Abstract
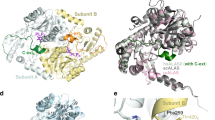
Ceruloplasmin is a multi-copper oxidase, which contains most of the copper present in the plasma. It is an acute-phase reactant that exhibits a two- to three-fold increase over the normal concentration of 300 μg/ml in adult plasma. However, the precise physiological role(s) of ceruloplasmin has been the subject of intensive debate and it is likely that the enzyme has a multi-functional role, including iron oxidase activity and the oxidation of biogenic amines. The three-dimensional X-ray structure of the human enzyme was elucidated in 1996 and showed that the molecule was composed of six cupredoxin-type domains arranged in a triangular array. There are six integral copper atoms per molecule (mononuclear sites in domains 2, 4 and 6 and a trinuclear site between domains 1 and 6) and two labile sites with roughly 50% occupancy. Further structural studies on the binding of metal cations by the enzyme indicated a putative mechanism for ferroxidase activity. In this paper we report medium-resolution X-ray studies (3.0–3.5 Å) which locate the binding sites for an inhibitor (azide) and various substrates [aromatic diamines, biogenic amines and (+)-lysergic acid diethylamide, LSD]. The binding site of the azide moiety is topologically equivalent to one of the sites reported for ascorbate oxidase. However, there are two distinct binding sites for amine substrates: aromatic diamines bind on the bottom of domain 4 remote from the mononuclear copper site, whereas the biogenic amine series typified by serotonin, epinephrine and dopa bind in close vicinity to that utilised by cations in domain 6 and close to the mononuclear copper. These binding sites are discussed in terms of possible oxidative mechanisms. The binding site for LSD is also reported.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 19 March 1999 / Accepted: 21 June 1999
Rights and permissions
About this article
Cite this article
Zaitsev, V., Zaitseva, I., Papiz, M. et al. An X-ray crystallographic study of the binding sites of the azide inhibitor and organic substrates to ceruloplasmin, a multi-copper oxidase in the plasma. JBIC 4, 579–587 (1999). https://doi.org/10.1007/s007750050380
Issue Date:
DOI: https://doi.org/10.1007/s007750050380