Abstract
Owing to their highly efficient catalytic effects and substrate specificity, the nucleic acid tool enzymes are applied as ‘nano-tools’ for manipulating different nucleic acid substrates both in the test-tube and in living organisms. In addition to the function as molecular scissors and molecular glue in genetic engineering, the application of nucleic acid tool enzymes in biochemical analysis has also been extensively developed in the past few decades. Used as amplifying labels for biorecognition events, the nucleic acid tool enzymes are mainly applied in nucleic acids amplification sensing, as well as the amplification sensing of biorelated variations of nucleic acids. With the introduction of aptamers, which can bind different target molecules, the nucleic acid tool enzymes-aided signal amplification strategies can also be used to sense non-nucleic targets (e.g., ions, small molecules, proteins, and cells). This review describes and discusses the amplification strategies of nucleic acid tool enzymes-aided biosensors for biochemical analysis applications. Various analytes, including nucleic acids, ions, small molecules, proteins, and cells, are reviewed briefly. This work also addresses the future trends and outlooks for signal amplification in nucleic acid tool enzymes-aided biosensors.

Nucleic acid tool enzymes-aided signal amplification sensing
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
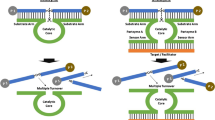
Nucleic acid tool enzyme is a kind of tool that can precisely cut, splice, and modify nucleic acids, and is widely used in genetic engineering. The nucleic acid tool enzymes include restriction enzymes, ligases, polymerases, nucleases, modifying enzymes, and so on. Different tool enzymes have different functions and characteristics in biology. Figure 1 schematically summarizes several prevalently used nucleic acid tool enzymes, demonstrating the unique substrate specificity and some substrate preferences. For example, polymerase replicates duplex DNA structures using nucleic acid templates as a guide [1], ligase facilitates the joining of DNA strands together by catalyzing the formation of a phosphodiester bond [2], and telomerase elongates single strands of nucleic acids along the template of its own RNA molecule [3]. The sequence-specific scission or nicking of duplex DNA substrates can hydrolyze by restriction endonucleases or nicking endonuclease, respectively [4, 5]. The duplex-specific nuclease (DSN) hydrolyzes only double strands DNA (dsDNA) or DNA in DNA/RNA hybrids, regardless of nucleotide sequence, and does not cleave single-stranded DNA (ssDNA) or RNA at all [6]. Furthermore, the RNase (RNase H) recognizes RNA/DNA heteroduplexes and specifically cleaves the RNA strands into smaller fragments [7]. Last but not least, exonuclease is a kind of sequence-independent tool enzyme that breaks phosphodiester bonds at the 3′ end or the 5′ end and cleaves nucleotides one at a time from the end of polynucleotide chain [8, 9]. Although these exonucleases do not require a specific recognition sequence in their substrates, they still display some substrate preference. For example, λ exonuclease needs a cleavable dsDNA strand to have a phosphate group on its 5′ end.
With the development of the tool enzymes, different nucleic acid tool enzymes are applied as ‘nano-tools’ for manipulating different nucleic acid substrates both in the test-tube and in living organisms. After the action of nucleic acid tool enzymes, the nucleic acid substrates will generate a series of structure switching, including the length of strand and the secondary and tertiary structures of substrates. Besides, they will also generate some by-products, such as repetitive ssDNA and free target, which can be used to initiate the subsequent reaction or target recycling. Thus, the diversity of nucleic acid tool enzymes can be widely applied on nucleic acids-based manipulation and nucleic acids-related signal amplification sensing. Compared with other signal amplification strategies, such as enzyme-linked immunosorbent assays [10, 11], immuno-PCR [12], mass spectrometric techniques [13], and nanomaterial-based strategies [14], the nucleic acid tool enzymes-aided signal amplification strategies have many remarkable merits: (1) the nucleic acid tool enzymes have highly efficient catalytic effects for substrates, thus the sensitivity can be guaranteed; (2) the reaction condition of enzymes involved is mild; (3) some of these enzymes display sequence-specificity or substrate preference, which can be used to design sensing mechanisms directly; (4) derived from lives cells, the nucleic acid tool enzymes display better biocompatibility in biochemistry analysis. So, the nucleic acid tool enzymes-aided signal amplification strategies have been studied in detail and developed in recent years [15–17].
The substrates of nucleic acid tool enzymes are nucleic acids, the nucleic acid tool enzymes-aided signal amplification strategies are then mainly used for nucleic acid (DNA or RNA) amplification sensing, as well as the amplification sensing of biorelated variations of nucleic acids such as DNA methylation and single nucleotide polymorphisms (SNPs). Moreover, some of the ions can mediate formation of non-natural base pairs in DNA duplexes, such as T-Hg2+-T or C-Ag+-C complexes [18]. Nucleic acid sequences that specifically bind Pb2+, Mg2+, or Cu2+ ions were found to yield supramolecular coiled structures that cleave specific DNA sequences [19]. The nucleic acid tool enzymes therefore can be used for amplification detection of ions. Recently, aptamers, the single stranded nucleic acid molecules selected from 1012 to 1015 combinatorial oligonucleotide libraries by a process called systematic evolution of ligands by exponential enrichment (SELEX) [20], have attracted more and more attention because of their homothetic characteristic of antibodies. In addition to binding to complementary nucleic acids, the aptamers can bind to non-nucleic acid targets, such as metal ions, small molecules, proteins, or even whole cells, with high affinity and specificity [21]. So, the introduction of aptamers provides unprecedented opportunities for nucleic acid tool enzymes to sense non-nucleic acid targets and dramatically widens the application areas of nucleic acid tool enzymes-aided signal amplification strategies.
In this review, we focus on the recent development of nucleic acid tool enzymes-aided signal amplification strategies in nucleic acid-related sensing and aptamer-based sensing. Various analytes detection by nucleic acid tool enzymes-aided signal amplification strategies are summarized (Fig. 2), such as DNA sensing, miRNA detection, SNPs identification, DNA methylation analysis, ions detection, small molecules sensing, and proteins and cells detection.
The detection of DNA
As one of the most exciting biopolymers in life science, DNA is not only the carrier of genetic information but also holds great promise for future applications in nanotechnology, material science, and biomedical analysis. Rapid growth of available sequence data has made the detection of specific nucleic acid sequences critical for gene therapy, mutation analysis, and clinical diagnosis [22–25]. Among the many methods devised for the detection of nucleic acids, amplification assays have received much attention because of the highest analytical sensitivity. As the classic target amplification method, polymerase chain reaction (PCR) can amplify trace amounts of DNA to levels that are detectable and improve the sensitivity to reach the level of single molecule detection limits [26]. However, PCR-based methods require special thermal cycling equipment and are prone to yielding false-positive results from artifact amplification, which may restrict its further application [27]. Thus, the design of alternative approaches for the sensitive detection of DNA with rapid, easy manipulation is in continuous demand. Recently, enzyme conjugates [28], DNAzymes [29], nucleic acid tool enzymes [16, 30], and nanomaterials [31] have been used as amplifying labels for biorecognition, and a series of amplified methods have been developed for DNA sensing. For example, Wang et al. explored a series of electrochemical approaches for the sensitive detection of DNA by coupling of several carbon nanotubes (CNT)-derived amplification processes [32, 33]. Mao and Tian reported an improved catalytic molecular beacon for DNA/RNA detection [34]. When the target DNA was presented, the DNAzymes became activated and constantly amplified the output signal through circulatory cleavage of molecular beacon, which greatly improved the detection sensitivity. Willner et al. have reported an amplified detection method for DNA sensing by the application of rotating functional magnetic particles [35]. Compared with signal amplification by DNAzymes and namomaterials, nucleic acid tool enzymes-aided signal amplification strategies have attracted considerable attention due to easy operation, high catalytic efficiency, and isothermal reaction condition. In this approach, each target molecule initiates multiple signaling events, and the sensitivity of DNA detection is thereby significantly increased [36]. By combining nucleic acid tool enzymes with different assay platforms, DNA can be quantified rapidly using colorimetric, fluorescent, or electrochemical assays (Table 1).
Nucleic acid tool enzymes-mediated colorimetric DNA assays
Colorimetric analysis is a simple process because the results can be monitored with the naked eye [58]. The inherent superiorities of colorimetry, such as simplicity, inexpensive, easy manipulation, and little or no required instrumentation, make it very attractive for DNA detection [59]. With the help of nucleic acid tool enzymes, the sensitivity of colorimetry gained great improvement. For example, Bi et al. reported a label-free colorimetric method for DNA detection based on target DNA triggered exonuclease-aided cascaded recycling amplification (Fig. 3) [38]. In this assay, they designed three kinds of unlabeled stem-loop DNA molecular beacons (denoted as MB1, MB2, and MB3, respectively). In the presence of target, MB1 was opened by hybridizing with target to form duplex DNA with a blunt 3′ terminus. Then, the Exo III bound to the duplex region of this hybrid and partially hydrolyzed the MB1 from the blunt 3′ terminus, releasing the target and trigger 1.
