Abstract
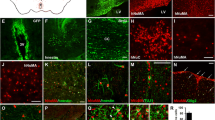
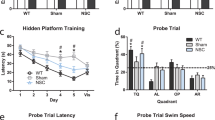
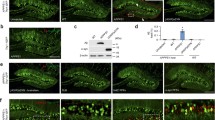
Abnormal processing of β-amyloid precursor protein (APP) and precipitation of amyloidogenic βA4 peptide have been implicated in the pathogenesis of Alzheimer’s disease (AD). In an attempt to generate an in vivo model of APP-associated pathology, we have introduced recombinant retroviral vectors harboring normal and mutant cDNAs for human neuron-specific APP 695 into fetal rat brain transplants. A minor population of recombinant neural cells with expression of APP was identified immunohistochemically in the grafts. The expression was maintained during an observation period of 9 months. Neurons with strong immunoreactivity for human APP exhibited markedly swollen perikarya and showed signs of cell degeneration. At 6 months post-transplantation, recombinant grafts developed APP-positive neuropil deposits with morphological features of neuritic plaques, but without βA4 peptide immunoreactivity. No difference was observed between wild-type APP and the mutant APP construct derived from a family with autosomal dominant AD. These observations indicate that long-term neuronal overexpression of human APP has the potential to generate APP plaques in the rodent brain.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 22 January 1996 / Accepted: 5 February 1996
Rights and permissions
About this article
Cite this article
Bayer, T., Foßgreen, A., Czech, C. et al. Plaque formation in brain transplants exposed to human β-amyloid precursor protein 695. Acta Neuropathol 92, 130–137 (1996). https://doi.org/10.1007/s004010050500
Issue Date:
DOI: https://doi.org/10.1007/s004010050500