Abstract
Background and Purpose
The objective of the current study was to evaluate the acute effects of cranial radiation therapy (CNS-RT) using different radiation doses (0, 1.8, 2, 3, ≥ 20 Gy) on cognitive function with special emphasis on memory. We assessed patients with and without intracranial tumors to distinguish between direct and indirect radiation effects on brain tissue.
Materials and Methods
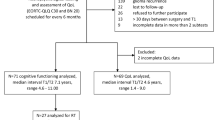
Eighty-two patients were evaluated with neuropsychological testing before and acutely after radiotherapy (RT). Sixty-four patients received RT to the brain (55 with, 9 without intracranial tumor). Eighteen patients treated with RT to the breast served as controls.
Results
Patients with intracranial tumor demonstrated attention (19–38th percentile) and verbal memory scores (34–46th percentile) below the population average at baseline. The average Verbal Memory score was significantly different between patients with intracranial tumor and controls both at baseline (38th vs. 58th percentile) and after irradiation (27th vs. 52th percentile). Patients with preexisting peritumoral edema performed worse than patients without edema and controls. Radiation dose-related deficits were seen for working memory performance in patients with intracranial tumor.
Conclusion
Our data indicate no measurable impairment of cognitive functioning acutely after prophylactic cranial irradiation. Patients with intracranial tumor show a deterioration of almost all memory functions with a dose-dependent impairment in working memory. Patients with preexisting peritumoral brain edema show the strongest deterioration.
Zusammenfassung
Hintergrund und Ziel
Die vorliegende Arbeit untersucht Akuteffekte der kraniellen Strahlentherapie (ZNS-RT) nach unterschiedlichen Bestrahlungsdosen (0, 1.8, 2, 3, ≥ 20 Gy) auf die kognitive Funktion unter besonderer Berücksichtigung des verbalen Gedächtnisses. Wir haben Patienten mit und ohne Hirntumor untersucht, um zwischen direkten und indirekten Bestrahlungseffekten auf das Hirngewebe zu unterscheiden.
Patienten und Methodik
82 Patienten wurden vor und unmittelbar nach Beginn der Radiotherapie (RT) neuropsychologisch untersucht. 64 Patienten wurden am ZNS bestrahlt (55 mit, 9 ohne Hirntumor). 18 Patientinnen, die an der Mamma bestrahlt wurden, dienten als Kontrollgruppe.
Ergebnisse
Vor RT-Beginn lagen Aufmerksamkeitsleistungen (Prozentränge von 19–38) und verbale Gedächtnisleistungen (Prozentränge von 34–46) der Patienten mit Hirntumor unterhalb des Mittelwertes für die Normalbevölkerung (Tabellen 4, 5). Die durchschnittliche verbale Gedächtnisleistung der Patienten mit Hirntumor unterschied sich vor (Prozentrang 38 vs. 58) und nach RT (Prozentrang 27 vs. 52) signifikant von der der Kontrollgruppe (Abbildung 1). Patienten mit einem peritumoralen Hirnödem vor ZNS-RT zeigen schlechtere Leistungen als Patienten ohne Hirnödem und Kontrollpatienten (Abbildung 3). Bestrahlungsdosisabhängige Effekte wurden für das Arbeitsgedächtnis bei Patienten mit ZNS-Tumor beobachtet (Abbildung 2).
Schlussfolgerung
Unsere Daten zeigen keine messbaren kognitiven Beeinträchtigungen unmittelbar nach Beginn einer prophylaktischen Ganzhirnbestrahlung. Patienten mit Hirntumor zeigen eine Verschlechterung der verbalen Gedächtnisfunktionen und eine dosisabhängige Beeinträchtigung im Arbeitsgedächtnis. Patienten mit einem peritumoralen Hirnödem vor ZNS-RT zeigen die größten Veränderungen.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Armstrong CL, Corn BW, Ruffer JE, et al. Radiotherapeutic effects on brain function: double dissociation of memory systems. Neuropsychiatry Neuropsychol Behav Neurol 2000;13:101–111.
Armstrong C, Ruffer J, Corn B, et al. Biphasic patterns of memory deficits following moderate-dose partial-brain irradiation: neuropsychologic outcome and proposed mechanisms. J Clin Oncol 1995;13:2263–2271.
Brunner R, Schaefer D, Hess K, et al. Effect of corticosteroids on short-term and long-term memory. Neurology 2005;64:335–337.
Cao Y, Tsien CI, Shen Z, et al. Use of magnetic resonance imaging to assess blood-brain/blood-glioma barrier opening during conformal radiotherapy. J Clin Oncol 2005;23:4127–4136.
