1.70 and 1.50. However, anti-TNF using SpA patients had an HR of 0.37 for MACE and an HR of 0.21 for cerebrovascular events compared to not using anti-TNFs. Based on these findings, the authors concluded that SpA is an independent risk factor for CVD and anti-TNF drugs reduce the increased CV risk in patients with SpA [36].
Obesity is one of the known traditional and modifiable risk factors for CVD. The adipose tissue is capable of producing different proinflammatory cytokines, adipokines, by influencing the immunological processes in obese people. Besides, it has been observed that the success rate of anti-TNF therapy in overweight or obese patients has been reduced. Other studies reported that women with AS were less responsive to the TNF-α blockers and had a shorter drug survival. This difference may also be due to the differences in body composition between genders. In a study investigating the relationship between the body composition and anti-TNF response in patients with AS, baseline higher body fat content was independently associated with a worse response to anti-TNF-α treatment. So, they concluded that obesity is a risk factor that should be considered in this respect [37].
In other systematic reviews and meta-analyses considering the 2021 EULAR recommendations for lifestyle improvements in people with rheumatic and musculoskeletal diseases, physical exercises have been recommended to maintain ideal and healthy body weight [38].
Smoking is another modifiable risk factor. It has been shown that the rate of neutralizing anti-drug antibodies was higher in smokers than nonsmokers (46% vs. 15%). Poor disease activity was associated with smoking in patients using anti-TNFs [39]. The negative effect of smoking on many rheumatic diseases has been emphasized in other systematic reviews and meta-analyses, considering the 2021 EULAR recommendations for lifestyle improvements in people with rheumatic and musculoskeletal diseases. Based on these recommendations, health professionals are expected to encourage patients to stop smoking and provide information about the destructive effects of smoking [40].
Male gender is another non-modifiable risk factor for CV risk. However, of 969 SpA patients using bDMARDS, we found that male sex is a predictive variable for the remission of the disease [41]. In a recent observational study, the authors reported that the remission rate of the anti-TNF treatment was significantly lower in female patients with inflammatory arthritis than in male patients [42]. Because of the significant differences in the burden of disease between male and female patients, gender differences should be considered during patient follow-up [42].
Interleukin 17 (IL-17) inhibitors
Experience with IL-17 inhibitors, proatherogenic cytokines, is relatively limited compared to TNF inhibitors. Studies monitoring the safety profiles of IL-17 inhibitors reported no significant changes in the number of CVEs or MACEs in patients using secukinumab and ixekizumab [5]. A recent study analyzed 9197 patients in 19 clinical phase 3 or 4 trials. The number of patients and trials and was 4742 psoriasis patients (eight trials), 2475 PsA patients (five trials), and 1980 axSpA patients (six trials). It has been shown that the treatment with secukinumab was associated with a reduction in high-sensitivity CRP and the neutrophil–lymphocyte ratio. Although the traditional CV risk factors remained stable in patients, high-sensitivity CRP and the neutrophil–lymphocyte ratio are critical biomarkers for systemic inflammation and CV risk [43].
Targeted synthetic DMARDs
Janus kinase (JAK) inhibitors, including tofacitinib, upadacitinib, and filgotinib, are currently investigated regarding their use in axSpA. Recently, studies have focused on using JAK inhibitors in SpA. However, there is no data about an increase in CV risk [5, 33]. An increased CV risk has not been reported in the SELECT-AXIS1 study [44] with active AS patients, the SELECT-AXIS 2 study [45] with nr-axSpA patients, and Phase 3 SELECT-PsA1 study [46] with PsA patients. The risk for MACE and VTE remained stable in these studies. However, in the final analysis of the ORAL Surveillance study in which tofacitinib use in RA was investigated, the risk of MACE and cancer development was significantly higher in patients using tofacitinib than in those using TNF inhibitors [47]. Therefore, a decision regarding the selection and follow-up protocols of the drugs should be made considering all the risks and benefits.
