Abstract.
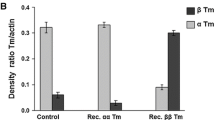
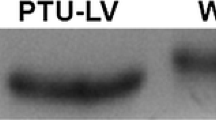
The head portion of the myosin heavy chain (MHC) is essential in force generation. As previously shown, Ca2+-activated fibres of mammalian skeletal muscle display a strong correlation between their MHC isoform complement and the kinetics of stretch activation, suggesting isoform-specific differences in kinetic properties of myosin heads. Using the same methodology on muscle strips of atria and ventricles of hyper- and hypothyroid rats, this study showed that the kinetics of cardiac αMHC are 3 times faster than those of cardiac βMHC under isometric conditions and maximal Ca2+ activation. Comparison of rat heart and skeletal muscle fibres revealed that 100% αMHC heart muscle strips exhibited faster stretch activation kinetics (time parameter t 3: 108±18 ms, mean±SD) than rat type-IIA fibres (t 3: 157±19 ms), but slower than type-IID fibres (t 3: 55±10 ms). The kinetics of 100% βMHC heart muscle strips (t 3: 351±44 ms) were faster than that of type-I fibres in rat skeletal muscle (t 3: 901±348 ms). This difference between the two muscle types calls in question the generally accepted identity of βMHC and MHCIβ.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Galler, S., Puchert, E., Gohlsch, B. et al. Kinetic properties of cardiac myosin heavy chain isoforms in rat. Pflugers Arch - Eur J Physiol 445, 218–223 (2002). https://doi.org/10.1007/s00424-002-0934-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00424-002-0934-6