Illustration of target DNA triggered exonuclease-aided cascaded recycling amplification. Reprinted from [38] with permission
The released target could catalyze a new cycle of probe transformation (recycling I). The trigger 1 hybridized with MB2 at its 3′ terminus to initiate the recycling II, which resemble recycling I. After recycling II, the trigger 2 was released and the recycling III was activated. In recycling III, the liberated single-stranded trigger was the same as that of target DNA. So the trigger, released from recycling III, in turn triggered recycling I and repeated the above cyclic process to achieve cascaded amplification. After finishing the whole network, trigger 1 could form the catalytic DNAzyme units in the presence of hemin and oxidize 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) disodium salt (ABTS2–) to produce the colored product, ABTS–, which has a maximal absorption at 419 nm.
Compared with those organic color reagents, optical nanomaterials such as gold nanoparticles (AuNPs) have high brightness, lower cytotoxicity, and better biocompatibility, making them suitable for constructing biosensors [60, 61]. For instance, the ultrahigh extinction coefficient (108–109) of the surface plasmon resonance (SPR) absorption of AuNPs provides an alternative opportunity for improving traditional colorimetric method [62]. In 2009, Liu et al. reported a homogeneous colorimetric DNA biosensor using nicking endonuclease-aided nanoparticle amplification, which allowed high sensitivity detection of long, single stranded oligonucleotides with single-base mismatch selectivity [37]. In this detection system, the nicking endonuclease was specifically designed to cleave only the linker DNA. In the absence of target DNA, the intact linker DNA would trigger oligonucleotide-modified gold nanoparticles aggregation and a concomitant color change. If the target was present, the linker strand would be cleaved by nicking endonuclease and particle aggregation would not occur.
Nucleic acid tool enzymes-mediated fluorescent DNA detection
Although colorimetric assays are amenable to point-of-care (POC) testing, the low sensitivity of colorimetry precludes it from being a strong competitor among various analysis strategies. Fluorescent analysis strategies are typically based on the measurement of the fluorescence intensity induced upon specific binding between fluorescence-labeled probes and target molecules. Compared with colorimetric assays, fluorescence assays enable extraordinary sensitivity and multiplex detection capability as well as automation. The nucleic acid tool enzymes-aided signal amplification has also been adopted in fluorescence assays for improving the sensitivity of DNA detection. Two well-known nucleic acid tool enzymes-aided target recycling for DNA detection assays, the nicking enzyme assay [42] and exonuclease III assay [47], are illustrated in Fig. 4a and b, respectively. In the nicking enzyme assay, a nicking enzyme recognition sequence was embedded into the loop portion of the molecular beacon (MB). When the target DNA was presented, it hybridized with MB and formed a full recognition site for the nicking enzyme. The nicking enzyme nicked the beacon strand and separated fluorophore and quencher, generating a high fluorescence signal. The released target strand could then hybridize with another MB to initiate the next cycle of cleavage, resulting in an amplified fluorescence signal. The exonuclease III assay mainly utilizes the cleavage function of Exo III on blunt or recessed 3′ hydroxyl termini of duplex DNAs. In this assay, an MB was modified with a CAL Fluor Red 610 (FR610) fluorophore at its 5′ end and a Black Hole Quencher (BHQ) at an internal position. When the target DNA was absent, the MB self-hybridized to form a stem-loop structure with a 3′ protruding terminus, which could resist Exo III hydrolysis. This closed stem-loop structure held the fluorophore in close proximity to the quencher, resulting in low fluorescence. However, in the presence of target DNA, the stem-loop structure was opened and formed a double-stranded structure with a blunt 3′ terminus.
Then, Exo III catalyzed the hydrolysis of beacon strand from blunt 3′ terminus, releasing the fluorophore and the target DNA. The released target DNA then hybridized with another MB probe to initiate a new round of hydrolysis.
Even though the Exo III is assumed to be dsDNA-specific exonuclease in most literature, recent research shows that Exo III has the activity of hydrolyzing ssDNA [63]. The hydrolysis of ssDNA target is one of the main causes for the ceasing of target recycling amplification. In order to solve this problem and obtain higher amplification, Yang et al. developed a dual-signal amplification method for DNA detection based on the hydrolyzing activity on ssDNA of Exo III. In this method, an X sequence was specially designed that could resist the Exo III digestion. The X sequence could cyclically trigger the molecular beacons digestion for a long time, resulting in four orders of magnitude in the detection limit [50].
Nucleic acid tool enzymes-mediated electrochemical DNA sensing
Electrochemical biosensor-based DNA detection is popular because of its simplicity, rapid response, high sensitivity, and low fabrication cost [64].
The reaction is monitored electrochemically, based on the measurement of current (amperometry), potential (potentiometry), or changes in the conductive properties of the medium between the electrodes (conductometry) [65]. Many electrochemical DNA sensors have also been developed based on nucleic acid tool enzymes-aided amplification [52–55]. For example, Ye et al. proposed a rapid, single step, ultrasensitive electrochemical DNA sensor using the Exo III to achieve the aim of signal amplification. As depicted in Fig. 5, a signaling probe with a thiol group at 5′ terminus self-assembled onto the gold electrode. Then, the fabricated gold electrode was exposed in RuHex buffer to adsorb RuHex molecules, in which RuHex/DNA/electrode system gave an initial redox signal arising from electron transfer between RuHex molecules and the gold electrode. Upon addition of the target DNA and Exo III, the signaling probe hybridized with target DNA to form a double-stranded structure with a 3′ blunt end. The Exo III specifically recognized the duplex and catalyzed the hydrolysis of signaling probe from blunt 3′ terminus, releasing the target DNA. The released target DNA could hybridize with another signaling probe to initiate a new round of digest process. After manifold cycles, most of the signaling probes were hydrolyzed and only a small quantity of RuHex was adsorbed onto the gold electrode, resulting in a decrease redox signal. By employing Exo III-aided target recycling strategy, the sensitivity of the proposed sensor has been greatly improved, to the detection limit of 20 fM [66].
Schematic representation of the label-free electrochemical DNA sensor based on Exo III-aided target recycling. Reprinted from [66] with permission
In order to avoid the enzyme recognition sites located on the target DNA/capture DNA duplexes, the targets should contain special sequences. Our group introduced a ‘Y’ junction structure, formed by a capture probe, an assistant probe, and a target DNA, for detecting different target DNAs. In this assay, a methylene blue labeled capture probes self-assembled onto the gold electrode through Au-S linkages. Upon addition of target DNA and assistant probe to this system, the target DNA was complementary to the capture probes at one-half of the segment and complementary to the assistant probe at the other half-segment. Meanwhile, the rest of assistant probe also hybridized with the remaining segment of capture probe. The specific cleavage site for restriction endonuclease was located on the assistant probe/capture probe duplexes, and isolated from target DNA. Thus, the restriction endonuclease recognized the specific cleavage site in assistant probe/capture probe duplexes and digested the capture probe, releasing the methylene blue labeled piece. At the same time, the target DNA could also be released and initiate a new round of digest process. By changing the sequences of assistant and capture strands, the proposed sensor was suitable for detecting different target sequences with high sensitivity [52]. More recently, in order to avoid the limitation of “signal-off” strategy, the ratiometric electrochemical assay, combining “signal-on” and “signal-off” strategies, has also gained widespread development and application because of its good analytical performance [57, 67, 68].
The detection of miRNA
miRNAs are a group of short, endogenous, noncoding RNAs involved in a variety of biological processes. Recent studies have indicated that abnormal miRNA expression is highly correlated with various diseases [69]. Nowadays, miRNAs are regarded as biomarker candidates in clinical diagnosis and therapy [70–72]. However, the unique characteristics of miRNAs, such as their sequence homology, small size, low abundance in total RNA samples, and susceptibility to degradation, make them difficult to be detected [73]. The Northern blotting technology and microarrays are the traditional approaches for miRNA detection. But it is unsatisfactory because of their low sensitivity and specificity [74, 75]. Recently, various methods have been developed for miRNAs detection, such as nanoparticle-based amplification strategy [76] and nucleic acid tool enzymes-aided amplification strategy [77, 78]. Among these approaches, multiple self-assembly steps or complicated operational requirements are needed by the former. The nucleic acid tool enzymes-aided amplification strategies have attracted considerable attention because of easy operation and isothermal reaction. However, some of these nucleic acid tool enzymes, such as nicking endonucleases and restriction endonucleases, require a specific nucleotide sequence for recognition and cleavage. Limited by the intrinsic properties of the nucleases used, most of the previous DNA amplification detection strategies are not suitable for the detection of miRNA. Fortunately, some sequence-independent nucleic acid tool enzymes, such as Exo III, S1 nuclease, and (DSN), can catalyze the cleavage of phosphodiester bonds in either DNA or RNA and have been widely used for miRNA detection [79–82].
For example, DSN has a substrate preference for hydrolyzing dsDNA and DNA in DNA-RNA hybrid duplexes, and does not cleave ss-DNA or RNA. Based on this property of DSN, Ye et al. developed a single-step, sensitive method for miRNA detection [83]. As shown in Fig. 6a, a Taqman probe, modified with a fluorophore and a quencher, acted as signal output. In the presence of target miRNA, the Taqman probe hybridized to a target miRNA to form DNA/RNA heteroduplex, which could be recognized and digested by DSN. After digestion by DSN, the fluorophore and quencher were separated, resulting in significant fluorescence signal. Meanwhile, the target miRNA was released and started a new digest process. This method allowed the detection of miRNA in the femtomolar range within 30 min, nearly five orders of magnitude lower than the conventional method.
