Abstract
In arid and semi-arid regions, a hydrological regime characterized by an annual cycle of drought and rainy seasons changes the volume and retention time of reservoirs. Such changes affect the limnological characteristics and lead to changes on phytoplankton community. Phytoplankton seasonal succession was studied in a Brazilian eutrophic semi-arid water supply reservoir (Cruzeta). In this study, the changes in the biomass and species composition of phytoplankton during two annual hydrological cycles were analyzed, and the driving factors were evaluated. The composition of phytoplankton alternated between filamentous cyanobacteria, such as Cylindrospermopsis raciborskii, under conditions of mixing, and colonial species, such as Microcystis panniformis and Sphaerocavum brasiliensis, under conditions of high water column stability. The lower water level during a severe drought favored adaptive phytoplankton species with low requirement for resources, such as diatoms and cryptomonads. Extreme events, i.e., torrential rains and severe droughts, governed by the hydrological regime of the semi-arid region led to strong altered availability of resources in the watershed, directing the spatial and temporal dynamics of the phytoplankton in the Cruzeta man-made lake. The results showed an unusual behavior of the phytoplankton community contradicting the expectations about the climatic change scenario. Instead of an expected increase in cyanobacteria, the severe drought led to low biomass and resources, favoring diatoms and cryptomonads.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cultural or artificial eutrophication or the excessive input in surface waters of nutrients such as nitrogen and phosphorus is considered the largest and most widespread water quality problem in the world, especially in urbanized watersheds (Paerl 2009; Schindler 2012). The increased nutrient availability leads to overgrowth of phytoplankton and, as a consequence to aggravated decomposition of organic matter, the depletion of dissolved oxygen that may cause death of fish and other aquatic organisms, loss of the scenic quality of the environment and toxic cyanobacteria proliferation that may further impair ecosystem services (Carpenter et al. 1998).
Those cyanobacterial blooms are widespread throughout the world and are considered one of the biggest problems in freshwater ecosystems. They are associated with changes in the organoleptic aspects of water, i.e., they are unsightly, produce unpleasant odors, cause ecological damage, such as changes in food chains, with potential effects on nutrient cycling and biodiversity, are a significant risk to human health and wildlife (Fernandes et al. 2009; Soares et al. 2009; Paerl et al. 2011) and lead to increased costs for water treatment (Carmichael et al. 2001).
The global expansion of cyanobacterial blooms is predominantly associated with the availability of nutrients in the watershed (eutrophication), increased surface temperature of the water caused by global climate change (Smith 1986; O’Neil et al. 2012), a stable water column (Reynolds 1987) and the high residence time of the reservoirs (Fernandes et al. 2009). Dominance of cyanobacteria in the phytoplankton community is related to the ability of these organisms to: (1) regulate their position in the water column (Reynolds 1987), (2) tolerate low light (Smith 1986) and an alkaline pH (Shapiro 1990), (3) store phosphorus (Blomqvist et al. 1994), (4) fix dissolved N2 (Padisák 1997) and (5) resist grazing due to unpalatability and production of toxins (Bouvy et al. 1999).
The incidence and intensity of cyanobacterial blooms are on the rise worldwide (O’Neil et al. 2012), and a link between global warming and the worldwide proliferation of cyanobacterial blooms has been suggested (Paerl and Huisman 2009; O’Neil et al. 2012; Paerl and Paul 2011). Also in Brazil, recordings of cyanobacterial blooms show a clear increase in intensity and frequency, also often with perennial dominance of cyanobacteria, especially in reservoirs (Huszar and Silva 1999; Bicudo et al. 1999; Beyruth 2000; Calijuri et al. 2002; Bouvy et al. 2003; Crossetti and Bicudo 2008; Soares et al. 2009, 2012). In northeastern Brazil, several studies have reported cyanobacteria dominance in various water supply sources, mostly occurring under stratified conditions and as a result of high temperatures and high nutrient levels (Huszar et al. 2000; Bouvy et al. 2000; Diniz 2005; Bittencourt-Oliveira et al. 2011).
