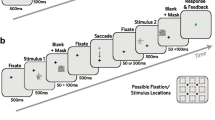
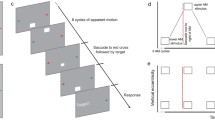
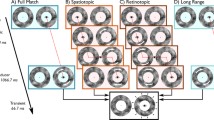
Abstract
Whenever we shift our gaze, any location information encoded in the retinocentric reference frame that is predominant in the visual system is obliterated. How is spatial memory retained across gaze changes? Two different explanations have been proposed: Retinocentric information may be transformed into a gaze-invariant representation through a mechanism consistent with gain fields observed in parietal cortex, or retinocentric information may be updated in anticipation of the shift expected with every gaze change, a proposal consistent with neural observations in LIP. The explanations were considered incompatible with each other, because retinocentric update is observed before the gaze shift has terminated. Here, we show that a neural dynamic mechanism for coordinate transformation can also account for retinocentric updating. Our model postulates an extended mechanism of reference frame transformation that is based on bidirectional mapping between a retinocentric and a body-centered representation and that enables transforming multiple object locations in parallel. The dynamic coupling between the two reference frames generates a shift of the retinocentric representation for every gaze change. We account for the predictive nature of the observed remapping activity by using the same kind of neural mechanism to generate an internal representation of gaze direction that is predictively updated based on corollary discharge signals. We provide evidence for the model by accounting for a series of behavioral and neural experimental observations.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amari S (1977) Dynamics of pattern formation in lateral-inhibition type neural fields. Biol Cybern 27(2): 77–87
Andersen RA, Mountcastle VB (1983) The influence of the angle of gaze upon the excitability of the light-sensitive neurons of the posterior parietal cortex. J Neurosci 3(3): 532–548
Andersen RA, Essick GK, Siegel RM (1985) Encoding of spatial location by posterior parietal neurons. Science 230: 456–458
Andersen RA, Snyder LH, Bradley DC, Xing J (1997) Multimodal representation of space in the posterior parietal cortex and its use in planning movements. Annu Rev Neurosci 20: 303–330
Aslin RN, Shea SL (1987) The amplitude and angle of saccades to double-step target displacements. Vision Res 27(11): 1925–1942
Avillac M, Denève S, Olivier E, Pouget A, Duhamel JR (2005) Reference frames for representing visual and tactile locations in parietal cortex. Nat Neurosci 8(7): 941–949
Bridgeman B, Stark L (1991) Ocular proprioception and efference copy in registering visual direction. Vision Res 31(11): 1903–1913
Brotchie PR, Andersen RA, Snyder LH, Goodman SJ (1995) Head position signals used by parietal neurons to encode locations of visual stimuli. Nature 375(6528): 232–235
Buneo CA, Andersen RA (2006) The posterior parietal cortex: Sensorimotor interface for the planning and online control of visually guided movements. Neuropsychologia 44(13): 2594–2606
Cassanello CR, Ferrera VP (2007) Computing vector differences using a gain field-like mechanism in monkey frontal eye field. J Physiol (Lond) 582: 647–664
Cavanagh P, Hunt AR, Afraz A, Rolfs M (2010) Visual stability based on remapping of attention pointers. Trends Cogn Sci 14(4): 147–153
Cisek P (2006) Integrated neural processes for defining potential actions and deciding between them: A computational model. J Neurosci 26(38): 9761–9770
Colby CL, Goldberg ME (1999) Space and attention in parietal cortex. Annu Rev Neurosci 22: 319–349
Denève S, Latham PE, Pouget A (2001) Efficient computation and cue integration with noisy population codes. Nat Neurosci 4: 826–831
Deubel H (2004) Localization of targets across saccades: Role of landmark objects. Vis Cogn 11(2–3): 173–202
Deubel H, Bridgeman B, Schneider WX (1998) Immediate post-saccadic information mediates space constancy. Vision Res 38: 3147–3159
Duhamel JR, Colby CL, Goldberg ME (1992) The updating of the representation of visual space in parietal cortex by intended eye movements. Science 255(5040): 90–92
Eggert J, van Hemmen JL (2001) Modeling neuronal assemblies: theory and implementation. Neural Comput 13(9): 1923–1974
Erlhagen W, Schöner G (2002) Dynamic field theory of movement preparation. Psychol Rev 109(3): 545–572
Fazl A, Grossberg S, Mingolla E (2009) View-invariant object category learning, recognition, and search: How spatial and object attention are coordinated using surface based attentional shrouds. Cogn Psychol 58(1): 1–48
Freedman EG, Stanford TR, Sparks DL (1996) Combined eye-head gaze shifts produced by electrical stimulation of the superior colliculus in rhesus monkeys. J Neurophysiol 76(2): 927–952
Freedman EG, Sparks DL (1997) Eye-head coordination during head-unrestrained gaze shifts in rhesus monkeys. J Neurophysiol 77(5): 2328–2348
Gardner JL, Merriam EP, Movshon JA, Heeger DJ (2008) Maps of visual space in human occipital cortex are retinotopic, not spatiotopic. J Neurosci 28(15): 3988–3999
Golomb JD, Marino AC, Chun MM, Mazer JA (2011) Attention doesn’t slide: spatiotopic updating after eye movements instantiates a new, discrete attentional locus. Atten Percept Psychophys 73(1): 7–14
Goossens HHLM, Van Opstal AJ (2006) Dynamic ensemble coding of saccades in the monkey superior colliculus. J Neurophysiol 95: 2326–2341