Although it is understood that the risk of CV morbidity and mortality is increased in SpA, the management of the disease has not been entirely determined. So, it is essential to identify the patients with higher CV risk as early as possible. Such an approach might include using complementary non-invasive imaging techniques for the CV system and the application of a CV risk stratification score. It is justified to show higher vigilance for identifying risk factors, followed by the appropriate interactions in all rheumatic patients. A screening approach for the traditional CV risk factors should be applied to all SpA patients. Besides, the SCORE algorithm as a CV risk stratification score should be calculated in these patients. The modified SCORE index is the most commonly used algorithm in European countries. The modification has been performed based on the recommendations proposed by the EULAR task force in 2017. Such approaches, such as the routine clinical assessment of the CV risk and the international SCORE tool to calculate the CV risk in these patients, may help patients increase their motivation level. These patients can improve their CV risk via lifestyle changes, including dietary efforts and smoking cessation [48, 49].
In conclusion, the risk of CV morbidity and mortality is increased in SpA. All physicians dealing with SpA treatment should know that CVE and CV risk factors have increased and that early diagnosis and appropriate preventive approaches should be made carefully. Routine evaluations should be made in terms of CV risk factors. Finally, the risks should be identified, managed, and treated early in accordance with national and international guidelines with a strict multidisciplinary approach. Reducing the activity of the underlying disease with an appropriate and early treatment will also reduce the risk of CVE. Good communication and cooperation should be established with the fields of expertise related to the multidisciplinary approach, healthcare professionals, and the patient. Lifestyle habits improving the CV health status should be given to the patients.
References
Rudwaleit M (2019) Classification and epidemiology of spondyloarthritis. In: Hochberg MC, Gravallase EM, Silman AJ, Smolen JS,Weinblatt ME, Weisman MH (eds.) Rheumatology, Elsevier, 7th edn pp 993–997
Wysham KD, Gensler LS (2019) Clinical features of axial spondyloarthritis. In: Hochberg MC, Gravallase EM, Silman AJ, Smolen JS,Weinblatt ME, Weisman MH (eds.) Rheumatology, Elsevier, 7th edn. pp 1002–1008
Cardelli C, Monti S, Terenzi R, Carli L (2021) One year in review 2021: axial spondyloarthritis. Clin Exp Rheumatol 39:1272–1281. https://doi.org/10.55563/clinexprheumatol/jlyd1l
Zhao SS, Ermann J, Xu C (2019) Comparison of comorbidities and treatment between ankylosing spondylitis and non-radiographic axial spondyloarthritis in the United States. Rheumatology (Oxford) 58:2025–2030. https://doi.org/10.1093/rheumatology/kez171
Toussirot E (2021) The risk of cardiovascular diseases in axial spondyloarthritis. Current insights Front Med (Lausanne) 8(8):782150. https://doi.org/10.3389/fmed.2021.782150
Moltó A, Etcheto A, van der Heijde D et al (2016) Prevalence of comorbidities and evaluation of their screening in spondyloarthritis: results of the international cross-sectional ASAS-COMOSPA study. Ann Rheum Dis 75:1016–1023. https://doi.org/10.1136/annrheumdis-2015-208174
Kerola AM, Kazemi A, Rollefstad S (2022) All-cause and cause-specific mortality in rheumatoid arthritis, psoriatic arthritis and axial spondyloarthritis: a nationwide registry study. Rheumatology (Oxford) 4:keac210. https://doi.org/10.1093/rheumatology/keac210