Hydrological and climatic factors play important roles in changes in water (and nutrients) inflow, outflow, water level and retention time of reservoirs that directly affect limnological variables (Naselli-Flores and Barone 2005; Soares et al. 2012; Romo et al. 2013). Therefore, seasonal fluctuations in the water level of reservoirs are reflected in changes in the physical, chemical and biological properties of these systems and may result in a decrease in the water quality in aquatic ecosystems during periods characterized by low rainfall and reduced water storage (Arfi 2003; Naselli-Flores and Barone 2005). These conditions, combined with high irradiation at elevated temperatures and eutrophication, may favor the growth of cyanobacteria year-round (Romo et al. 2013).
Semi-arid regions are characterized by peculiar climatic and hydrological conditions, with long periods of drought, few and short periods of rain, a relatively low-to-moderate wind speed, high evaporation rate and, hence, a relatively high residence time of water in the ponds, lakes and reservoirs (Chellappa et al. 2009). In addition, during periods of prolonged drought, which is typical of these regions, there is a significant loss of water due to evaporation, where the quality and availability of water can become a critical factor for the economic development of a region (Freire et al. 2009). As a consequence, the climatic characteristics of these regions, manifested in annual cycles of drought and rain, cause large fluctuations in the volume of water accumulated in reservoirs, leading to changes in the stability of the water column and in the community composition and biomass of cyanobacteria (Bouvy et al. 2003; Dantas et al. 2011, 2012). Additionally, in these warmer low-latitude systems, predicted increase in extreme events—like higher precipitation intensities and consequently higher flushing rates—may result in a stronger reset of the biomass in these systems as well as a community dominated by species more closely adapted to variable environments (De Senerpont Domis et al. 2013a).
Stimulated by a recent discussion on expected phytoplankton dynamics in the tropics (De Senerpont Domis et al. 2013a, b; Sarmento et al. 2013) and the resulting plea for more data on plankton dynamics from the tropics, we present here a study on phytoplankton dynamics in a man-made lake (Cruzeta) in the semi-arid northeastern Brazil. Based on the eutrophic conditions of the Cruzeta reservoir, we hypothesized that the hydrological regime of this tropical semi-arid region is a key factor in the availability of resources, which influences the composition and species dominance of cyanobacteria. The aim of this study was to analyze changes in the biomass and species composition during annual hydrological cycles and to evaluate the driving factors of phytoplankton dynamics in a tropical semi-arid reservoir. As our study includes a normal annual cycle, as well as extreme events (heavy rains followed by severe drought) that are predicted to occur more frequently in the future, i.e., prolonged drought spells alternating with intense precipitation (Marengo et al. 2010), it also gives an insight in expected, more common future scenarios.
Materials and methods
Study area
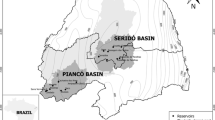
The Cruzeta man-made lake (06°24′42′′S, 36°47′23′′W) is located in Cruzeta city, in a semi-arid region in northeastern Brazil. The climate is warm, with an average rainfall of 550 mm/year, characterized by a rainy season between the months of February–June, with other months marked by a significant scarcity of rainfall (see Fig. 1 in Supplementary information).
This tropical reservoir was built in 1929 from the São José River and is used as water supply, for irrigation, fishing, recreation and watering livestock. In actuality, the maximum accumulation capacity is approximately 23,546,000 m3, with a surface area of 6.16 km2, maximum depth of 8.7 m and average depth of 4.7 m. The average retention time is 120 days with a regular water cycle (Bezerra et al. 2014).
Sampling
Water samples were taken at monthly intervals from July 2010 to June 2012 at two sampling stations in the pelagic zone of the Cruzeta man-made lake (Fig. 1). Sampling station 1 (P1) was located near the dam (z max = 8.7 m), and sampling station 2 (P2) was located near to the main tributary and under the influence of the river during the rainy season (z max = 4.4 m).