Moreover, multiplexed miRNAs detection could be easily realized by designing different fluorophore-labeled Taqman probes because DSN enzyme has no requirement for specific recognition sequence. Similarly, Yang et al. [84] designed a sensitive method for miRNA detection based on a specific molecular beacon (MB) and DSN-aided target recycling (Fig. 6b). Since DSN can also cleave dsDNA, the stem of the MB is prone to be recognized and digested by DSN. To overcome this problem, a specific MB, modified with 2-OMe-RNA on its stem, was designed to resist DSN hydrolysis. In the absence of miRNA, the MB showed low fluorescence intensity since the fluorophore and the quencher were in close proximity. In the presence of miRNA, the MB would bind to the target miRNA and produce a fluorescence signal because of the spatial separation of the fluorophore from the quencher. The formed MB–miRNA duplex would be recognized and digested by DSN, releasing the target miRNA. Subsequently, the released miRNA could hybridize with another MB, which leads to a cyclic reaction and fluorescent signal amplification. In order to further improve the sensitivity, by combining T7 exonuclease-aided cyclic amplification and rolling circle amplification strategy, they developed a dual amplification method for ultrasensitive detection of miRNA [85]. This dual amplification method could achieve a detection limit as low as 12 fM. More recently, based on polydopamine nanospheres-protected ssDNA probes and DNase I-aided signal amplification strategy, they developed a simple fluorescent method for sensitive miRNA detection [86].
The analysis of biorelated variations of nucleic acids
The analysis of single nucleotide polymorphisms
Because gene mutations play an important role in carcinogenesis, the identification of DNA variations is another important aspect of cancer therapy. Single nucleotide polymorphisms (SNPs) are the most common type of genetic variation, and they have been regarded as the key diagnostic markers for cancer progression, genetic disease, and pharmacogenomics [87–89]. SNPs identification is therefore of vital importance for early diagnosis, clinical prognostics, and disease treatment. Usually, the detection of single-base mismatch is the basis of all hybridization-based detection approaches for SNPs analysis. Up to now, many nucleic acid tool enzymes-based methodologies have been proposed for single base mismatch detection [90–93]. For example, Zhao et al. reported a novel colorimetric method for single-base mismatch detection based on S1 nuclease and gold nanoparticles (Fig. 7) [94].
Schematic illustration of the label-free optical detection of single-base mismatches. Reprinted from [94] with permission
S1 nuclease has a substrate preference for cleaving ssDNA and base-pair mismatches in dsDNA, and does not cleave perfectly matched dsDNA [95]. In previous work they have found that nucleoside monophosphates (dNMPs) could better stabilize unmodified gold nanoparticles (AuNPs) than ssDNA and dsDNA with the same base-composition and concentration. As shown in Fig. 7, the 16-bp perfectly matched duplex DNA could not be cleaved by S1 nuclease. The intact dsDNA was not able to stabilize AuNPs, leading the AuNP aggregation upon adding salt solution and resulting in purple-blue solution. In contrast, in the presence of single-base mismatched duplex DNA, the color of AuNPs slightly changed into red-purple because of the incomplete cleavage of the single-base mismatched dsDNA by S1 nuclease. Single-base mutations present in target DNA with varied sites were all effectively detected by this method.
In order to improve the detection sensitivity of AuNPs-based SNPs assay, Xiao et al. [96] presented a remarkably simple platform that enables sensitive naked-eye detection of SNPs based on the Exo III aided target recycle at room temperature. In this assay, one set of DNA capture probe (CP) was modified into the AuNPs with Au-S linkages. If the target sequence is perfectly matched to the CP sequence on the AuNPs, the intact dsDNA could digest by Exo III, leading to AuNP aggregation and yielding a blue color solution. However, in the case of a single-base mismatch, the sensor failed to aggregate and remained red. This method could achieve a detection limit as low as 2 nM by naked eye, which was 100 times lower than that unmodified AuNPs.
The detection of DNA methylation and DNA methyltransferases
DNA methylation is an important component of epigenetic regulation and plays an essential role in regulating gene expression in organisms [97]. DNA methylation is catalyzed by different DNA methyltransferases (MTases) [98, 99]. As abnormal DNA methylation can be regarded as biomarkers for early cancers and DNA MTases is regarded as a potential target for anticancer drugs, the detection of DNA methylation and assay of MTase activities is of significance for both fundamental biochemical research and drug discovery [100–102]. So far, the methods for detection of DNA methylation mainly include immuno-based recognition [103], methylation-sensitivity restriction enzyme digestion [104], bisulfite treatment [105, 106], and various polymerase chain reaction techniques [107]. For the immuno-based methods, the preparation of antibody is labor-intensive and expensive, which restrict its further application. In the restriction enzyme-based assay, a dsDNA with specific recognition site for restriction enzyme was usually designed. The restriction enzymes recognize the methylation/demethylation position, and selectively scissor the specific DNA sequence. When the specific position is demethylated/methylated, the cleavage will be inhibited. As in Fig. 8a [108], first, target DNA hybridized to the probe and formed dsDNA with two 3′ protruding termini, which could be resistant to hydrolysis by Exo III. Hpa II endonuclease was used to recognize the 5′-CCGG-3′ sequences and cleave them, resulting in two split short dsDNA with 3′ recessed termini. Then, Exo III could bind to the split dsDNA and hydrolyze it from recessed 3′ hydroxyl termini to generate ssDNA. The fluorescence intensity of the added thiazole orange (TO) was weak because of weak interaction between the TO and ssDNA. If dsDNA was methylated by M.SssI MTase, the function of Hpa II was inhibited and Exo III could not work. The added TO intercalated into its double strand and showed strong fluorescence intensity.
a Schematic illustration of the label-free fluorescent method for the detection of site-specific DNA methylation and MTase activity based on restriction enzyme and Exo III. Reprinted from [108] with permission. b Schematic illustration of DNA methylation assay through the treatment of bisulfite. Reprinted from [109] with permission
The methods based on restriction enzymes can accurately determine the methylation sites, but are confined to detect limited DNA methylation due to the selectivity of restriction enzymes. In the bisulfite-based assay, the cytosine (C) in substrate is deaminated and converted to uracil (U) through the treatment of bisulfite. If C was methylated (mC), mC remains almost unchanged. Therefore, this method could identify the methylated cytosine at any site in DNA and is therefore considered to be a gold standard assay. As in Fig. 8b [109], after the sodium bisulfite treatment, the cytosine in both methylated and unmethylated DNA was changed to uracil, but 5-methylcytosine (mC) remained unchanged. The methylated DNA specifically bound to the domain I of template and worked as a primer to initiate an extension reaction in the presence of dNTPs and polymerase. With the assistance of nicking enzyme I, the extension reaction could generate a large number of trigger, which could hybridize to the QF probes and initiate the nicking enzymeII-aided signal amplification.
The unmethylated DNA with six-base mismatch was unable to hybridize with the template and could not initiate the subsequent signal amplification. More recently, by combining the advantages of LCR amplification and AuNPs-based colorimetric strategy, Li et al. developed a low-cost method for detection of DNA methylation with high sensitivity [110].
In many cases, the DNA methylation states are linked to the activity of DNA MTase [111, 112]. Alterations of MTase activity may lead to abnormal DNA methylation. So, the DNA MTase has also been regarded as a potential therapeutic target in many types of cancer [113]. Strategies to detect DNA MTase are therefore also important in the area of clinical diagnostics. Recently, isothermal amplification techniques, such as strand displacement amplification (SDA) [114], rolling circle amplification (RCA) [115, 116], nicking enzyme signal amplification (NESA) [117, 118], and methylation-sensitivity restriction enzyme digestion [119–121], are frequently applied for DNA MTase assay because of the wide variety of choices. In 2013, Zhang et al. [116] developed a highly sensitive chemiluminescence method for the MTase assay based on hairpin probe (HP) and rolling circle amplification (RCA). In the presence of DNA MTase, the HP was methylated and cleaved by the methylation-sensitive restriction endonuclease. The cleaved HP could function as a primer to initiate RCA, which generated a large number of horseradish peroxidase-mimicking DNAzyme chains. The DNAzyme chains could fold into a biocatalytic G-quadruplex/hemin structure and catalyze the oxidation of luminal to generate distinct chemiluminescence signal in the presence of H2O2 and luminol. In the absence of Dam MTase, the methylation and cleavage reaction of HP would not occur. The intact HP could not function as a primer to initiate subsequent RCA, and no chemiluminescence signal was obtained. More recently, they developed a sensitive fluorescence method for DNA MTase detection based on DSN and T7 RNA polymerase aided signal amplification [122].
The amplification detection of ions
As is known, DNA forms a double-helix structure mainly through the formation of Watson-Crick base pairs, such as adenine-thymine (A-T) and guanine-cytosine (G-C) base pairs [123]. Recently, the Ono and Urata groups in a series of articles have reported that silver specifically stabilizes the cytosine-cytosine (C-C) mismatches (C-Ag-C) and mercury ions specifically stabilize the thymine-thymine (T-T) mismatches (T-Hg-T) in oligodeoxynucleotide duplexes [19, 124, 125]. In these non-natural base pairs, the hydrogen bonds present in natural DNA are replaced by coordinative bonds. These features were widely applied to develop T-/C-rich DNA for the detection of Hg2+ or Ag+ through the ion-induced conformational switch [126, 127]. Both the objective of metal ion mediated non-natural base pairs and nucleic acid tool enzymes’ substrates are nucleic acids, and the nucleic acid tool enzymes-aided amplification strategies were also applied in the detection of Hg2+ and Ag+ ions [128–132]. For example, Hsing and Xuan [128] reported an ultrasensitive electrochemical method for Hg2+ detection based on Hg2+ trigger digestion of the designed e-T-rich probe by Exo III (Fig. 9a). In this assay, an e-T-rich probe was modified with methylene blue at an internal T base close to the 3′ end. In the presence of Hg2+, the linear e-T-rich probe could fold into a hairpin structure with a blunt 3′ terminus and activate the hydrolytic ability of the Exo III enzyme, releasing a thymidine monophosphate with the methylene blue tag (eNT). The eNT possesses a higher diffusivity toward the negatively charged ITO electrode than that of the e-T-rich probe due to its less negative charge and smaller size.