Agca R, Heslinga SC, Rollefstad S et al (2017) EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann Rheum Dis 76:17–28. https://doi.org/10.1136/annrheumdis-2016-209775
Drosos GC, Vedder D, Houben E et al (2022) EULAR recommendations for cardiovascular risk management in rheumatic and musculoskeletal diseases, including systemic lupus erythematosus and antiphospholipid syndrome. Ann Rheum Dis 81:768–779. https://doi.org/10.1136/annrheumdis-2021-221733
van der Heijde D, Ramiro S, Landewé R et al (2016) update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Ann Rheum Dis 76:978–991. https://doi.org/10.1136/annrheumdis-2016-210770
Kim JH, Choi IA (2021) Cardiovascular morbidity and mortality in patients with spondyloarthritis: a meta-analysis. Int J Rheum Dis 24:477–486. https://doi.org/10.1111/1756-185X.13970
Martín-Martínez MA, Castañeda S, González-Juanatey C et al (2019) Incidence of first cardiovascular event in Spanish patients with inflammatory rheumatic diseases: prospective data from the CARMA project. Clin Exp Rheumatol 37:731–739
Martín-Martínez MA, Castañeda S, Sánchez-Alonso F et al (2021) Cardiovascular mortality and cardiovascular event rates in patients with inflammatory rheumatic diseases in the CARdiovascular in rheuMAtology (CARMA) prospective study-results at 5 years of follow-up. Rheumatology (Oxford) 60:2906–2915. https://doi.org/10.1093/rheumatology/keaa737
Bengtsson K, Forsblad-d’Elia H, Lie E et al (2017) Are ankylosing spondylitis, psoriatic arthritis and undifferentiated spondyloarthritis associated with an increased risk of cardiovascular events? A prospective nationwide population-based cohort study. Arthritis Res Ther 18(19):102. https://doi.org/10.1186/s13075-017-1315-z
López-Medina C, Jiménez-Gómez Y, Moltó A et al (2018) Cardiovascular risk factors in patients with spondyloarthritis from Northern European and Mediterranean countries: an ancillary study of the ASAS-COMOSPA project. Joint Bone Spine 85:447–453. https://doi.org/10.1016/j.jbspin.2017.07.006
Çay HF, Alkan Melikoğlu M, Yurdakul FG et al (2023) Real-life data on the comorbidities in spondyloarthritis from our multicenter nationwide registry: BioStaR Arch Rheumatol. https://doi.org/10.46497/ArchRheumatol.2023.9793
Ferraz-Amaro I, Rueda-Gotor J, Genre F et al (2021) Potential relation of cardiovascular risk factors to disease activity in patients with axial spondyloarthritis. Ther Adv Musculoskelet Dis 28(13):1759720X211033755. https://doi.org/10.1177/1759720X211033755
Navarini L, Currado D, Marino A et al (2022) Persistence of C-reactive protein increased levels and high disease activity are predictors of cardiovascular disease in patients with axial spondyloarthritis. Sci Rep 7(12):7498. https://doi.org/10.1038/s41598-022-11640-8
Bhattad PB, Kulkarni M, Patel PD, Roumia M (2022) Cardiovascular morbidity in ankylosing spondylitis: a focus on inflammatory cardiac disease. Cureus 14:e25633. https://doi.org/10.7759/cureus.25633
Forsblad-d’Elia H, Wallberg H, Klingberg E et al (2013) Cardiac conduction system abnormalities in ankylosing spondylitis: a cross-sectional study. BMC Musculoskelet Disord 14:237
Bengtsson K, Forsblad-d’Elia H, Lie E et al (2018) Risk of cardiac rhythm disturbances and aortic regurgitation in different spondyloarthritis subtypes in comparison with general population: a register-based study from Sweden. Ann Rheum Dis 77:541–548. https://doi.org/10.1136/annrheumdis-2017-212189
Bengtsson K, Klingberg E, Deminger A et al (2019) Cardiac conduction disturbances in patients with ankylosing spondylitis: results from a 5-year follow-up cohort study. RMD Open 13(5):e001053. https://doi.org/10.1136/rmdopen-2019-001053
