Abstract.
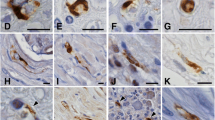
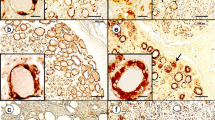
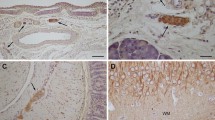
In Schwann cells, the transmembrane glycoprotein β-dystroglycan composes the dystroglycan complex together with the extracellular glycoprotein α-dystroglycan, which binds laminin-2 (α2/β1/γ1), a major component of the Schwann cell basal lamina. In the Schwann cell cytoplasm, β-dystroglycan is anchored to a dystrophin isoform, Dp116. In this study, we investigated the expression of β-dystroglycan, Dp116 and the laminin-α2 chain in satellite cells of rat dorsal root ganglia (DRGs). Immunohistochemical study showed that immunoreactivities for β-dystroglycan and Dp116 were both localized to the outer rim of neuron-satellite cell and axon-Schwann cell units, indicating that both satellite and Schwann cells expressed these proteins in DRGs. Immunoreactivity for the laminin-α2 chain was detected in a similar location, indicating that the basal lamina surrounding satellite and Schwann cells in DRGs contained laminin-2. Ultrastructurally, immunoreactivity for the cytoplasmic domain of β-dystroglycan as well as that for Dp116 was most intense in the cytoplasm just underlying the outer membrane of satellite cells. The immunoreactivity for laminin was associated with the outer surface of those cells, suggesting that it was localized in the surrounding basal lamina. These results indicate that the dystroglycan complex is expressed in the satellite cell outer membrane and involved in the adhesion with the basal lamina through the interaction with laminin-2.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Revised, accepted: 28 June 2000
Electronic Publication
Rights and permissions
About this article
Cite this article
Masaki, T., Matsumura, K., Hirata, A. et al. Expression of dystroglycan complex in satellite cells of dorsal root ganglia. Acta Neuropathol 101, 174–178 (2001). https://doi.org/10.1007/s004010000276
Received:
Issue Date:
DOI: https://doi.org/10.1007/s004010000276