Thus, methylene blue could close to the ITO electrode and generate a distinct increase in the electrochemical signal. The released Hg2+ can bind with other e-T-rich probe to initiate a new cycle of cleavage. However, in the absence of Hg2+, the linear e-T-rich probe could hardly get into contact with the ITO electrode and therefore a negligible electrochemical response was observed. Through the Exo III aided signal amplification, this immobilization-free electrochemical Hg2+ sensor platform has a detection limit of 0.2 nM and a total assay time within minutes. Similarly, Zhang et al. presented a new strategy for the detection of Ag+ based on ion-triggered conformation change and Exo III-aided target cycle.
In addition, since it was first found by two independent research groups in the late 20th century, the SELEX technique was widely used to select aptamers or DNAymes. The DNAzymes are specific single strand DNA sequences that can be activated in the presence of added ions. For example, nucleic acid sequences that specifically bind Pb2+, Mg2+, or Cu2+ ions were found to yield supramolecular coiled structures that cleave specific DNA sequences [18]. Based on this, researchers developed some new biosensors for the detection of these ions in recent years [133, 134] that overcome the limitations of the traditional protocols. Recently, by combining the powerful amplification of RCA and the highly specificity cleavage efficiency of ion-dependent DNAzyme, Zhang et al. [135] developed a novel, highly sensitive, and selective electrochemical sensing system for lead ions detection (Fig. 9b). In the absence of Pb2+, the DNAzyme was inactivated, and neither cleaving nor RCA reaction could be initiated. When the Pb2+ was present, the DNAzyme was activated and the substrate strand was cleaved into two DNA fragments. The free catalytic strand could be hybridized with the padlock probe. After addition of ligase, polymerase, and dNTPs, the RCA process was triggered, producing a long ssDNA with repeating sequence units. Subsequently, the signal probes (CdS QD-ssDNA) were hybridized with the long ssDNA and formed a long DNA duplex. After magnetic separation, the unhybridized CdS QD-ssDNA was removed; the captured CdS QDs were then dissolved by nitric acid. The released cadmium cations could be quantified by square wave voltammograms. In order to further simplify the detection process, Guo et al. employed a DNA circular template instead of the padlock probe, which makes the RCA process easier and more accurate for application in the detection [136]. Similarly, by combining the self-cleaving DNAzyme and the nucleic acid tool enzymes-aided reaction cycles, Cao et al. report a label-free fluorescent assay for the amplified detection of copper ions [137].
Aptamer-based signal amplification sensing
Aptamers are single-stranded nucleic acid molecules isolated from random-sequence nucleic acids libraries by SELEX [138, 139]. They generally have high binding affinity and selectivity for specific targets, ranging from small molecules to macromolecules, and even cells [21]. Being oligonucleotides, aptamers share the common properties of nucleic acids, including programmability through Watson-Crick base pairing, predictable secondary or tertiary structures, and high chemical stability. The binding of aptamers to their specific targets depends on their secondary and tertiary structures [140]. A variety of structure switching mechanisms can be developed to facilitate the transduction of target recognition events to either target- or signal-based amplifications [141–146]. Therefore, the introduction of aptamers provides unprecedented opportunities to detect non-nucleic acid targets, such as small molecules, proteins, and cell, which dramatically widen the application areas of nucleic acid tool enzymes-aided signal amplification strategies.
The amplification detection of small molecules
Evaluation of small molecules associated with different disease states can also serve as useful information for identification of disease and cancer pharmacogenomics [147].
Many small molecules, such as thrombin, adenosine, cocaine, and vascular endothelial growth factor, have been utilized as target for aptamers identification [148, 149]. Based on target molecules triggered aptamers structure switch and nucleic acid tool enzymes aided signal amplification, many novel signal amplification strategies have also been developed for these molecules [150–152]. For example, by integrating structure-switching aptamers with Exo III, Yang et al. developed a rapid, selective, and ultrahighly sensitive approach for nucleic acid and cocaine detection (Fig. 10) [48]. In the cocaine sensing assay, the aptamer of cocaine was been split into two fragments, fragment 1 and 2, to eliminate false negative signals. Fragment 2 was designed to be a linear molecular beacon with a fluorophore and quencher attached to 3′ terminal and penultimate nucleotides, respectively. In the absence of cocaine, the two fragments remained irrelevant with no fluorescent signal owing to the fluorophore and the quencher in close proximity. However, in the presence of cocaine, the two aptamer fragments interacted with the target and assembled into an aptamer–target complex. The linear molecular beacon formed a duplex-like structure with a blunt 3′ terminus, which could serve as the substrate of Exo III. Exo III hydrolyzed this duplex-like structure from the blunt 3′ terminus, liberating the fluorophore and releasing the target. This method could be used to detect cocaine down to 0.67 uM.
The mechanism of Exo III-aided cyclic amplification method for the detection of cocaine using linear molecular beacons as signal probes. Reprinted from [48] with permission
The amplification detection of proteins
Similar to the detection of small molecules, nucleic acid tool enzymes-aided signal amplification strategies are also used in protein sensing [153–155]. For example, Xing et al. developed a novel, simple, and homogeneous approach for protein detection based on nicking enzyme aided fluorescence signal amplification [153]. They extended an aptamer to form a hairpin probe (Fig. 11).
Aptamer and NEase-aided fluorescence signal-amplification detection of protein. Reprinted from [153] with permission
The binding of the aptamer to thrombin opened the hairpin, which enabled hybridization with a signaling sequence that is dual labeled with a fluorophore and a quencher. This initiated cyclic cleavage of the signaling sequence, which generated an amplified fluorescence signal. A detection limit of 100 pM was obtained. More recently, this method was further used in detecting membrane protein on single living cells in microfluidic droplets [156].
Willner et al. combined the split aptamer subunits with Exo III-aided target recycling for the analysis of proteins [157, 158]. In their system, one of the aptamer subunits was modified with a fluorophore at the 5′ terminus and a quencher at the 3′ terminus, respectively. In the absence of target protein, the fluorophore was in close proximity to the quencher, resulting in a low fluorescence signal. When the target protein was present, it induced the aptamer subunits to self-assemble into a structure with recessed 3′ terminus, resulting in cyclic enzymatic amplification and fluorescence enhancement. Similarly, by converting the protein detection to nucleic acid detection, our group developed a simpler fluorescence method for protein detection based on enzyme-aided strand displacement amplification [159]. This method was dependent on the simultaneous and proximal recognition of target-protein by pairs of proximity probes.
The amplification detection of cells
Many high-affinity aptamers that specifically bind to different target cancer cells have also been created using cell-SELEX in recent years [146]. The aptamers selected are also used for highly selective detection of cancer cells because of their good selectivity [160–162]. So, many novel aptasensors for cancer cells have been developed by nucleic acid tool enzymes-aided signal amplification [163–167].
For example, Kong et al. [163] developed a simple, highly sensitive colorimetric method for visually detecting rare cancer cells based on cell-triggered cyclic enzymatic signal amplification (Fig. 12). In the absence of target cells, the hairpin aptamer probes (HAPs) and linker DNAs stably coexist in solution. Then, the linker DNA could assemble DNA-AuNPs, producing a purple solution. In contrast, when the HAP was incubated with target cancer cells, the specific binding of HAPs to the target cells triggers a conformational switch that results a nicking endonuclease-strand scission cycle. In the subsequent step, the cleaved linker DNA could no longer assemble two DNA-AuNPs and the solution was red. This method could detect target cells with high sensitivity and high selectivity. The linear response for target CCRF-CEM cells in a concentration range from 102 to 104 cells was obtained with a detection limit of 40 cells, which is approximately 20 times lower than the normal AuNP-based methods without amplification.
Schematic illustration of a highly sensitive colorimetric method for the detection of rare cancer cells based on cell-triggered cyclic enzymatic signal amplification. Reprinted from [163] with permission
Moreover, Ding et al. [164] constructed a cascade signal amplification strategy for detection of cancer cells by combining the RCA technique with oligonucleotide functionalized nanoparticles, and anodic stripping voltammetric detection. Based on DNA-RCA-directed enzyme-catalyzed polymerization, Zheng et al. demonstrated an ultrasensitive electrochemical sensor for cancer cells and successfully applied it for the electrochemical cytosensing of MCF-7 cells [167].