Romand X, Adeline F, Dalecky M et al (2022) Systematic assessment of heart valves and cardiac function by echocardiography in axial spondyloarthritis: a systematic review and meta-analysis. Joint Bone Spine 89:105375. https://doi.org/10.1016/j.jbspin.2022.105375
Derakhshan MH, Goodson NJ, Packham JC et al (2019) Increased risk of hypertension associated with spondyloarthritis disease duration: results from the ASAS-COMOSPA study. J Rheumatol 46:701–709. https://doi.org/10.3899/jrheum.180538
Chou CH, Lin MC, Peng CL et al (2014) A nationwide population-based retrospective cohort study: increased risk of acute coronary syndrome in patients with ankylosing spondylitis. Scand J Rheumatol 43:132–136. https://doi.org/10.3109/03009742.2013.822097
Feng KM, Chien WC, Chen YH et al (2022) Increased risk of acute coronary syndrome in ankylosing spondylitis patients with uveitis: a population-based cohort study. Front Immunol 10(13):890543. https://doi.org/10.3389/fimmu.2022.890543
González Mazón I, Rueda-Gotor J, Ferraz-Amaro I et al (2021) Subclinical atherosclerotic disease in ankylosing spondylitis and non-radiographic axial spondyloarthritis. A multicenter study on 806 patients. Semin Arthritis Rheum 51:395–403. https://doi.org/10.1016/j.semarthrit.2021.02.003
Rueda-Gotor J, Ferraz-Amaro I, Genre F et al (2022) Factors associated with atherosclerosis in radiographic and non-radiographic axial spondyloarthritis. A multicenter study on 838 patients. Semin Arthritis Rheum 55:152037. https://doi.org/10.1016/j.semarthrit.2022.152037.
Ben Tekaya A, Boukriba S, Fendri A et al (2022) Endothelial dysfunction and increased carotid intima-media thickness in patients with spondyloarthritis without traditional cardiovascular risk factors. RMD Open 8:e002270. https://doi.org/10.1136/rmdopen-2022-002270
Hammer RE, Maika SD, Richardson JA, Tang JP, Taurog JD (1990) Spontaneous inflammatory disease in transgenic rats expressing HLA-B27 and human beta 2m: an animal model of HLA-B27-associated human disorders. Cell 63:1099–1112. https://doi.org/10.1016/0092-8674(90)90512-d
Poddubnyy D, Sieper J, Arévalo M et al (2020) Role of HLA-B27 in the comorbidities observed in Axial Spondyloarthritis: data from COMOSPA. Joint Bone Spine 87:445–448. https://doi.org/10.1016/j.jbspin.2020.03.012
Lai Y, ZhangY MoS et al (2022) Prevalence of comorbidities and risk factors in spondyloarthritis: results of a cross-sectional study. Ann Rheum Dis 81:e43. https://doi.org/10.1136/annrheumdis-2020-217313
Poddubnyy D, Sieper J (2020) Treatment of axial spondyloarthritis: what does the future hold? Curr Rheumatol Rep 22:47. https://doi.org/10.1007/s11926-020-00924-5
Kao CM, Wang JS, Ho WL et al (2022) Factors associated with the risk of major adverse cardiovascular events in patients with ankylosing spondylitis: a nationwide, population-based case-control study. Int J Environ Res Public Health 19:4098. https://doi.org/10.3390/ijerph19074098
Atzeni F, Nucera V, Galloway J, Zoltán S, Nurmohamed M (2020) Cardiovascular risk in ankylosing spondylitis and the effect of anti-TNF drugs: a narrative review. Expert Opin Biol Ther 20:517–524. https://doi.org/10.1080/14712598.2020.1704727
Chan SCW, Teo CK, Li PH, Lau KK, Lau CS, Chung HY (2021) Cardiovascular risk in patients with spondyloarthritis and association with anti-TNF drugs. Ther Adv Musculoskelet Dis 13:1759720X211032444. https://doi.org/10.1177/1759720X211032444
Ibáñez Vodnizza SE, Nurmohamed MT, Visman IM et al (2017) Fat mass lowers the response to tumor necrosis factor-α blockers in patients with ankylosing spondylitis. J Rheumatol 44:1355–1361. https://doi.org/10.3899/jrheum.170094
Gwinnutt JM, Wieczorek M, Cavalli G et al (2021) Effects of physical exercise and body weight on disease-specific outcomes of people with rheumatic and musculoskeletal diseases (RMDs): systematic reviews and meta-analyses informing the 2021 EULAR recommendations for lifestyle improvements in people with RMDs. RMD Open 8:e002168. https://doi.org/10.1136/rmdopen-2021-002168
