Abstract
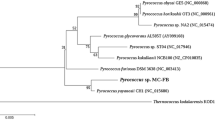
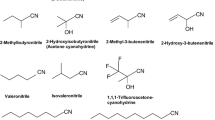
A novel, nitrile-degrading, thermophilic microorganism belonging to the genus Bacillus and most closely related to strain DSM 2349 has been isolated. The strain grew optimally at 65°C with the constitutive expression of a thermostable intracellular nitrile hydratase. No aromatic-specific "benzonitrilase" activity was detected under any conditions. The enzyme, an α2β2 heterotetramer with a native molecular weight of 110 kDa, was purified to homogeneity. N-terminal sequence data showed no homology to known bacterial α subunit sequences but had a high level of identity with other bacterial N-terminal β subunit sequences. The purified enzyme had a broad pH-activity range (50% activity limits were pH 5.1 and 8.7) and was stable in aqueous solution up to 60°C in the absence of either substrates or substrate analogues. Substrate specificity was restricted to aliphatic nitriles, but an unusual preference for branched and cyclic aliphatic nitriles was noted. Turnover rates under optimum reaction conditions were 746 and 4580 sec−1 for acetonitrile and valeronitrile, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: December 1, 1997 / Accepted: February 24, 1998
Rights and permissions
About this article
Cite this article
Pereira, R., Graham, D., Rainey, F. et al. A novel thermostable nitrile hydratase. Extremophiles 2, 347–357 (1998). https://doi.org/10.1007/s007920050078
Issue Date:
DOI: https://doi.org/10.1007/s007920050078