The vertical profiles for the temperature, pH, dissolved oxygen and conductivity were measured in situ using a multi-sensor probe (Hydrolab DS5) at 1-m intervals from the surface to the bottom. Water transparency was estimated using the Secchi disk extinction depth. Integrated samples (between 0.5 and 3 m depth) for nutrients and phytoplankton analyses were collected with a Van Dorn bottle (2 L). Phytoplankton samples were fixed with Lugol for later identification and counting.
Sample analysis
The turbidity (NTU) was measured using a turbidimeter (HACH 2100P). The total nutrients (nitrogen and phosphorus) were measured using a spectrophotometric method (Valderrama 1981). Soluble reactive phosphorus (SRP) was measured spectrophotometrically on water filtered on 0.45-µm glass-fiber filters (Murphy and Riley 1962). Total and fixed suspended solids (=inorganic solids) were determined by gravimetry after drying the filters overnight at 100 °C and ignition of filters at 500 °C for 3 h (APHA 2012). The volatile suspended solids (=organic solids) were measured by the difference between total suspended solids and fixed suspended solids (APHA 2012).
The identification and counting of phytoplankton was performed using, respectively, standard optical microscope (1000×) and inverted microscope (400×). Individuals (cells, colonies, filaments) are listed in random fields (Uhelinger 1964) using a sedimentation technique (Utermöhl 1958), and at least 100 specimens of the most abundant species were counted (Lund et al. 1958).
The biovolume (mm3 L−1) was calculated from the approximate geometric shapes (Hillebrand et al. 1999) and assuming the unit fresh weight, expressed as mass 1 mm3 L−1 = 1 mg L−1 (Wetzel and Likens 2000). The species considered dominants were defined as those that contributed more than 5 % of the total biomass and that together in the sample, accounted for more than 80 % of the biomass. Species diversity (H’) was estimated using the Shannon–Weaver index (Shannon and Weaver 1963) based on biomass and is expressed in bits per milligram.
Monthly rainfall data and the historical averages were provided by the Agricultural Research Company of the Rio Grande do Norte State (EMPARN). The rainy months of the study were considered those with precipitation equal to or above the historical average rainfall in the region, whereas the dry months were considered those with precipitation equal to or below the historical average.
The hydraulic retention time of the reservoir was calculated from the ratio of the average volume (m3) and the total outflow of the reservoir (m3 year−1), which was obtained by adding the outflow, water spill, to the water supply for irrigation and evaporation over the entire study period.
The euphotic zone (z eu) depth was estimated by multiplying the Secchi depth with a factor 2.7 (Cole 1994). The temperature profile was used to determine the mixing and stratification of the water column using a minimum difference of 0.5 °C for the thermal gradient (Dantas et al. 2012). The euphotic zone and maximum depth ratio (z eu/z max) were used to assess the availability of light (Jensen et al. 1994).
Data analysis
As a first approach, we performed an exploratory analysis as a descriptive statistic by Statistica®. For a description of the relationships between the dominant cyanobacteria species and the environmental variables investigated, a redundancy analysis (RDA) was performed. The data on the species’ abundances were previously analyzed by correspondence analysis and are not biased by detrended correspondence analysis (DCA), indicating that a linear ordering model would be more appropriate. These ordinations were performed using the software PC-ORD version 6.0 (McCune and Mefford 2011). The significance of the variables was analyzed using the Monte Carlo permutation test.
Results
Meteorological and hydrological scenario
During the study (July 2010–June 2012), three distinct periods were identified, defined by rainfall (Fig. 2) and water level fluctuations (Fig. 3): (I) normal dry season and low water level (months from July 2010 to January 2011) with an atypical rainfall of approximately 90 mm during October; (II) heavy rainy season with a higher rainfall above the historical average and high water level with overflow (from February to August 2011); and (III) severe drought and low water level (from September 2011 to July 2012). This period was considered an extended dry season, because the months that should rain (from January to May) had rainfall below to historical average (Fig. 2).