Michelsen B, Berget KT, Kavanaugh A, Haugeberg G (2022) Association between TNFi anti-drug antibodies, smoking, and disease activity in patients with inflammatory arthritis: results from a Norwegian cross-sectional observational study. Rheumatol Ther 9:1171–1179. https://doi.org/10.1007/s40744-022-00464-7
Wieczorek M, Gwinnutt JM, Ransay-Colle M et al (2022) Smoking, alcohol consumption and disease-specific outcomes in rheumatic and musculoskeletal diseases (RMDs): systematic reviews informing the 2021 EULAR recommendations for lifestyle improvements in people with RMDs. RMD Open 8:e002170. https://doi.org/10.1136/rmdopen-2021-002170
Bodur H, Yurdakul FG, Ataman S et al (2022) Where we are in treat to target era? Predictive factors for remission and drug switching in patients with axial spondyloarthritis: a real-life evidence from BioStaR nationwide registry. Clin Rheumatol 41:2053–2063. https://doi.org/10.1007/s10067-022-06145-8
Michelsen B, Berget KT, Loge JH, Kavanaugh A, Haugeberg G (2022) Sex difference in disease burden of inflammatory arthritis patients treated with tumor necrosis factor inhibitors as part of standard care. PLoS ONE 17:e0266816. https://doi.org/10.1371/journal.pone.0266816
Merola JF, McInnes IB, Deodhar AA et al (2022) Effect of secukinumab on traditional cardiovascular risk factors and inflammatory biomarkers: post hoc analyses of pooled data across three indications. Rheumatol Ther 9:935–955. https://doi.org/10.1007/s40744-022-00434-z
van der Heijde D, Song IH, Pangan AL et al (2019) Efficacy and safety of upadacitinib in patients with active ankylosing spondylitis (SELECT-AXIS 1): a multicentre, randomised, double-blind, placebo-controlled, phase 2/3 trial. Lancet 394:2108–2117. https://doi.org/10.1016/S0140-6736(19)32534-6
Deodhar A, Van den Bosch F, Poddubnyy D et al (2022) Upadacitinib for the treatment of active non-radiographic axial spondyloarthritis (SELECT-AXIS 2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 400:369–379. https://doi.org/10.1016/S0140-6736(22)01212-0
McInnes IB, Kato K, Magrey M et al (2021) Upadacitinib in patients with psoriatic arthritis and an inadequate response to non-biological therapy: 56-week data from the phase 3 SELECT-PsA 1 study. RMD Open 7:e001838. https://doi.org/10.1136/rmdopen-2021-001838. (Erratum in: RMD Open. 2021 Nov;7(3).)
Ytterberg SR, Bhatt DL, Mikuls TR et al (2022) Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med 386(4):316–326. https://doi.org/10.1056/NEJMoa2109927
Castañeda S, Vicente-Rabaneda EF, García-Castañeda N et al (2020) Unmet needs in the management of cardiovascular risk in inflammatory joint diseases. Expert Rev Clin Immunol 16:23–36. https://doi.org/10.1080/1744666X.2019.1699058
Yagensky V, Schirmer M (2022) Cardiovascular risks and risk stratification in inflammatory joint diseases: a cross-sectional study. Front Med (Lausanne) 9:786776. https://doi.org/10.3389/fmed.2022.786776
Author information
Authors and Affiliations
Contributions
Study conception and design: HB. Acquisition of data: HB. Analysis and interpretation of data: HB. Drafting of manuscript: HB. Literature search: HB.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that there is no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Part of the Topical Collection entitled ‘Cardiovascular Issues in Rheumatic Diseases’
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bodur, H. Cardiovascular comorbidities in spondyloarthritis. Clin Rheumatol 42, 2611–2620 (2023). https://doi.org/10.1007/s10067-022-06473-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06473-9