Abstract
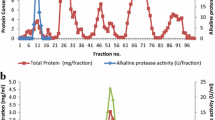
Alkalophilic Bacillus sp. KSM-K16 produced three alkaline proteases, as detected by polyacrylamide gel electrophoresis (PAGE). The major protease, designated M protease, was recently purified to homogeneity and its properties were characterized. In the present study, two minor proteases, designated H protease and N protease, were purified to homogeneity from cultures of this organism. H protease had a molecular mass of 28 kDa, as estimated by sodium dodecyl sulfate/PAGE (SDS-PAGE) and its maximum activity against casein was observed at pH 11.0 and at 55°C. N protease consisted of two polypeptide chains with molecular masses of 12.5 kDa and 14.5 kDa, as estimated by SDS-PAGE, although it migrated as a single protein band during non-denaturing PAGE. Its maximum activity was observed at pH 11.0 and at 60°C. The amino-terminal sequences of H protease and of the 14.5-kDa polypeptide of N protease were identical to that of M protease. The electrophoretic relationship between the three enzymes was examined after they had been stored at different pH values and at 5°C. M protease was converted to H protease more rapidly at pH 11 than at pH 8 or below, and H protease was converted to M protease at pH 8 or below but not at pH 11. N protease appeared to be the autolytic product of the M and H proteases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Edelhoch H (1967) Spectroscopic determination of tryptophan and tyrosine in proteins. Biochemistry 6: 1948–1954
Flatmark T (1964) On the heterogeneity of beef heart cytochrome c. I. Separation and isolation of subfractions by disc electrophoresis and column chromatography. Acta Chem Scand 18: 1656–1666
Flatmark T (1966) On the heterogeneity of beef heart cytochrome c. II. Some physico-chemical properties of the main subfractions (CyI-CyIII). Acta Chem Scand 20: 1476–1486
Flatmark T, Sletten K (1968) Multiple forms of cytochrome c in rat. J Biol Chem 243: 1623–1629
Funakoshi S, Deutsch HF (1969) Human carbonic anhydrases. J Biol Chem 244: 3438–3446
Hakamada Y, Kobayashi T, Hitomi J, Kawai S, Ito S (1994) Molecular cloning and nucleotide sequence of the gene for an alkaline protease from the alkalophilic Bacillus sp. KSM-K16. J Ferment Bioeng 78: 105–108
Hattori M, Ogawa H (1980) Characteristics of fibrous protein and the disulphide cross-linking band in human Stratum corneum. In: Bernstein IA, Seiji M (eds) Biochemistry of normal and abnormal epidermal differentiation. University of Tokyo Press, Tokyo, pp 379–390
Ichishima E, Maeba H, Amikura T, Sakata H (1984) Multiple forms of protyrosinase from Aspergillus oryzae and their mode of activation at pH 3.0. Biochim Biophys Acta 786: 25–31
Ichishima E, Hanzawa S, Watanabe M, Takeuchi M (1986) Electrophoretic heterogeneity and aged-related change of serine proteinase from Aspergillus sojae. Curr Microbiol 13: 231–235
Kobayashi T, Hakamada Y, Adachi S, Hitomi J, Yoshimatsu T, Koike K, Kawai S, Ito S (1995) Purification and properties of an alkaline protease from alkalophilic Bacillus sp KSM-K16. Appl Microbiol Biotechnol 43: 473–481
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Lowry OH, Rosebrough NJ, Farr AL, Randall JR (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193: 265–275
Makino K, Ogaki J, Nishihara T, Ichikawa T, Kondo M (1983) Studies on protease from marine bacteria 2. Properties of extracellular protease from marine Pseudomonas sp 145-2. Microbios 36: 7–20
Taber HW, Sherman F (1964) Spectrophotometric analyzers for disk electrophoresis; studies of yeast cytochrome c. Ann NY Acad Sci 121: 600–615
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedures and some applications. Proc Natl Acad Sci USA 76: 4350–4354
Yamagata H, Tanaka K, Kasai Z (1979) Isoenzymes of acid phosphatase in aleurone particles of rice grains and their interconversion. Agric Biol Chem 43: 2059–2066
Yamane T, Kani T, Hatanaka T, Suzuki A, Ashida T, Kobayashi T, Ito S, Yamashita O (1995) Structure of a new alkaline serine protease (M-protease) from Bacillus sp KSM-K16. Acta Crystallogr D 51: 199–206
Yang JT, Wu CSC, Martinez HM (1986) Calculation of protein conformation from circular dichroism. Methods Enzymol 130: 208–269
Yoshida K, Hidaka H, Miyado S, Shibata U, Saito K, Yamada Y (1977) Purification and some properties of Bacillus sphaericus protease. Agric Biol Chem 41: 745–754
Zuidweg MHJ, Bos CJK, Van Welzen H (1972) Proteolytic components of alkaline proteases of Bacillus strains. Zymograms and electrophoretic isolation. Biotechnol Bioeng 14: 685–714
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kobayashi, T., Hakamada, Y., Hitomi, J. et al. Purification of alkaline proteases from a Bacillus strain and their possible interrelationship. Appl Microbiol Biotechnol 45, 63–71 (1996). https://doi.org/10.1007/s002530050650
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002530050650