Conclusions and perspective
The need for ultrasensitive bioassays and the trend towards fast assays has made the nucleic acid tool enzyme one of the most useful tools for manipulating nucleic acids and for signal amplification. A wide variety of nucleic acid tool enzymes with different catalytic properties have been introduced into biosensing for signal amplification because of their highly efficient catalytic effects. Because the substrates of nucleic acid tool enzymes are nucleic acids, the nucleic acid tool enzymes-aided signal amplification strategies are mainly used to detect nucleic acid, as well as biorelated variations of nucleic acids. On the other hand, based on different configurations of aptamers bound to targets, nucleic acid tool enzymes-aided signal amplification strategies have also been used for amplified detection of non-nucleic acid targets such as ions, small molecules, proteins, and cells.
In addition, some biosensors incorporating multiple tool enzymes are developed for cascade signal amplification and achieving higher sensitivity. For example, exonucleases and endonuclease can be used to regenerate primers for RCA reactions [116]; endonuclease can produce blunt 3′ termini for exonuclease digestion [168]; nicking endonuclease can be used to generate primers for polymerization cycle [169]; RCA reactions can generate prolonged ssDNA for nicking endonuclease-based amplification [39]. In order to further improve sensor performance, the combination of enzymatic and other non-enzymatic amplification approaches is becoming popular. For instance, Li et al. report a dual-amplification strategy for highly sensitive detection of DNA based on the dumbbell-shaped DNA probe with a magnetic bead at 5′ terminus and a fluorescein-encapsulated liposome at 3′ terminus [51].
All these nucleic acid tool enzymes-aided signal amplification strategies provide an effective tool for quantitative analysis trace amounts of analytes and supply valuable information for biochemical research, clinical diagnostics, and environmental monitoring. There are still many challenges in the development of this field. (1) With amplification, the detection time of some assays may be prolonged, which is unpopular in fast detection. So, more advanced, efficient, and stable nucleic acid tool enzymes urgently need to be explored for potential applications. (2) Their performance in practical samples is unsatisfactory (for example, cell detection), and commercialization of these nucleic acid tool enzymes-aided biosensors is difficult. Therefore, the reaction conditions need to be strictly controlled and the pretreatment of complex samples should be progressed. (3) Up to now, the number of aptamers with specific targets is limited. Only a few non-nucleic acid targets could be detected using such strategies. In order to extend the potential utility of nucleic acid tool enzymes for non-nucleic acid targets, more converting mechanisms are needed to transfer the non-nucleic acid targets detection to nucleic acid detection. With the persistent development of nucleic acid tool enzymes, we envision that the utilization of nucleic acid tool enzymes will bring higher sensitivity, reagent-less biosensing, and point-of-care diagnosis.
References
Berdis AJ. Mechanisms of DNA polymerases. Chem Rev. 2009;109:2862–79.
Barany F. Genetic disease detection and DNA amplification using cloned thermostable ligase. Proc Natl Acad Sci U S A. 1991;88:189–93.
Rhyu MS. Telomeres, telomerase, and immortality. J Natl Cancer Inst. 1995;87:884–94.
Roberts RJ, Murray K. Restriction endonuclease. Crit Rev Biochem Mol. 1976;4:123–64.
Zheleznaya LA, Kachalova GS, Artyukh RI, Yunusova AK, Perevyazova TA, Matvienko NI. Nicking endonucleases. Biochemistry. 2009;74:1457–66.
Shagin DA, Rebrikov DV, Kozhemyako VB, Altshuler IM, Shcheglov AS, Zhulidov PA, et al. A novel method for SNP detection using a new duplex-specific nuclease from crab hepatopancreas. Genome Res. 2002;12:1935–42.
Lee JE, Frank BC, Cooper TA. RNase H-mediated degradation of toxic RNA in myotonic dystrophy type 1. Proc Natl Acad Sci U S A. 2012;109:4221–6.
Sayers JR, Schmidt W, Eckstein F. 5′-3′ Exonucleases in phosphorothioate-based oligonucleotide-directed mutagenesis. Nucleic Acids Res. 1988;16:791–802.
Shevelev IV, Hübscher U. The 3′-5′ Exonucleases. Nat Rev Mol Cell Biol. 2002;3:364–76.
Zhang H, Wang S, Fang G. Applications and recent developments of multi-analyte simultaneous analysis by enzyme-linked immunosorbent assays. J Immunol Methods. 2011;368:1–23.
Haab BB. Applications of antibody array platforms. Trends Biotechnol. 2006;17:415–21.
Niemeyer CM, Adler MR. Immuno-PCR: high sensitivity detection of proteins by nucleic acid amplification. Trends Biotechnol. 2005;23:208–16.
Chiang CK, Chen WT, Chang HT. Nanoparticle-based mass spectrometry for the analysis of biomolecules. Chem Soc Rev. 2011;40:1269–81.
Lei JP, Ju HX. Signal amplification using functional nanomaterials for biosensing. Chem Soc Rev. 2012;41:2122–34.
Taniguchi K, Kajiyama T, Kambara H. Quantitative analysis of gene expression in a single cell by qPCR. Nat Methods. 2009;6:503–6.
Miao P, Tang Y, Wang B, Yin J, Ning L. Signal amplification by enzymatic tools for nucleic acids. TrAC Trends Anal Chem. 2015;67:1–15.
Gerasimova YV, Kolpashchikov DM. Enzyme-assisted target recycling (EATR) for nucleic acid detection. Chem Soc Rev. 2014;43:6405–38.
Willner I, Shlyahovsky B, Zayats M, Willner B. DNAzymes for sensing, nanobiotechnology and logic gate applications. Chem Soc Rev. 2008;37:1153–65.
Scharf P, Müller J. Nucleic acids with metal-mediated base pairs and their applications. ChemPlusChem. 2013;78:20–34.
Hamula CLA, Guthrie JW, Zhang HQ, Li XF, Le CX. Selection and analytical applications of aptamers. TrAC Trends Anal Chem. 2006;25:681–91.
Cho EJ, Lee JW, Ellington AD. Applications of aptamers as sensors. Annu Rev Anal Chem. 2009;2:241–64.
Lubin AA, Plaxco KW. Folding-based electrochemical biosensors: the case for responsive nucleic acid architectures. Acc Chem Res. 2010;43:496–505.
Li D, Song SP, Fan CH. Target-responsive structural switching for nucleic acid-based sensors. Acc Chem Res. 2010;43:631–41.
Wang F, Elbaz J, Orbach R, Magen N, Willner I. Amplified analysis of DNA by the autonomous assembly of polymers consisting of DNAzyme wires. J Am Chem Soc. 2011;133:17149–51.
Andras SC, Power JB, Cocking EC, Davey MR. Strategies for signal amplification in nucleic acid detection. Mol Biotechnol. 2001;19:29–44.
Lie YS, Petropoulos CJ. Advances in quantitative PCR technology: 5′ nuclease assays. Curr Opin Biotechnol. 1998;9:43–8.
Wassenegger M. Advantages and disadvantages of using PCR techniques to characterize transgenic plants. Mol Biotechnol. 2001;17:73–82.
Saghatelian A, Guckian KM, Thayer DA, Thayer DA, Ghadiri MR. DNA detection and signal amplification via an engineered allosteric enzyme. J Am Chem Soc. 2003;125:344–5.
Liu J, Lu Y. Accelerated color change of gold nanoparticles assembled by DNAzymes for simple and fast colorimetric Pb2+ detection. J Am Chem Soc. 2004;126:12298–305.
Guo X, Liu P, Yang X, Wang K, Wang Q, Guo Q, et al. A multiple amplification strategy for nucleic acid detection based on host-guest interaction between the β-cyclodextrin polymer and pyrene. Analyst. 2015;140:2016–22.
Wu L, Xiong E, Zhang X, Zhang X, Chen J. Nanomaterials as signal amplification elements in DNA-based electrochemical sensing. Nano Today. 2014;9:197–211.
Wang J, Liu G, Jan MR. Ultrasensitive electrical biosensing of proteins and DNA: carbon-nanotube derived amplification of the recognition and transduction events. J Am Chem Soc. 2004;126:3010–1.
Wang J. Carbon-nanotube based electrochemical biosensors: a review. Electroanalysis. 2005;17:7–14.
Tian Y, Mao C. DNAzyme amplification of molecular beacon signal. Talanta. 2005;67:532–7.
Weizmann Y, Patolsky F, Katz E, Willner I. Amplified DNA sensing and immunosensing by the rotation of functional magnetic particles. J Am Chem Soc. 2003;125:3452–4.
Lu J, Paulsen IT, Jin D. Application of exonuclease III-aided target recycling in flow cytometry: DNA detection sensitivity enhanced by orders of magnitude. Anal Chem. 2013;85:8240–5.
Xu W, Xue X, Li T, Zeng H, Liu X. Ultrasensitive and selective colorimetric DNA detection by nicking endonuclease assisted nanoparticle amplification. Angew Chem Int Ed. 2009;48:6849–52.
Bi S, Li L, Cui Y. Exonuclease-assisted cascaded recycling amplification for label-free detection of DNA. Chem Commun. 2012;48:1018–20.
Xu W, Xie X, Li D, Yang Z, Li T, Liu X. Ultrasensitive colorimetric DNA detection using a combination of rolling circle amplification and nicking endonuclease-assisted nanoparticle amplification (NEANA). Small. 2012;8:1846–50.
Shen W, Deng H, Gao Z. Gold nanoparticle-enabled real-time ligation chain reaction for ultrasensitive detection of DNA. J Am Chem Soc. 2012;134:14678–81.