DeQuervain DJF, Roozendaal B, Nitsch RM, et al. Acute cortisone administration impairs retrieval of long-term declarative memory in humans. Nat Neurosci 2000;3:313–314.
Duchstein S, Gademann G, Peters B. Early and late effects of local high dose radiotherapy of the brain on memory and attention. Strahlenther Onkol 2003;179:441–451.
Ernst-Stecken A, Jeske I, Hess A, et al. Hypofractionated stereotactic radiotherapy to the rat hippocampus. Determination of dose response and tolerance. Strahlenther Onkol 2007;183:440–446.
Fuss M, Poljanc K, Hug EB. Full Scale IQ (FSIQ) changes in children treated with whole brain and partial brain irradiation. A review and analysis. Strahlenther Onkol 2000;176:573–581.
Gregor A, Cull A, Traynor E, et al. Neuropsychometric evaluation of long-term survivors of adult brain tumours: relationship with tumour and treatment parameters. Radiother Oncol 1996;41:55–59.
Hawighorst H, Engenhart R, Knopp MV, et al. Intracranial meningeomas: time- and dose-dependent effects of irradiation on tumor microcirculation monitored by dynamic MR imaging. Magn Reson Imaging 1997;15:423–432.
Helmstädter C, Lendt M, Lux S. VLMT-Verbaler Lern- und Merkfähigkeitstest. Göttingen: Hogrefe, 2001.
Henzel M, Gross MW, Hamm K, et al. Stereotactic radiotherapy of meningiomas. Symptomatology, acute and late toxicity. Strahlenther Onkol 2006;182:382–388.
Herrmann C, Buss U, Snaith RP. HADS-D: Hospital anxiety and depression scale. Deutsche Version [German version]. Bern: Huber, 1995.
Hoffman R, Al’Absi M. The effect of acute stress on subsequent neuropsychological test performance (2003). Arch Clin Neuropsychol 2004;19:497–506.
Kim YH, Choi BI, Cho WH, et al. Dynamic contrast-enhanced MR imaging of VX2 carcinomas after X-irradiation in rabbits: comparison of gadopentetate dimeglumine and a macromolecular contrast agent. Invest Radiol 2003;38:539–549.
Kirschbaum C, Wolf OT, May M, et al. Stress- and treatment-induced elevations of cortisol levels associated with impaired declarative memory in healthy adults. Life Sci 1996;58:1475–1483.
Krueck WG, Schmiedl UP, Maravilla KR, et al. MR assessment of radiation-induced blood-brain barrier permeability changes in rat glioma model. AJNR 1994;15:625–632.
Lehrl S. Mehrfachwahl-Wortschatz-Intelligenztest MWT-B [The multiple choice vocabulary test]. 4th ed. Balingen: Spitta Verlag, 1999.
Lezak MD. Neuropsychological assessment. 3rd ed. New York, NY: Oxford University Press, 1995.
Limoli CL, Giedzinski E, Rola R, et al. Radiation response of neural precursor cells: linking cellular sensitivity to cell cycle checkpoints, apoptosis and oxidative stress. Radiat Res 2004;161:17–27.
Lupien SJ, Fiocco A, Wan N, et al. Stress hormones and human memory function across the lifespan. Psychoneuroendocrinology 2005;30:225–242.
Lupien SJ, Gaudreau S, Tchiteya MN, et al. Stress-induced declarative memory impairment in healthy elderly subjects: relationship to cortisol reactivity. J Clin Endocrinol Metab 1997;82:2070–2075.
Madsen TM, Kristjansen PEG, Bolwig TG, et al. Arrested neuronal proliferation and impaired hippocampal function following fractionated brain irradiation in the adult rat. Neuroscience 2003;119:635–642.
Majhail NS, Chander S, Mehta VS, et al. Factors influencing early complications following Gamma Knife radiosurgery. A prospective study. Stereotact Funct Neurosurg 2001;76:36–46.
Meador KJ, Taylor HS, Loring DW. Medical College of Georgia (MCG) Complex figures. Augusta: Medical College of Georgia, 1991.
Meyers CA, Smith JA, Bezjak A, et al. Neurocognitive function and progression in patients with brain metastases treated with whole-brain radiation and Motexafin Gadolinium: Results of a randomized phase III trial. J Clin Oncol 2004;22:157–165.
Mizumatsu S, Monje ML, Morhardt DR, et al. Extreme sensitivity of adult neurogenesis to low doses of X-irradiation. Cancer Res 2003;63:4021–4027.
Monje ML, Mizumatsu S, Fike JR, et al. Irradiation induces neural precursorcell dysfunction. Nat Med 2002;8:955–962.
Newcomer JW, Craft S, Hershey T, et al. Glucocorticoid-induced impairment in declarative memory performance in adult humans. J Neurosci 1994;14:2047–2053.
