Abstract.
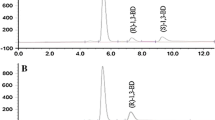
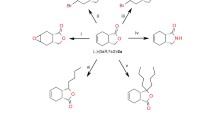
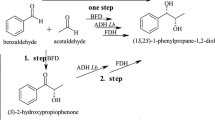
Diastereoselective reduction of diketones with Lactobacillus kefir DSM 20587 was examined. The reduction of both oxo-functions proceeded highly diastereoselectively. (2R,5R)-Hexanediol 3 was produced starting from (2,5)-hexanedione 1 in quantitative yields with enantiomeric excess >99% and diastereomeric excess >99%. The reaction conditions were optimized: maximum yield of (2R,5R)-hexanediol was reached at pH 6, 30°C and with equal amounts of substrate and cosubstrate. The applicability of the system in fed-batch experiments was demonstrated. The feed specific biomass concentration required to reach maximal yield and selectivity in fed-batch mode was determined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Haberland, .J., Kriegesmann, .A., Wolfram, .E. et al. Diastereoselective synthesis of optically active (2R,5R)-hexanediol. Appl Microbiol Biotechnol 58, 595–599 (2002). https://doi.org/10.1007/s00253-002-0936-5
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00253-002-0936-5