Xing Y, Wang P, Zang Y, Ge Y, Jin Q, Zhao J, et al. A colorimetric method for H1N1 DNA detection using rolling circle amplification. Analyst. 2013;138:3457–62.
Li JJ, Chu Y, Lee BYH, Xie XS. Enzymatic signal amplification of molecular beacons for sensitive DNA detection. Nucleic Acids Res. 2008;36:e36.
Zou B, Ma Y, Wu H, Zhou G. Ultrasensitive DNA detection by cascade enzymatic signal amplification based on Afu flap endonuclease coupled with nicking endonuclease. Angew Chem Int Ed. 2011;50:7395–8.
Gao F, Lei J, Ju H. Assistant DNA recycling with nicking endonuclease and molecular beacon for signal amplification using a target-complementary arched structure. Chem Commun. 2013;49:4006–8.
Wang HB, Ou LJ, Huang KJ, Wen XG, Wang LL, Liu YM. A sensitive biosensing strategy for DNA detection based on graphene oxide and T7 exonuclease assisted target recycling amplification. Can J Chem. 2013;91:1266–71.
Liu L, Lei J, Gao F, Ju H. A DNA machine for sensitive and homogeneous DNA detection via lambda exonuclease assisted amplification. Talanta. 2013;115:819–22.
Zuo X, Xia F, Xiao Y, Plaxco KW. Sensitive and selective amplified fluorescence DNA detection based on exonuclease III-aided target recycling. J Am Chem Soc. 2010;132:1816–8.
Yang CJ, Liang C, Huang J, Ling Y, Lin X, Wang C, et al. Linear molecular beacons for highly sensitive bioanalysis based on cyclic Exo III enzymatic amplification. Biosens Bioelectron. 2011;27:119–24.
Zhang M, Guan YM, Ye BC. Ultrasensitive fluorescence polarization DNA detection by target assisted exonuclease III-catalyzed signal amplification. Chem Commun. 2011;47:3478–80.
Cai Z, Chen Y, Lin C, Wu Y, Yang CJ, Wang Y, et al. A dual-signal amplification method for the dna detection based on exonuclease III. Biosens Bioelectron. 2014;61:370–3.
Zhou F, Li B. Exonuclease III-assisted target recycling amplification coupled with liposome-assisted amplification: one-step and dual-amplification strategy for highly sensitive fluorescence detection of DNA. Anal Chem. 2015;87:7156–62.
Wang Q, Yang L, Yang X, Wang K, He L, Zhu J, et al. An electrochemical DNA biosensor based on the “Y” junction structure and restriction endonuclease-aided target recycling strategy. Chem Commun. 2012;48:2982–4.
Ji H, Yan F, Lei J, Ju H. Ultrasensitive electrochemical detection of nucleic acids by template enhanced hybridization followed with rolling circle amplification. Anal Chem. 2012;84:7166–71.
Liu S, Wang C, Zhang C, Wang Y, Tang B. Label-free and ultrasensitive electrochemical detection of nucleic acids based on autocatalytic and exonuclease III-assisted target recycling strategy. Anal Chem. 2013;85:2282–8.
Liu S, Lin Y, Wang L, Liu T, Cheng C, Wei W, et al. Exonuclease III-aided autocatalytic DNA biosensing platform for immobilization-free and ultrasensitive electrochemical detection of nucleic acid and protein. Anal Chem. 2014;86:4008–15.
Tao C, Yan Y, Xiang H, Zhu D, Cheng W, Ju H, et al. A new mode for highly sensitive and specific detection of DNA based on exonuclease III-assisted target recycling amplification and mismatched catalytic hairpin assembly. Chem Commun. 2015;51:4220–2.
Xiong E, Zhang X, Liu Y, Zhou J, Yu P, Li X, et al. Ultrasensitive electrochemical detection of nucleic acids based on the dual-signaling electrochemical ratiometric method and exonuclease III-assisted target recycling amplification strategy. Anal Chem. 2015;87:7291–6.
Xia F, Zuo X, Yang R, Xiao Y, Kang D, Vallée-Bélisle A, et al. Colorimetric detection of DNA, small molecules, proteins, and ions using unmodified gold nanoparticles and conjugated polyelectrolytes. Proc Natl Acad Sci U S A. 2010;107:10837–41.
Lodeiro C, Capelo JL, Mejuto JC, Oliveira E, Santos HM, Pedras B, et al. Light and colour as analytical detection tools: a journey into the periodic table using polyamines to bio-inspired systems as chemosensors. Chem Soc Rev. 2010;39:2948–76.
Tansil NC, Gao Z. Nanoparticles in biomolecular detection. Nano Today. 2006;1:28–37.
Rosi NL, Mirkin CA. Nanostructures in biodiagnostics. Chem Rev. 2005;105:1547–62.
Link S, El-Sayed MA. Spectral properties and relaxation dynamics of surface plasmon electronic oscillations in gold and silver nanodots and nanorods. J Phys Chem B. 1999;103:8410–26.
Yang Z, Sismour AM, Benner SA. Nucleoside alpha-thiotriphosphates, polymerases, and the exonuclease III analysis of oligonucleotides containing phosphorothioate linkages. Nucleic Acids Res. 2007;35:3118–27.
Ronkainen NJ, Halsall HB, Heineman WR. Electrochemical biosensors. Chem Soc Rev. 2010;39:1747–63.
Chang BY, Park SM. Electrochemical impedance spectroscopy. Annu Rev Anal Chem. 2010;3:207–29.
Wu D, Yin BC, Ye BC. A label-free electrochemical DNA sensor based on exonuclease III-aided target recycling strategy for sequence-specific detection of femtomolar DNA. Biosens Bioelectron. 2011;28:232–8.
Yu P, Zhou J, Wu L, Xiong E, Zhang X, Chen J. A ratiometric electrochemical aptasensor for sensitive detection of protein based on aptamer-target-aptamer sandwich structure. J Electroanal Chem. 2014;732:61–5.
Yan D, Byung Joon L, Bingling L, Yu Sherry J, Sessler JL, Ellington AD. Reagentless ratiometric electrochemical DNA sensors with improved robustness and reproducibility. Anal Chem. 2014;86:8010–6.
He L, Hannon GJ. MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet. 2004;5:22–531.
Sawyers CL. The cancer biomarker problem. Nature. 2008;452:548–52.
Tricoli JV, Jacobson JW. MicroRNA: potential for cancer detection, diagnosis, and prognosis. Cancer Res. 2007;67:4553–5.
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A. 2008;105:10513–8.
Cissell KA, Shrestha S, Deo SK. MicroRNA detection: challenges for the analytical chemist. Anal Chem. 2007;79:4754–61.
Válóczi A, Hornyik C, Varga N, Burgyán J, Kauppinen S, Havelda Z. Sensitive and specific detection of microRNAs by northern blot analysis using LNA-modified oligonucleotide probes. Nucleic Acids Res. 2004;32:e175.
Lee I, Ajay SS, Chen H, Maruyama A, Wang N, McInnis MG, et al. Discriminating single-base difference miRNA expressions using microarray Probe Design Guru (ProDeG). Nucleic Acids Res. 2008;36:e27.
Fang S, Lee HJ, Wark AW, Corn RM. Attomole microarray detection of microRNAs by nanoparticle-amplified SPR imaging measurements of surface polyadenylation reactions. J Am Chem Soc. 2006;128:14044–6.
Qiu X, Zhang H, Yu H, Jiang T, Luo Y. Duplex-specific nuclease-mediated bioanalysis. Trends Biotechnol. 2015;33:180–8.
Xu F, Shi H, He X, Wang K, He D, Guo Q, et al. Concatemeric dsDNA-templated copper nanoparticles strategy with improved sensitivity and stability based on rolling circle replication and its application in microRNA detection. Anal Chem. 2014;86:6976–82.
Yin BC, Liu YQ, Ye BC. Sensitive detection of microRNA in complex biological samples via enzymatic signal amplification using DNA polymerase coupled with nicking endonuclease. Anal Chem. 2013;85:11487–93.
Ge J, Zhang LL, Liu SJ, Yu RQ, Chu X. A highly sensitive target-primed rolling circle amplification (TPRCA) method for fluorescent in situ hybridization detection of microRNA in tumor cells. Anal Chem. 2014;86:1808–15.
Duan R, Zuo X, Wang S, Quan X, Chen D, Chen Z, et al. Quadratic isothermal amplification for the detection of microRNA. Nat Protoc. 2014;9:597–607.
Guo X, Yang X, Liu P, Wang K, Wang Q, Guo Q, et al. Multiple amplification detection of microRNA based on the host–guest interaction between β-cyclodextrin polymer and pyrene. Analyst. 2015;140:4291–7.
Yin BC, Liu YQ, Ye BC. One-step multiplexed fluorescence detection of microRNAs based on duplex-specific nuclease signal amplification. J Am Chem Soc. 2012;134:5064–7.
Lin X, Zhang C, Huang Y, Zhu Z, Chen X, Yang C. Backbone-modified molecular beacons for highly sensitive and selective detection of microRNAs based on duplex specific nuclease signal amplification. Chem Commun. 2013;49:7243–5.
Cui L, Zhu Z, Lin N, Zhang H, Guan Z, Yang C. A T7 exonuclease-assisted cyclic enzymatic amplification method coupled with rolling circle amplification: a dual-amplification strategy for sensitive and selective microRNA detection. Chem Commun. 2014;13:1576–8.
