Abstract.
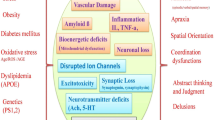
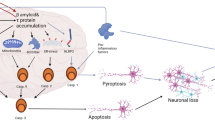
Multiple studies suggest that neuronal death in Alzheimer's disease (AD) is the result of an apoptotic mechanism. However, the stereotypical manifestations that define the terminal phases of apoptosis, such as chromatin condensation, apoptotic bodies, and blebbing, are not seen in AD. In this study, we show that the caspases, such as caspase 6, which cleave amyloid-β protein precursor (AβPP) and presenilins, are localized to the pathological lesions associated with AD. However, while upstream caspases such as 8 and 9 are clearly found in association with the intraneuronal pathology in AD, downstream caspases such as 3, 6 and 7 are present only at control levels. Given that execution of apoptosis requires amplification of the caspase-mediated apoptotic signal, our results indicate that in AD there is a lack of effective apoptotic signal propagation to downstream caspase effectors. Therefore, while the presence of caspases, especially caspase 6, in association with extracellular deposits of amyloid-β, could obviously have important ramifications on the proteolytic processing of AβPP and, thereby, on disease pathogenesis, it seems that AD represents the first in vivo situation reported in which the initiation of apoptosis does not proceed to caspase-dependent cell death. This novel phenomenon of apoptotic avoidance, which we term abortive apoptosis, or abortosis, may represent an exit from the caspase-induced apoptotic program that leads to neuronal survival in AD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Raina, A., Hochman, A., Zhu, X. et al. Abortive apoptosis in Alzheimer's disease. Acta Neuropathol 101, 305–310 (2001). https://doi.org/10.1007/s004010100378
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s004010100378