Guthrie BL, Porter JD, Sparks DL (1983) Corollary discharge provides accurate eye position information to the oculomotor system. Science 221(4616): 1193–1195
Hallett PE, Lightstone AD (1976) Saccadic eye movements to flashed targets. Vision Res 16(1): 107–114
Heiser LM, Colby CL (2006) Spatial updating in area lip is independent of saccade direction. J Neurophysiol 95: 2751–2767
Johnson JS, Spencer JP, Schöner G (2008) Moving to higher ground: the dynamic field theory and the dynamics of visual cognition. New Ideas Psychol 26(2): 227–251
Karn KS, Møller P, Hayhoe MM (1997) Reference frame in saccadic targeting. Exp Brain Res 115(2): 267–282
Keith GP, Blohm G, Crawford JD (2010) Influence of saccade efference copy on the spatiotemporal properties of remapping: A neural network study. J Neurophysiol 103(1): 117–139
Kopecz K, Schöner G (1995) Saccadic motor planning by integrating visual information and pre-information on neural dynamic fields. Biol Cybern 73(1): 49–60
Kusunoki M, Goldberg ME (2003) The time course of perisaccadic receptive field shifts in the lateral intraparietal area of the monkey. J Neurophysiol 89: 1519–1527
Mays LE, Sparks DL (1980) Saccades are spatially, not retinocentrically, coded. Science 208: 1163–1165
Nakamura K, Chung HH, Graziano MSA, Gross CG (1999) Dynamic representation of eye position in the parieto-occipital sulcus. J Neurophysiol 81(5): 2374–2385
Ottes FP, Van Gisbergen JAM, Eggermont JJ (1984) Metrics of saccade responses to visual double stimuli: two different modes. Vision Res 24(10): 1169–1179
Pouget A, Sejnowski TJ (1997) Spatial transformations in the parietal cortex using basis functions. J Cogn Neurosci 9(2): 222–237
Pouget A, Denève S, Duhamel JR (2002) A computational perspective on the neural basis of multisensory spatial representations. Nature Rev Neurosci 3: 741–747
Quaia C, Optican LM, Goldberg ME (1998) The maintenance of spatial accuracy by the perisaccadic remapping of visual receptive fields. Neural Netw 11(7–8): 1229–1240
Quaia C, Lefèvre P, Optican LM (1999) Model of the control of saccades by superior colliculus and cerebellum. J Neurophysiol 82(2): 999–1018
Salinas E, Abbott LF (1996) A model of multiplicative neural responses in parietal cortex. Proc Natl Acad Sci USA 93(21): 11,956– 11,961
Simmering VR, Schutte AR, Spencer JP (2008) Generalizing the dynamic field theory of spatial cognition across real and developmental time scales. Brain Res 1202: 68–86
Snyder LH, Grieve KL, Brotchie P, Andersen RA (1998) Separate body- and world-referenced representations of visual space in parietal cortex. Nature 394(6696): 887–891
Sommer MA, Wurtz RH (2004a) What the brain stem tells the frontal cortex. i. oculomotor signals sent from superior colliculus to frontal eye field via mediodorsal thalamus. J Neurophysiol 91(3): 1381–1402
Sommer MA, Wurtz RH (2004b) What the brain stem tells the frontal cortex. ii. role of the sc-md-fef pathway in corollary discharge. J Neurophysiol 91(3): 1403–1423
Sommer MA, Wurtz RH (2006) Influence of the thalamus on spatial visual processing in frontal cortex. Nature 444: 374–377
Stricanne B, Andersen RA, Mazzoni P (1996) Eye-centered, head-centered, and intermediate coding of remembered sound locations in area lip. J Neurophysiol 76(3): 2071–2076
Tanaka M (2007) Spatiotemporal properties of eye position signals in the primate central thalamus. Cereb Cortex 17(7): 1504–1515
Thelen E, Schöner G, Scheier C, Smith LB (2001) The dynamics of embodiment: a field theory of infant perseverative reaching. Behav Brain Sci 24(1): 1–34
Trappenberg TP, Dorris MC, Munoz DP, Klein RM (2001) A model of saccade initiation based on the competitive integration of exogenous and endogenous signals in the superior colliculus. J Cogn Neurosci 13(2): 256–271
Umeno MM, Goldberg ME (2001) Spatial processing in the monkey frontal eye field. II. Memory responses. J Neurophysiol 86(5): 2344–2352
Wang XJ (2001) Synaptic reverberation underlying mnemonic persistent activity. Trends Neurosci 24(8): 455–463
White III RL, Snyder LH (2004) A neural network model of flexible spatial updating. J Neurophysiol 91(4): 1608–1619
Wilimzig C, Schneider S, Schöner G (2006) The time course of saccadic decision making: dynamic field theory. Neural Netw 19(8): 1059–1074
Wilson HR, Cowan JD (1973) A mathematical theory of the functional dynamics of cortical and thalamic nervous tissue. Biol Cybern 13(2): 55–80
Wurtz RH (2008) Neuronal mechanisms of visual stability. Vision Res 48(20): 2070–2089
Xing J, Andersen RA (2000) Models of the posterior parietal cortex which perform multimodal integration and represent space in several coordinate frames. J Cogn Neurosci 12(4): 601–614
Zipser D, Andersen RA (1988) A back-propagation programmed network that simulates response properties of a subset of posterior parietal neurons. Nature 331(6158): 679–684
Acknowledgments
The authors acknowledge support from the German Federal Ministry of Education and Research within the National Network Computational Neuroscience—Bernstein Fokus: “Learning behavioral models: From human experiment to technical assistance,” grant FKZ 01GQ0951.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Schneegans, S., Schöner, G. A neural mechanism for coordinate transformation predicts pre-saccadic remapping. Biol Cybern 106, 89–109 (2012). https://doi.org/10.1007/s00422-012-0484-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-012-0484-8