At the beginning of the rainy season (January 2011), Cruzeta reservoir was only 35 % of the total storage capacity. Due to heavy rains in the Period II, with records above the historical average (Fig. 2) and consequently the high flushing (water input), the reservoir rapidly filled up reaching its maximum capacity in May 2011. The reservoir remained completely filled until August 2011, and all surplus water from additional rain resulted in an overflow (Fig. 3). From September 2011 (Period III) onward, there was a progressive decrease in volume caused by the drought, reaching its lowest accumulation capacity, approximately 30 % of the total storage capacity (Fig. 3). The hydraulic retention time was 164 days for the period of study.
Limnological scenario
The temperature profile at sample station P1 (near to the dam) indicates that the system was mixed for most of the study period and that it could be classified as warm polymictic with some episodes of stratification (Fig. 4). During the end of the period I (normal dry season) and the beginning of the period II (heavy rainy season 2011), as well as late in period III (severe drought), the surface water temperature exceeded 30 °C (Fig. 4).
The reservoir showed low average depths throughout the study and during all periods, with the greatest depth observed at a sampling station near the dam (P1) at the period II (rainy season) and the lowest recorded depth at a sampling station near the river tributary (P2) during the periods I and III (dry seasons) (Table 1).
The Cruzeta reservoir is a turbid environment, with low light availability (low values of the ratio z eu/z max) during both dry and wet periods (Fig. 5). Temporal variation of the photic zone behaved similarly at both sampling stations (Table 1). The lowest light availability occurred during the 2010 drought (z eu = 0.6 m) in the deepest part of the reservoir (P1) with 9 % of the incident light availability. The low light availability in Cruzeta was due to different sources of turbidity during the study period. In period I, light extinction was mainly caused by the high phytoplankton biomass, which is reflected in high values of volatile suspended solids (Table 1), whereas in period III—mainly at the end of the study—high levels of inorganic particles (fixed suspended solids) caused high turbidity (Fig. 5).
The reservoir could be classified as eutrophic based on the total nitrogen and total phosphorus concentrations measured in all hydrological periods (Table 1). During the dry season of 2010 (period I), volatile suspended solids, total phosphorus, soluble reactive phosphorus and total nitrogen showed higher median values compared to the other periods (Table 1). During period III (severe drought), lower values of soluble reactive phosphorus (Fig. 6) and total phosphorus were measured at both sampling stations (Table 1).
Phytoplankton dynamics
A total of 62 phytoplankton taxa were identified during the study. Green algae (Chlorophyceae) contributed most to all taxa (24), followed by cyanobacteria (23), diatoms (Bacillariophyceae) (7), Cryptophyceae (5) and Euglenophyceae (2). However, the density and phytoplankton biomass were dominated by cyanobacteria during all periods studied (99 % of the total biomass).
The cyanobacteria species Cylindrospermopsis raciborskii (Woloszyska) Seenayya et Subba Raju, Microcystis panniformis Komárek et al. and Sphaerocavum brasiliensis Azevedo and Sant’Anna together contributed 97.8 % to the total biomass in P1 and P2 and 90.4 % throughout the study. Other taxa of cyanobacteria were considered dominants, such as Planktothrix isothrix (Skuja) Komárek and Komárková and Aphanizomenon gracile Lemmerrmann.
The phytoplankton biomass was highest during period I (dry season of 2010), decreasing during subsequent periods (Fig. 6), with the lowest mean value at both sampling stations found during the severe drought (Period III) (Table 1). Phytoplankton biomass at sampling station 1 (P1) was higher than at P2 (Table 1).
Seasonal composition of phytoplankton was driven by hydrological scenario and seasonal conditions (Fig. 7):
Period I: Normal dry season with low water level: Dominance of colonial cyanobacteria species M. panniformis and Sphaerocavum brasiliensis (Fig. 7). The highest phytoplankton biomass values (median value = 1334.6 mg L−1) were found in this period (Fig. 6), while species richness and diversity were very low (Table 1). Nutrient concentrations were higher compared to the other periods (Table 1), especially SRP (Fig. 6).