Xie Y, Lin X, Huang Y, Pan R, Zhu Z, Zhou L, et al. Highly sensitive and selective detection of miRNA: DNase I-assisted target recycling using DNA probes protected by polydopamine nanospheres. Chem Commun. 2015;51:2156–8.
Ching ADA, Caldwell KS, Jung M, Dolan M, Smith OS, Tingey S, et al. SNP frequency, haplotype structure, and linkage disequilibrium in elite maize inbred line. BMC Genet. 2002;3:19–33.
Mehta AM, Jordanova ES, Corver WE, van Wezel T, Uh HW, Kenter GG, et al. Single nucleotide polymorphisms in antigen processing machinery component ERAP1 significantly associate with clinical outcome in cervical carcinoma. Genes Chromosom Cancer. 2009;48:410–8.
Nam RK, Zhang WW, Trachtenberg J, Seth A, Klotz LH, Stanimirovic A, et al. Utility of incorporating genetic variants for the early detection of prostate cancer. Clin Cancer Res. 2009;15:1787–93.
Lichtenstein A, Willner I. Detection of single-base DNA mutations by enzyme-amplified electronic transduction. Nat Biotechnol. 2001;19:253–7.
Wei W, Ni Q, Pu Y, Yin L, Liu S. Electrochemical biosensor for DNA damage detection based on exonuclease III digestions. J Electroanal Chem. 2014;714:25–9.
Wu T, Xiao X, Zhang Z, Zhao M. Enzyme-mediated single-nucleotide variation detection at room temperature with high discrimination factor. Chem Sci. 2015;6:1206–11.
Zou Z, Qing Z, He X, Wang K, He D, Shi H, et al. Ligation-rolling circle amplification combined with γ-cyclodextrin mediated stemless molecular beacon for sensitive and specific genotyping of single-nucleotide polymorphism. Talanta. 2014;125:306–12.
Liu M, Yuan M, Lou X, Mao H, Zheng D, Zou R, et al. Label-free optical detection of single-base mismatches by the combination of nuclease and gold nanoparticles. Biosens Bioelectron. 2011;26:4294–300.
Till BJ, Burtner C, Comai L, Henikoff S. Mismatch cleavage by single-strand specific nucleases. Nucleic Acids Res. 2004;32:2632–41.
Wu S, Liang P, Yu H, Xu X, Liu Y, Lou X, et al. Amplified single base-pair mismatch detection via aggregation of exonuclease-sheared gold nanoparticles. Anal Chem. 2014;86:3461–7.
Reik W, Walter J. Genomic imprinting: parental influence on the genome. Nat Rev Genet. 2001;2:21–32.
Okano M, Bell DW, Haber DA, Li E. DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell. 1999;99:247–57.
Palmer BR, Marinus MG. The DAM and DCM strains of Escherichia coli—a review. Gene. 1994;143:1–12.
Robertson KD, Wolffe AP. DNA methylation in health and disease. Nat Rev Genet. 2000;1:11–9.
Laird PW, Jaenisch R. The role of DNA methylation in cancer genetics and epigenetics. Annu Rev Genet. 1996;30:441–64.
McCabe MT, Low JA, Daignault S, Imperiale MJ, Wojno KJ, Day ML. Inhibition of DNA methyltransferase activity prevents tumorigenesis in a mouse model of prostate cancer. Cancer Res. 2006;66:385–92.
Kurita R, Niwa O. DNA methylation analysis triggered by bulge specific immuno-recognition. Anal Chem. 2012;84:7533–8.
Ma L, Su M, Li T, Wang Z. Microarray-based resonance light scattering assay for detecting DNA methylation and human DNA methyltransferase simultaneously with high sensitivity. Analyst. 2014;139:3537–40.
Ehrich M, Zoll S, Sur S, Van Den Boom D. A new method for accurate assessment of DNA quality after bisulfite treatment. Nucleic Acids Res. 2007;35:e29.
Wang Y, Wee EJH, Trau M. Highly sensitive DNA methylation analysis at CpG resolution by surface-enhanced raman scattering via ligase chain reaction (LCR). Chem Commun. 2015;51:10953–6.
Hong T, Wang T, He Z, Ma J, Xiao H, Huang J, et al. Qualitative and quantitative detection of methylation at CpG sites using the fluorescein-dGTP incorporated asymmetric PCR assay strategy. Chem Commun. 2014;50:6653–5.
Gao C, Li H, Liu Y, Wei W, Zhang Y, Liu S. Label-free fluorescence detection of DNA methylation and methyltransferase activity based on restriction endonuclease Hpa II and exonuclease III. Analyst. 2014;139:6387–92.
Zhu G, Yang K, Zhang C. Sensitive detection of methylated DNA using the short linear quencher–fluorophore probe and two-stage isothermal amplification assay. Biosens Bioelectron. 2013;49:170–5.
Su F, Wang L, Sun Y, Liu C, Duan X, Li Z. Highly sensitive detection of CpG methylation in genomic DNA by AuNP-based colorimetric assay with ligase chain reaction. Chem Commun. 2015;51:3371–4.
Li W, Wu P, Zhang H, Cai C. Signal amplification of graphene oxide combining with restriction endonuclease for site-specific determination of DNA methylation and assay of methyltransferase activity. Anal Chem. 2012;84:7583–90.
Zhang H, Li M, Fan M, Gu J, Wu P, Cai C. Electrochemiluminescence signal amplification combined with a conformation-switched hairpin DNA probe for determining the methylation level and position in the Hsp53 tumor suppressor gene. Chem Commun. 2014;50:2932–4.
Mutze K, Langer R, Schumacher F, Becker K, Ott K, Novotny A, et al. DNA methyltransferase 1 as a predictive biomarker and potential therapeutic target for chemotherapy in gastric cancer. Eur J Cancer. 2011;47:1817–25.
Zhu C, Wen Y, Peng H, Long Y, He Y, Huang Q, et al. A methylation-stimulated DNA machine: an autonomous isothermal route to methyltransferase activity and inhibition analysis. Anal Bioanal Chem. 2011;399:3459–64.
Liu P, Yang XH, Wang Q, Huang J, Liu JB, Zhu Y, et al. Sensitive detection of DNA methyltransferase activity based on rolling circle amplification technology. Chin Chem Lett. 2014;25:1047–51.
Zeng Y, Hu J, Long Y, Zhang CY. Sensitive detection of DNA methyltransferase using hairpin probe-based primer generation rolling circle amplification-induced chemiluminescence. Anal Chem. 2013;85:6143–50.
Xue Q, Lv Y, Xu S, Zhang Y, Wang L, Li R, et al. Highly sensitive fluorescence assay of DNA methyltransferase activity by methylation-sensitive cleavage-based primer generation exponential isothermal amplification-induced G-quadruplex formation. Biosens Bioelectron. 2015;66:547–53.
Xue Q, Wang L, Jiang W. Label-free molecular beacon-based quadratic isothermal exponential amplification: a simple and sensitive one-pot method to detect DNA methyltransferase activity. Chem Commun. 2015;51:13538–41.
He X, Su J, Wang Y, Wang K, Ni X, Chen Z. A sensitive signal-on electrochemical assay for MTase activity using AuNPs amplification. Biosens Bioelectron. 2011;28:298–303.
Su J, He X, Wang Y, Wang K, Chen Z, Yan G. A sensitive signal-on assay for MTase activity based on methylation-responsive hairpin-capture DNA probe. Biosens Bioelectron. 2012;36:123–8.
Wang Y, He X, Wang K, Su J, Chen Z, Yan G, et al. A label-free electrochemical assay for methyltransferase activity detection based on the controllable assembly of single wall carbon nanotubes. Biosens Bioelectron. 2013;41:238–43.
Zhang Y, Xu W, Zeng Y, Zhang C. Sensitive detection of DNA methyltransferase activity by transcription-mediated duplex-specific nuclease-assisted cyclic signal amplification. Chem Commun. 2015;51:13968–71.
Watson JD, Crick FH. Letters to nature: molecular structure of nucleic acid. Nature. 1953;171:738.
Ding W, Xu M, Zhu H, Liang H. Mechanism of the hairpin folding transformation of thymine-cytosine-rich oligonucleotides induced by Hg (II) and Ag (I) ions. Eur Phys J E. 2013;36:1–8.
Fortino M, Marino T, Russo N. Theoretical study of silver-ion-mediated base pairs: the case of C-Ag-C and C-Ag-A systems. J Phys Chem A. 2014;119:5153–7.
He X, Qing Z, Wang K, Zou Z, Shi H, Huang J. Engineering a unimolecular multifunctional DNA probe for analysis of Hg2+ and Ag+. Anal Methods. 2012;4:345–7.
Tortolini C, Bollella P, Antonelli ML, Antiochia R, Mazzei F, Favero G. DNA-based biosensors for Hg2+ determination by polythymine-methylene blue modified electrodes. Biosens Bioelectron. 2015;67:524–31.
Xuan F, Luo X, Hsing IM. Conformation-dependent exonuclease III activity mediated by metal ions reshuffling on thymine-rich DNA duplexes for an ultrasensitive electrochemical method for Hg2+ detection. Anal Chem. 2013;85:4586–93.
Chen J, Zhou S, Wen J. Disposable strip biosensor for visual detection of Hg2+ based on Hg2+-triggered toehold binding and exonuclease III-assisted signal amplification. Anal Chem. 2014;86:3108–14.