Nieder C, Astner ST, Grosu AL, et al. The role of postoperative radiotherapy after resection of a single brain metastasis. Combined analysis of 643 patients. Strahlenther Onkol 2007;183:576–580.
Peißner W, Kocher M, Treuer H, et al. Ionizing radiation-induced apoptosis of proliferating stem cells in the dentate gyrus of the adult rat hippocampus. Brain Res Mol Brain Res 1999;71:61–68.
Penitzka S, Steinvorth S, Sehlleier S, et al. Assessment of cognitive function after preventive and therapeutic whole brain irradiation using neuro-psychological testing. Strahlenther Onkol 2002;178:252–258.
Porter RJ, Barnett NA, Idey A, et al. Effects of hydrocortisone administration on cognitive function in the elderly. J Psychopharmacol 2002;16:65–71.
Raber J, Rola R, LeFevour A, et al. Radiation induced cognitive impairments are associated with changes in indicators of hippocampal neurogenesis. Radiat Res 2004;162:39–47.
Ris MD, Packer R, Goldwein J, et al. Intellectual outcome after reduced-dose radiation therapy plus adjuvant chemotherapy for medulloblastoma: a Children’s Cancer Group study. J Clin Oncol 2001;19:3470–3476.
Rola R, Raber J, Rizk A, et al. Radiation induced impairment of hippocampal neurogenesis is associated with cognitive deficits in young mice. Exp Neurol 2004;188:316–330.
Salander P, Karlsson T, Bergenheim T, et al. Long-term memory deficits in patients with malignant glioma. J Neurooncol 1995;25:227–238.
Selch MT, Ahn E, Laskari A, et al. Stereotactic radiotherapy for treatment of cavernous sinus meningiomas. Int J Radiat Oncol Biol Phys 2004;59:101–111.
Silasi G, Diaz-Heijtz R, Besplug J, et al. Selective brain responses to acute and chronic low-dose X-ray irradiation in males and females. Biochem Biophys Res Commun 2004;325:1223–1235.
Spiegler BJ, Bouffet E, Greenberg ML, et al. Change in neurocognitive functioning after treatment with cranial radiation in childhood. J Clin Oncol 2004;22:706–713.
Spreen O, Strauss E. A compendium of neuropsychological tests: administration, norms, and commentary. 2nd ed. New York (NY): Oxford University Press, 1998.
Steinvorth S, Welzel G, Fuss M, et al. Neuropsychological outcome after fractionated stereotactic radiotherapy (FSRT) for base of skull meningiomas: a prospective 1-year follow-up. Radiother Oncol 2003;69:177–182.
Steinvorth S, Wenz F, Wildermuth S, et al. Cognitive function in patients with cerebral arteriovenous malformations after radiosurgery: prospective long-term follow-up. Int J Radiat Oncol Biol Phys 2002;54:1430–1437.
Van Oosterhout AG, Ganzevles PG, Wilmink JT, et al. Sequelae in long-term survivors of small cell lung cancer. Int Radiat Oncol Biol Phys 1996;34:1037–1044.
Vollmann H, Wölfel S, Ohneseit P, et al. Differential expression of Egr1 and activation of microglia following irradiation in the rat brain. Strahlenther Onkol 2007;183:248–255.
Welzel G, Steinvorth S, Wenz F. Cognitive effects of chemotherapy and/or cranial irradiation in adults. Strahlenther Onkol 2005;181:141–156.
Wenz F, Steinvorth S, Lohr F, et al. Prospective evaluation of delayed central nervous system (CNS) toxicity of hyperfractionated total body irradiation (TBI). Int J Radiat Oncol Biol Phys 2000;48:1497–1501.
Wenz F, Steinvorth S, Lohr F, et al. Acute central nervous system (CNS) toxicity of total body irradiation (TBI) measured using neuropsychological testing of attention functions. Int J Radiat Oncol Biol Phys 1999;44:891–894.
Wenz F, Steinvorth S, Wildermuth S, et al. Assessment of neuropsychological changes in patients with arteriovenous malformation (AVM) after radiosurgery. Int J Radiat Oncol Biol Phys 1998;42:995–999.
Zimmermann P, Fimm B. Testbatterie zur Aufmerksamkeitsprüfung [Test for attentional performance, TAP]. Herzogenrath: PsyTest, 1995.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Welzel, G., Fleckenstein, K., Mai, S.K. et al. Acute neurocognitive impairment during cranial radiation therapy in patients with intracranial tumors. Strahlenther Onkol 184, 647–654 (2008). https://doi.org/10.1007/s00066-008-1830-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-008-1830-6