Period II: Rainy season (heavy rains) with higher water level and overflow: Dominance of filamentous cyanobacteria species C. raciborskii, A. gracile and P. isothrix (Fig. 7), mainly in the mixing period (before the overflow) at the sampling station 1 (P1). Phytoplankton diversity was consistently low; however, species richness was somewhat higher than in the previous period (Table 1). The biomass declined (Fig. 6). The high water volume with overflow (May–August 2011) resulted in more light in the reservoir in this period compared to period I.
Period III: Severe drought with low water level—in the beginning of the period, there was dominance of the heterocystous species C. raciborskii (cyanobacteria), during a mixing thermal profile (Fig. 7). M. panniformis, S. brasiliensis and C. raciborskii were coexisting during the following period with a thermal stratification. In the end of the studied period, lower dominance of cyanobacteria and higher incidence of cryptomonads (Cryptomonas spp.) and diatoms (Aulacoseira granulata, Cyclotella striata and Nitzschia palea) were observed at both sampling stations (Fig. 7). The lowest phytoplankton biomass and SRP occurred in this period of severe drought (Fig. 6).
The samples were distributed in the RDA according to the main features of the periods and sampling stations. Axis 1 of the RDA accounted for explaining 17.6 % of the relationship between the phytoplankton species and environmental variables and Axis 2 10.9 % (Fig. 8). The Monte Carlo test showed that the canonical axes were significant (Axis 1, P < 0.001; permutation 1999).
Redundancy analysis (RDA) ordination diagram of the Cruzeta reservoir, including abiotic variables, dominant species and periods. Vol volume of the reservoir, DO dissolved oxygen, FS fixed suspended solids, VS volatile suspended solids, SRP soluble reactive phosphorus, z eu euphotic zone, Biom phytoplankton biomass. Aph gra = Aphanizomenon gracile; Cylindr = Cylindrospermopsis raciborskii; Crypto ac = Cryptomonas acuta; Crypto er = Cryptomonas erosa; Crypto py = Cryptomonas pyrenoidifera; Crypt sp = Cryptomonas sp.; Micro pan = Microcystis panniformis; Plankt s = Planktothrix sp.; Sph bras = Sphaerocavum brasiliensis
Samples were distributed in RDA triplot according to the main characteristics of the three periods, and the first axis was supported by the hydrological regime of the semi-arid region (Fig. 8). Clearly, three distinct clusters appeared: (1) one characteristic for the normal dry season, (2) one characteristic for the rainy season and (3) one characteristic for the extreme drought. The positive side identified the sampling units of the period I (dry season of 2010), characterized by lower volumes and light, and higher values of volatile suspended solids (r = 0.41), fixed suspended solids (r = 0.82), soluble reactive phosphorus (r = 0.63) and phytoplankton biomass (r = 0.61), contributing to a better performance of the colonial cyanobacteria M. panniformis and S. brasiliensis. On the negative side, the sampling units were found that predominantly could be characterized by a higher water volume (r = −0.86), higher euphotic zone (r = −0.68) in the period II, consequence of higher rainfall, conditions that contributed to the dominance of C. raciborskii, P. isothrix, A. gracile species. Cryptomonas spp. and diatoms occurred in the end of the period, with the severe drought (period III) with lower values of soluble reactive phosphorus and phytoplankton biomass.
Discussion
Arid and semi-arid regions are characterized by seasonal fluctuations in the level of the water supply due to long drought periods and short rainy periods. The absence of rainfall during the dry season affects the water consumption rate, reduces the volume of the reservoirs and increases the water retention time, while the rivers and streams dry up. The water level in reservoirs is determined by rainfall in the drainage basin and water use, so that rainfall and anthropogenic uses influence the light regime (Xiao et al. 2011). Hence, such water level changes can modify the trophic state of the systems and, consequently, influence the dynamics of the phytoplankton (Bouvy et al. 2003; Barbosa et al. 2012).
The results indicated that the water quality and the phytoplankton composition were driven by extreme events, torrential rains and severe drought, characteristic of the hydrological regime of a semi-arid region, changing the fluctuation of the volume of the reservoir.