Yin J, He X, Jia X, Wang K, Xu F. Highly sensitive label-free fluorescent detection of Hg2+ ions by DNA molecular machine-based Ag nanoclusters. Analyst. 2013;138:2350–6.
Xu G, Wang G, He X, Zhu Y, Chen L, Zhang X. An ultrasensitive electrochemical method for detection of Ag+ based on cyclic amplification of exonuclease III activity on cytosine-Ag+-cytosine. Analyst. 2013;138:6900–6.
Zhou Y, Xing XJ, Pang DW, Tang HW. An exonuclease III-aided “turn-on” fluorescence assay for mercury ions based on graphene oxide and metal-mediated “molecular beacon”. RSC Adv. 2015;5:12994–9.
Zhao W, Lam JCF, Chiuman W, Brook MA, Li Y. Enzymatic cleavage of nucleic acids on gold nanoparticles: a generic platform for facile colorimetric biosensors. Small. 2008;4:810–6.
Shen L, Chen Z, Li Y, He S, Xie S, Xu X, et al. Electrochemical DNAzyme sensor for lead based on amplification of DNA-Au Bio-Bar codes. Anal Chem. 2008;80:6323–8.
Tang S, Tong P, Li H, Tang J, Zhang L. Ultrasensitive electrochemical detection of Pb2+ based on rolling circle amplification and quantum dots tagging. Biosens Bioelectron. 2013;42:608–11.
Peng Y, Li L, Yi X, Guo L. Label-free picomolar detection of Pb2+ using atypical icosahedra gold nanoparticles and rolling circle amplification. Biosens Bioelectron. 2014;59:314–20.
He JL, Zhu SL, Wu P, Li PP, Li T, Cao Z. Enzymatic cascade based fluorescent DNAzyme machines for the ultrasensitive detection of Cu (II) ions. Biosens Bioelectron. 2014;60:112–7.
Ellington AD, Szostak JW. In vitro selection of RNA molecules that bind specific ligands. Nature. 1990;346:818–22.
Tuerk C, Gold L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science. 1990;249:505–10.
Liu J, Cao Z, Lu Y. Functional nucleic acid sensors. Chem Rev. 2009;109:1948–98.
Cho EJ, Yang L, Levy M, Ellington AD. Using a deoxyribozyme ligase and rolling circle amplification to detect a non-nucleic acid analyte, ATP. J Am Chem Soc. 2005;127:2022–3.
Shi H, He X, Wang K, Wu X, Ye X, Guo Q, et al. Activatable aptamer probe for contrast-enhanced in vivo cancer imaging based on cell membrane protein-triggered conformation alteration. Proc Natl Acad Sci U S A. 2011;108:3900–5.
Lu CH, Wang F, Willner I. Amplified optical aptasensors through the endonuclease-stimulated regeneration of the analyte. Chem Sci. 2012;3:2616–22.
Li F, Zhang H, Wang Z, Newbigging AM, Reid MS, Li XF, et al. Aptamers facilitating amplified detection of biomolecules. Anal Chem. 2014;87:274–92.
Yin J, He X, Wang K, Xu F, Shangguan J, He D, et al. Label-free and turn-on aptamer strategy for cancer cells detection based on a DNA-silver nanocluster fluorescence upon recognition-induced hybridization. Anal Chem. 2013;85:12011–9.
Tan W, Donovan MJ, Jiang J. Aptamers from cell-based selection for bioanalytical applications. Chem Rev. 2013;113:2842–62.
Holm RH, Kennepohl P, Solomon EI. Structural and functional aspects of metal sites in biology. Chem Rev. 1996;96:2239–314.
Deng B, Lin Y, Wang C, Li F, Wang Z, Zhang H, et al. Aptamer binding assays for proteins: the thrombin example-a review. Anal Chim Acta. 2014;837:1–15.
Feng C, Dai S, Wang L. Optical aptasensors for quantitative detection of small biomolecules: A review. Biosens Bioelectron. 2014;59:64–74.
Huang Y, Chen J, Zhao S, Shi M, Chen ZF, Liang H. Label-free colorimetric aptasensor based on nicking enzyme assisted signal amplification and DNAzyme amplification for highly sensitive detection of protein. Anal Chem. 2013;85:4423–30.
Liu S, Wang Y, Zhang C, Lin Y, Li F. Homogeneous electrochemical aptamer-based ATP assay with signal amplification by exonuclease III assisted target recycling. Chem Commun. 2013;49:2335–7.
Huang Y, Liu X, Zhang L, Hu K, Zhao S, Fang B, et al. Nicking enzyme and graphene oxide-based dual signal amplification for ultrasensitive aptamer-based fluorescence polarization assays. Biosens Bioelectron. 2015;63:178–84.
Xue L, Zhou X, Xing D. Sensitive and homogeneous protein detection based on target-triggered aptamer hairpin switch and nicking enzyme assisted fluorescence signal amplification. Anal Chem. 2012;84:3507–13.
Gao T, Ning L, Li C, Wang H, Li G. A colorimetric method for protein assay via exonuclease III-assisted signal attenuation strategy and specific DNA–protein interaction. Anal Chim Acta. 2013;788:171–6.
Zheng AX, Wang JR, Li J, Song XR, Chen GN, Yang HH. Nicking enzyme based homogeneous aptasensors for amplification detection of protein. Chem Commun. 2011;48:374–6.
Li L, Wang Q, Feng J, Tong L, Tang B. Highly sensitive and homogeneous detection of membrane protein on a single living cell by aptamer and nicking enzyme assisted signal amplification based on microfluidic droplets. Anal Chem. 2014;86(10):5101–7.
Liu X, Freeman R, Willner I. Amplified fluorescence aptamer-based sensors using exonuclease III for the regeneration of the analyte. Chem Eur J. 2012;18:2207–11.
Freeman R, Girsh J, Fang-ju Jou A, Ho JAA, Hug T, Dernedde J, Willner I. Optical aptasensors for the analysis of the vascular endothelial growth factor (VEGF). Anal Chem. 2012;84:6192–6198.
Tan Y, Guo Q, Zhao X, Yang X, Wang K, Huang J, et al. Proximity-dependent protein detection based on enzyme-assisted fluorescence signal amplification. Biosens Bioelectron. 2014;51:255–60.
Song J, Lv F, Yang G, Liu L, Yang Q, Wang S. Aptamer-based polymerase chain reaction for ultrasensitive cell detection. Chem Commun. 2012;48:7465–7.
Bamrungsap S, Chen T, Shukoor MI, Chen Z, Sefah K, Chen Y, et al. Pattern recognition of cancer cells using aptamer-conjugated magnetic nanoparticles. ACS Nano. 2012;6:3974–81.
Herr JK, Smith JE, Medley CD, Shangguan D, Tan W. Aptamer-conjugated nanoparticles for selective collection and detection of cancer cells. Anal Chem. 2006;78:2918–24.
Zhang X, Xiao K, Cheng L, Chen H, Liu B, Zhang S, et al. Visual and highly sensitive detection of cancer cells by a colorimetric aptasensor based on cell-triggered cyclic enzymatic signal amplification. Anal Chem. 2014;86:5567–72.
Ding C, Liu H, Wang N, Wang Z. Cascade signal amplification strategy for the detection of cancer cells by rolling circle amplification and nanoparticles tagging. Chem Commun. 2012;48:5019–21.
Zhao J, Zhang L, Chen C, Jiang J, Yu R. A novel sensing platform using aptamer and RNA polymerase-based amplification for detection of cancer cells. Anal Chim Acta. 2012;745:106–11.
Li Y, Zeng Y, Ji X, Li X, Ren R. Cascade signal amplification for sensitive detection of cancer cell based on self-assembly of DNA scaffold and rolling circle amplification. Sensors Actuators B Chem. 2012;171:361–6.
Sheng Q, Cheng N, Bai W, Zheng J. Ultrasensitive electrochemical detection of breast cancer cells based on DNA-rolling-circle-amplification-directed enzyme-catalyzed polymerization. Chem Commun. 2015;51:2114–7.
Li W, Liu X, Hou T, Li H, Li F. Ultrasensitive homogeneous electrochemical strategy for DNA methyltransferase activity assay based on autonomous exonuclease III-assisted isothermal cycling signal amplification. Biosens Bioelectron. 2015;70:304–9.
Joneja A, Huang X. Linear nicking endonuclease-mediated strand-displacement DNA amplification. Anal Biochem. 2011;414:58–69.
Acknowledgments
This work was supported by the Key Project of Natural Science Foundation of China (grant no. 21175039, 21322509, 21305035, 21190044, and 21221003), Research Fund for the Doctoral Program of Higher Education of China (grant no. 20110161110016), and the project supported by Hunan Provincial Innovation Foundation for Postgraduate (grant no. CX2015B072).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Published in the topical collection featuring Young Investigators in Analytical and Bioanalytical Science with guest editors S. Daunert, A. Baeumner, S. Deo, J. Ruiz Encinar, and L. Zhang.
Rights and permissions
About this article
Cite this article
Qing, T., He, D., He, X. et al. Nucleic acid tool enzymes-aided signal amplification strategy for biochemical analysis: status and challenges. Anal Bioanal Chem 408, 2793–2811 (2016). https://doi.org/10.1007/s00216-015-9240-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-9240-y