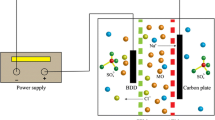
Abstract
Continuous anodic oxidation of azo dye C.I. Acid Red 18 by using PbO2 electrode in aqueous solution was studied. To reach the best conditions of the process, the influence of various operating parameters such as pH, current density, hydraulic retention time (HRT) and dye concentration on the removal rate of chemical oxygen demand (COD) and color, as indexes showing the amount of efficiency, was investigated. The findings showed that, respectively, 99.9% and 80.0% of the dye and COD were removed (at optimized conditions). Mineralization current efficiency results indicated that at the beginning of the reaction mineralization occurred quickly at a low current density, whereas at high amounts the rate of mineralization the efficiency decreased. At the optimum conditions, the majority of COD was removed only with 38.2 kWh/kg COD of energy consumption in 120 min. By controlling HO•/dye concentration ratio via the parameters adjustment, particularly HRT and current density, this system can treat Acid Red 18 well even at high concentrations. Furthermore, the voltammetry study illustrated that electroactive intermediates created during the process were mineralized at current density of 8.6mA/cm2.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J. Basiri Parsa, M. Golmirzaei and M. Abbasi, J. Ind. Eng. Chem., 20, 689 (2014).
E. Hosseini Koupaie, M. Alavi Moghaddam and S. Hashemi, J. Hazard. Mater., 195, 147 (2011).
S. S. Martínez and E. V. Uribe, Ultrason. Sonochem., 19, 174 (2012).
E. J. Ruiz, C. Arias, E. Brillas, A. Hernández-Ramírez and J. Peralta-Hernández, Chemosphere, 82, 495 (2011).
S. Asad, M. Amoozegar, A. A. Pourbabaee, M. Sarbolouki and S. Dastgheib, Bioresour. Technol., 98, 2082 (2007).
H. Lin, H. Zhang, X. Wang, L. Wang and J. Wu, Sep. Purif. Technol., 122, 533 (2014).
M. Shirzad-Siboni, S. J. Jafari, O. Giahi, I. Kim, S. M. Lee and J. K. Yang, J. Ind. Eng. Chem., 20, 1432 (2014).
A. Pandey, P. Singh and L. Iyengar, Int. Biodeter. Biodegr., 59, 73 (2007).
E. Hosseini Koupaie, M. Alavi Moghaddam and S. Hashemi, Bioresour. Technol., 127, 415 (2013).
E. Hosseini Koupaie, M. Alavi Moghaddam and S. Hashemi, Int. Biodeter. Biodegr., 71, 43 (2012).
S. Mozia, M. Tomaszewska and A. W. Morawski, Desalination, 198, 183 (2006).
S. Mozia, M. Tomaszewska and A. W. Morawski, Desalination, 185, 449 (2005).
G. Kazem, G. Azarian, D. Nematollahi, A. R. Rahmani and H. Zolghadrnasab, Res. J. Chem. Environ., 16, 98 (2012).
A. R. Rahmani, D. Nematollahi, K. Godini and G. Azarian, Sep. Purif. Technol., 107, 166 (2013).
M. Hossain, I. Mahmud, S. Parvez and H. M. Cho, Environ. Eng. Res., 18, 157 (2013).
W. C. Lin, C. H. Chen, H. Y. Tang, Y. C. Hsiao, J. R. Pan, C. C. Hu and C. Huang, Appl. Catal. B-Environ., 140, 32 (2013).
X. Florenza, A. M. S. Solano, F. Centellas, C. A. Martínez-Huitle, E. Brillas and S. Garcia-Segura, Electrochem. Acta, 142, 276 (2014).
Y. Z. Song, Dyes Pigm., 87, 39 (2010).
J. Sim, H. Seo and J. Kim, Korean J. Chem. Eng., 29, 483 (2012).
K. Godini, G. Azarian, A. R. Rahmani and H. Zolghadrnasab, J. Res. Health Sci., 13, 188 (2013).
S. Song, L. Zhan, Z. He, L. Lin, J. Tu, Z. Zhang, J. Chen and L. Xu, J. Hazard. Mater., 175, 614 (2010).
S. Song, J. Fan, Z. He, L. Zhan, Z. Liu, J. Chen and X. Xu, Electrochem. Acta, 55, 3606 (2010).
I. Sirés, E. Brillas, G. Cerisola and M. Panizza, J. Electroanal. Chem., 613, 151 (2008).
S. Curteanu, K. Godini, C. G. Piuleac, G. Azarian, A. R. Rahmani and C. Butnariu, Ind. Eng. Chem. Res., 53, 4902 (2014).
H. S. Awad and N. A. Galwa, Chemosphere, 61, 1327 (2005).
A. R. Rahmani, D. Nematollahi, G. Azarian, K. Godini and Z. Berizi, Korean J. Chem. Eng., 32, 1570 (2015).
W. Horwitz (Ed.), Standard Methods for the Examination of Water and Wastewater, 20th Ed., APHA, Washington, D.C. (2005).
H. Xu, A. P. Li, Q. Qi, W. Jiang and Y. M. Sun, Korean J. Chem. Eng., 29, 1178 (2012).
Y. Cong, Z. Wu and Y. Li, Korean J. Chem. Eng., 25, 727 (2008).
W. Mook, M. Aroua, M. Chakrabarti, C. Low, P. V. Aravind and N. Brandon, Electrochim. Acta, 94, 327 (2013).
J. Gao, H. Zhao, F. Cao, J. Zhang and C. Cao, Electrochim. Acta, 54, 2595 (2009).
A. R. Rahmani, K. Godini, D. Nematollahi and G. Azarian, Desalin. Water Treat., 56, 2234 (2015).
M. Panizza and C. A. Martinez-Huitle, Chemosphere, 90, 1455 (2013).
H. An, H. Cui, W. Zhang, J. Zhai, Y. Qian, X. Xie and Q. Li, Chem. Eng. J., 209, 86 (2012).
B. Krishnakumar and M. Swaminathan, Spectrochim. Acta A, 81, 739 (2011).
D. Nematollahi and H. Shayani-Jam, J. Org. Chem., 73, 3428 (2008).
D. Nematollahi, A. Amani and E. Tammari, J. Org. Chem., 72, 3646 (2007).
D. Nematollahi, A. Afkhami, E. Tammari, T. Shariatmanesh, M. Hesari and M. Shojaeifard, Chem. Commun., 2, 162 (2007).
D. Nematollahi, M. S. Workentin and E. Tammari, Chem. Commun., 15, 1631 (2006).
L. Yue, K. Wang, J. Guo, J. Yang, X. Luo, J. Lian and L. Wang, J. Ind. Eng. Chem., 20, 725 (2014).
Z. He, C. Huang, Q. Wang, Z. Jiang, J. Chen and S. Song, Int. J. Electrochem. Sci., 6, 4341 (2011).
H. Beiginejad, D. Nematollahi, M. Bayat, F. Varmaghani and A. Nazaripour, J. Electrochem. Soc., 160, H693 (2013).
H. Beiginejad, D. Nematollahi, F. Varmaghani, M. Bayat and H. Salehzadeh, J. Electrochem. Soc., 160, G3001 (2013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rahmani, A.R., Godini, K., Nematollahi, D. et al. Degradation of azo dye C.I. Acid Red 18 using an eco-friendly and continuous electrochemical process. Korean J. Chem. Eng. 33, 532–538 (2016). https://doi.org/10.1007/s11814-015-0175-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-015-0175-y