Abstract
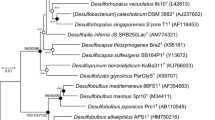
Acetate-degrading anaerobic microorganisms in freshwater sediment were quantified by the most probable number technique. From the highest dilutions a methanogenic, a sulfate-reducing, and a nitrate-reducing microorganism were isolated with acetate as substrate. The methanogen (culture AMPB-Zg) was non-motile and rod-shaped with blunted ends (0.5–1 μm × 3–4 μm long). Doubling times with acetate at 30–35°C were 5.6–8.1 days. The methanogen grew only on acetate. Analysis of the 16S rRNA sequence showed that AMPB-Zg is closely related toMethanosaeta concilii. The isolated sulfate-reducing bacterium (strain ASRB-Zg) was rod-shaped with pointed ends (0.5–0.7 μm × 1.5–3.5 μm long), weakly motile, spore forming, and gram positive. At the optimum growth temperature of 30°C the doubling times with acetate were 3.9–5.3 days. The bacterium grew on a range of organic acids, such as acetate, butyrate, fumarate, and benzoate, but did not grow autotrophically with H2, CO2, and sulfate. The closest relative of strain ASRB-Zg isDesulfotomaculum acetoxidans. The nitrate-reducing bacterium (strain ANRB-Zg) was rod-shaped (0.5–0.7 μm × 0.7–1 μm long), weakly motile, and gram negative. Optimum growth with acetate occurred at 20–25°C. The bacterium grew on a range of organic substrates, such as acetate, butyrate, lactate, and glucose, and did grow autotrophically with H2, CO2, and oxygen but not with nitrate. In the presence of acetate and nitrate, thiosulfate was oxidized to sulfate. Phylogenetically, the closest relative of strain ANRB-Zg isVariovorax paradoxus.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Azegami K, Nishiyama K, Watanabe Y, Kadota I, Ohuchi A, Fukazawa C (1987)Pseudomonas plantarii sp. nov., the causal agent of rice seedling blight. Int J Syst Bacteriol 37:144–152
Bazylinski DA, Palome E, Blakemore NA, Blakemore RP (1986) Denitrification byChromobacterium violaceum. Appl Environ Microbiol 52:696–699
Brysch K, Schneider C, Fuchs G, Widdel F (1987) Lithoautotrophic growth of sulfate-reducing bacteria, and description ofDesulfobacterium autrophicum gen. nov., sp. nov. Arch Microbiol 148:264–274
Cavalca L, Hartmann A, Rouard N, Soulas G (1999) Diversity of tfdC genes: distribution and polymorphism among 2,4-dichlorophenoxyacetic acid degrading soil bacteria. FEMS Microbiol Ecol 29:45–58
Finster K, Coates JD, Liesack W, Pfennig N (1997)Desulfuromonas thiophila sp. nov, a new obligately sulfur-reducing bacterium from anoxic freshwater sediment. Int J Syst Bacteriol 47:754–758
Hungate RE (1969) A roll tube method for cultivation of strict anaerobes. In: Norris JR, Ribbons DW (eds) Methods in Microbiology, Vol 3b. Academic Press, New York, pp. 117–132
Huser BA, Wuhrmann K, Zehnder AJB (1982)Methanothrix soehngenii gen. nov. sp. nov., a new acetotrophic nonhydrogen-oxidizing methane bacterium. Arch Microbiol 132:1–9
Jetten MSM, Stams AJM, Zehnder AJB (1992) Methanogenesis from acetate—a comparison of the acetate metabolism inMethanothrix soehngenii andMethanosarcina spp. FEMS Microbiol Rev 88:181–197
Jones WJ (1991) Diversity and physiology of methanogens. In: Rogers JE, Whitman WB (eds) Microbial Production and Consumption of Greenhouse Gases: Methane, Nitrogen Oxides, and Halomethanes. American Society for Microbiology, Washington, DC, pp 39–55
Jukes T, Cantor CR (1969) Evolution of protein molecules. In: Munro HN (ed) Mammalian Protein Metabolism. Academic Press, New York, pp 21–132
Kamagata Y, Fulthorpe RR, Tamura K, Takami H, Forney LJ, Tiedje JM (1997) Pristine environments harbor a new group of oligotrophic 2,4-dichlorophenoxyacetic acid-degrading bacteria. Appl Environ Microbiol 63:2266–2272
Kiene RP (1991) Production and consumption of methane in aquatic systems. In: Rogers JE, Whitman WB (eds) Microbial Production and Consumption of Greenhouse Gases: Methane, Nitrogen Oxides, and Halomethanes. American Society for Microbiology, Washington, DC, pp 111–146
Nogales B, Moore ERB, Abraham W-F, Timmis KN (1999) Identification of the metabolically active members of a bacterial community in a polychlorinated biphenyl-polluted moorland soil. Environ Microbiol 1:199–212
Oude Elferink SJWH, Maas RN, Harmsen HJM, Stams AJM (1995)Desulforhabdus amnigenus gen nov sp nov, a sulfate reducer isolated from anaerobic granular sludge. Arch Microbiol 164:119–124
Oude Elferink SJWH, Luppens SBI, Marcelis CLM, Stams AJM (1998) Kinetics of acetate oxidation by two sulfate reducers isolated from anaerobic granular sludge. Appl Environ Microbiol 64:2301–2303
