Abstract.
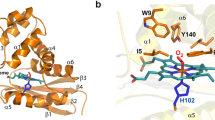
We have recently discovered heme-containing signal transducers from the archaeon Halobacterium salinarum (HemAT-Hs) and the gram-positive bacterium Bacillus subtilis (HemAT-Bs). These proteins bind diatomic oxygen and trigger aerotactic responses. We identified that HemAT oxygen-sensing domains contain a globin-coupled sensor (GCS) motif, which exists as a two-domain transducer, having no similarity to the PAS domain (Period circadian protein, Ah receptor nuclear translocator protein, Single-minded protein) superfamily transducers. Using the GCS motif, we predicted that a 439-amino-acid protein annotated as a methyl-accepting chemotaxis protein (MCP) in the facultatively alkaliphilic bacterium Bacillus halodurans is a globin-coupled oxygen sensor. We cloned, expressed, and purified GCS Bh and performed its spectral analysis. GCS Bh binds heme and shows myoglobin-like spectra. This suggests that GCS Bh acts as an oxygen sensor and transmits a conformational signal through a linked signaling domain to trigger an aerotactic response in B. halodurans.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Hou, S., Belisle, C., Lam, S. et al. A globin-coupled oxygen sensor from the facultatively alkaliphilic Bacillus halodurans C-125. Extremophiles 5, 351–354 (2001). https://doi.org/10.1007/s007920100220
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s007920100220