The results of this study showed that normally Cruzeta man-made lake is a shallow eutrophic ecosystem dominated by cyanobacteria, reinforcing the results found in other recent studies in the same system (Freitas et al. 2011; Bezerra 2011). Eutrophic conditions, characterized by high concentrations of nutrients, particularly phosphorus, are strongly related to the dominance of cyanobacteria in water bodies (Padisák and Reynolds 1998; Haande et al. 2011).
The dynamics of the phytoplankton displayed a difference in seasonality ruled by fluctuations in rainfall and the water level of the water supply reservoir. Naselli-Flores (2000) demonstrated that water level fluctuations influence the composition of phytoplankton in reservoirs of different trophic states. The volume reduction (90 %) in the semi-arid Mediterranean reservoirs over a period of extremely high demand and water use interferes with the stability of the water column, with consequences on the dynamics of phytoplankton and nutrients from those reservoirs, leading to eutrophication and an increased incidence of potentially toxic cyanobacteria species such as Microcystis (Naselli-Flores 2003). Studies of phytoplankton composition in the Brazilian semi-arid tropical reservoirs have also shown the same pattern of influence of volume changes in the temporal distribution of phytoplankton biomass (Costa et al. 2009; Dantas et al. 2012).
In general, the phytoplankton biomass in Cruzeta man-made lake was dominated by cyanobacteria, but due to the water level fluctuation and availability of the resources, the cyanobacteria composition changed between colonial and filamentous morphotypes as a result of different adaptive strategies. During the dry season of 2010 (period I), low water volume, thermal stratification, increased availability of phosphorus and a lower light availability favored colonial cyanobacteria M. panniformis and S. brasiliensis. The high biomass of these species did not hold up with the washout due to the high rainfall, and biomass rapidly declined, which is in line with general predictions (De Senerpont Domis et al. 2013a). In the subsequent period II, turbulent mixing favored filamentous species, as C. raciborskii and Planktothrix isothrix. These species have similar ecological traits, share some dimensions of the ecological niche (Bonilla et al. 2012) and are known to be tolerant to turbulent conditions (mixed water column), buoyancy regulation and shade tolerance (Reynolds 1993; Padisák and Reynolds 1998; Mischke 2003; Bonilla et al. 2012).
Colonial cyanobacteria reappeared and co-existed with C. raciborskii in the reservoir during the beginning of the period III, despite the lower biomass, when the reservoir displayed a decreasing water volume. Cyanobacteria further declined with ongoing lowering in water volume and eventually were replaced by diatoms and cryptophytes. Inasmuch as strongly reduced rainfall also implies less nutrient input in the system, this decrease in nutrient availability could have been one of the underlying mechanisms as reflected in low concentrations of available phosphorus. Diatoms can be potent competitors for nutrients, especially phosphorus, (Sommer 1988) and also thrive well under relatively low light (Reynolds 2006). Low light requirement seems to be a key factor in the success of cryptomonads species (Klaveness 1988). Indeed, cryptomonads flourished in high turbid, shallow drying up pond (Barone and Naselli-Flores 2003). Hence, the high inorganic turbidity in Cruzeta might have been in favor of these species. The lower water table makes sediment ressuspension more probable that could either negatively affect phytoplankton through turbidity (inorganic particles), and also promote settling of cells when water column is stable, or promote phytoplankton through release of nutrients. The latter is not expected in Cruzeta reservoir with iron-rich soils and clay (Red Yellow Latosol Soil; Embrapa 1971) and favorable redox conditions in the water column and near the sediment (Chellappa et al. 2008).