Oude Elferink SJWH, Akkermans-van Vliet WM, Bogte JJ, Stams AJM (1999)Desulfobacca acetoxidans gen. nov. sp. nov., a novel acetate-degrading sulfate reducer isolated from sulfidogenic granular sludge. Int J Syst Bacteriol 49:345–350
Pichinoty F, Mandel M, Garcia J-L (1979) The properties of novel denitrifyingBacillus cultures found in tropical soils. J Gen Microbiol 115:419–430
Reinhold B, Hurek T, Fendik I, Pot B, Gillis M, Kersters K, Thielemans S, De Ley J (1987)Azospirrilum halopraeferens sp. nov., a nitrogen-fixing organism associated with roots of Kallar grass (Leptochloa fusca (L.) Kunth). Int J Syst Bacteriol 37:43–51
Robertson LA, Kuenen JG (1992) The colorless sulfur bacteria. In: Balows A, Trüper HG, Dworkin M, Harder W, Schleifer K-H (eds) The Prokaryotes, Vol 1. Springer, Berlin, pp 385–413
Rothfuss F, Conrad R (1993) Thermodynamics of methanogenic intermediary metabolism in littoral sediment of lake constance. FEMS Microbiol Ecol 12:265–276
Sambrook J, Fritsch EJ, Maniatis T (1989) Moleculair Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Scholten JCM, Stams AJM (1995) The effect of sulfate and nitrate on methane formation in a freshwater sediment. Antonie Van Leeuwenhoek 68:309–315
Scholten JCM, van Bodegom PM, Vogelaar J, van Ittersum A, Hordijk K, Roelofsen W, Stams AJM (2000) Effect of sulphate and nitrate on acetate consumption by methanogenic, sulphate-and nitrate-reducing populations in mixed freshwater sediment. Biogeochem, in press.
Scholten JCM, Conrad R, Stams AJM (2000) Effect of 2-bromoethane sulfonate, molybdate and chloroform on acetate consumption by methanogenic and sulfate-reducing communities in a freshwater sediment. FEMS Microbiol Ecol 32:35–42
Schütz H, Seiler W, Conrad R (1989) Processes involved in formation and emission of methane in rice paddies. Biogeochem 7:33–53
Stams AJM, Vandijk JB, Dijkema C, Plugge CM (1993) Growth of syntrophic propionate-oxidizing bacteria with fumarate in the absence of methanogenic bacteria. Appl Environ Microbiol 59:1114–1119
Stevenson FJ (1994) Humus Chemistry; Genesis, Composition, Reactions, 2nd ed. John Wiley & Sons, New York
Strunk O, Gross O, Reichel B, May M, Hermann S, Stuckman N, Nonhoff B, Lenke M, Finhart A, Vilbig A, Ludwig T, Bode A, Schleifer K-H, Ludwig W (1998) ARB: a software environment for sequence data. http://www.mikro.biologie.tumuenchen.de. Department of Microbiology, Technische Universität München, Munich, Germany.
Thauer RK, Moller-Zinkhan D, Spormann AM (1989) Biochemistry of acetate catabolism in anaerobic chemotrophic bacteria. Ann Rev Microbiol 43:43–67
Tomlinson GA, Jahnke LL, Hochstein LI (1986)Halobacterium denitrificans sp. nov., an extremely halophilic denitrifying bacterium. Int J Syst Bacteriol 36:66–70
Torreblanca M, Rodriguez-Valera F, Juez G, Ventosa A, Kamekura M, Kates M (1986) Classification of non-alkaliphilic Halobacteria based on numerical taxonomy and polar lipid composition, and descriptionof Haloarcula gen. nov. andHaloferax gen. nov. Syst Appl Microbiol 8:89–99
Van der Meer JR (1994) Genetic adaptation of bacteria to chlorinated aromatic compounds. FEMS Microbiol Rev 15:239–249
Widdel F (1988) Microbiology and ecology of sulfate- and sulfur-reducing bacteria. In: Zehnder AJB (ed) Biology of Anaerobic Microorganisms. John Wiley & Sons, New York, pp 469–585
Widdel F (1992) The genusDesulfotomaculum. In: Balows A, Trüper HG, Dworkin M, Harder W, Schleifer K-H (eds) The Prokaryotes, Vol 2. Springer, Berlin, pp 1792–1799
Widdel F, Hansen TA (1992) The dissimilatory sulfate- and sulfur-reducing bacteria. In: Balows A, Trüper HG, Dworkin M, Harder W, Schleifer K-H (eds) The Prokaryotes, Vol 1. Springer, Berlin, pp 583–624
Wolin EA, Wolin MJ, Wolfe RS (1963) Formation of methane by bacterial extracts. J Biol Chem 238:2882–2886
Wind T, Stubner S, Conrad R (1999) Sulfate-reducing bacteria in rice field soil and on rice roots. Syst Appl Microbiol 22:269–279
Zumft WG (1992) The denitrifying prokaryotes. In: Balows A, Trüper HG, Dworkin M, Harder W, Schleifer K-H (eds) The Prokaryotes, Vol 1. Springer, Berlin, pp 554–581
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scholten, J.C.M., Stams, A.J.M. Isolation and characterization of acetate-utilizing anaerobes from a freshwater sediment. Microb Ecol 40, 292–299 (2000). https://doi.org/10.1007/s002480000070
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002480000070