Abstract.
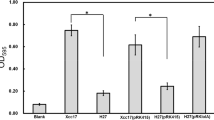
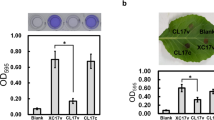
Myxococcus xanthus is a gram-negative soil bacterium that initiates a complex developmental program in response to starvation. A transposon insertion (Tn5-lac Ω109) mutant with developmental deficiencies was isolated and characterized in this study. A strain containing this insertion mutation in an otherwise wild-type background showed delayed developmental aggregation for about 12 h and sporulated at 1–2% of the wild-type level. Tn5-lac Ω109 was found to have disrupted the M. xanthus wbgB gene, which is located 2.1 kb downstream of the M. xanthus lipopolysacharide (LPS) O-antigen biosynthesis genes wzm wzt wbgA. The deduced polypeptide sequence of WbgB shares significant similarity with bacterial glycosyltransferases including M. xanthus WbgA. The wbgB::Tn5-lac Ω109 mutant was found to be defective in LPS O-antigen synthesis by immunochemical analysis. Further mutational analysis indicated that the defects of the wbgB::Tn5-lac Ω109 mutant were not the result of polar effects on downstream genes. Various motility assays demonstrated that the Tn5-lac Ω109 mutation affected both social (S) and adventurous (A) gliding motility of M. xanthus cells. The pleiotrophic effects of wbgB mutations indicate the importance of LPS O-antigen biosynthesis for various cellular functions in M. xanthus.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Yang, Z., Guo, D., Bowden, M. et al. The Myxococcus xanthus wbgB gene encodes a glycosyltransferase homologue required for lipopolysaccharide O-antigen biosynthesis. Arch Microbiol 174, 399–405 (2000). https://doi.org/10.1007/s002030000226
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002030000226