Cylindrospermopsis and Microcystis are common genera in tropical waters (Huszar and Silva 1999; Hoeger et al. 2004; Soares et al. 2009). Sphaerocavum is typically found in eutrophic waters and co-occurs with Microcystis species, as they are genera that inhabit similar environmental conditions (Fonseca and Bicudo 2008; Carvalho et al. 2008). The success of these populations has often been associated with a high tolerance to high insolation and stratified conditions (Reynolds 2006; Romo et al. 2013), corroborating their dominance during the summer and autumn months in tropical reservoirs (Soares et al. 2009) or during late summer months in temperate systems (Reynolds 2006) when most of the deep systems are stratified. However, in semi-arid tropical reservoirs, cyanobacteria of genus Microcystis have been reported during periods of transition and during the rainy season (Costa et al. 2009), while in the semi-arid Mediterranean reservoirs, blooms of Microcystis species occur during the summer with high stability of the water column (Naselli-Flores 2003). Our results also report the dominance of Microcystis and Sphaerocavum in dry periods during a reduced water level with stratified water column and high phosphorus. But, during the rainy season with torrential rains, the reservoir was quickly filled with water outflow, resulting in the disappearing of the biomass of these species, highlighting the importance of the hydrological regime.
Cylindrospermopsis raciborskii was present during almost the entire study period, except during periods of lower reservoir volume when there was a total dominance of colonial cyanobacteria (normal dry season) or diatoms and cryptomonads (extreme drought). The presence of this nitrogen-fixing species has been associated with a greater plasticity of adaptations to environmental variables (Bonilla et al. 2012; Bittencourt-Oliveira et al. 2011). The main factors affecting C. raciborskii blooms are related to advantages over other phytoplankton species, such as the ability to tolerate high water temperatures, low nutrient concentrations, low water clarity and high salinities and pH (Briand et al. 2002; Bouvy et al. 2003; Bonilla et al. 2012; Bittencourt-Oliveira et al. 2011). Based on a dataset analysis performed by Soares et al. (2013), cyanobacterial blooms of Cylindrospermopsis were found at lower latitudes in Brazil, being most commonly reported during dry periods and in mixed systems, or forming a perennial dominance under high phosphorus concentrations. Additionally, the success of C. raciborskii is also described to their ability to store phosphorus and to use the low light under conditions of destratification or mixing (Burford et al. 2006). Cylindrospermopsis can be considered well adapted to both stratified (Berger et al. 2006) and mixing periods (Burford et al. 2006), and periods of its dominance are commonly reported during mixed conditions, which underpin the wide tolerance of Cylindrospermopsis to mixing (Bonilla et al. 2012). This pattern was observed in the Cruzeta, where the dominance of C. raciborskii species occurred during the rainy season with larger volumes of water and also during the following dry seasons with mixing of the water column.
In a recent discussion about the plankton dynamics in a fast-changing world (De Senerpont Domis et al. 2013a, b; Sarmento et al. 2013), it was postulated that in tropical systems, temporal variability in precipitation, and consequently hydrological cycle, can be an important driver of the seasonal dynamics of plankton communities. The intensification of rainfall in tropical areas, defended by De Senerpont Domis et al. (2013a, b), will have stronger consequences for hydrology-driven systems, where increasing the incidence of high flushing events will lead to increase washout. With anticipated higher precipitation intensities in these systems, higher flushing rates may result in a stronger reset of the biomass build-up as well as a community dominated by species more closely adapted to variable environments (De Senerpont Domis et al. 2013a, b). But, as argued by Sarmento et al. (2013), the events of drought, as predicted by regional climate models for South America (Marengo et al. 2010), have to be considered too. Semi-arid watersheds of northeast Brazil are more sensitive to reduced rainfall and water flow, so this region will tend to be more severely affected by climate change compared to the humid tropical systems (Roland et al. 2012). Indeed, our results indicated that a high biomass with cyanobacteria dominance can be found in a dry season, but that extreme events (both torrential rains with overflow and severe drought) can change the hydrological scenario of the semi-arid systems, resulting in a washout during the rainy season, and an unexpected response during severe drought. The severe drought event in 2012 is the worst drought in the last 30 years according to the Brazilian National Water Agency (ANA). Projection models claim with confidence that arid and semi-arid areas are more susceptible to climate changes (IPCC 2013). Vulnerability to climate variability in these regions is characterized mainly by an increase in the frequency of prolonged drought and decrease water flow, causing the deterioration of water quality (IPCC 2007; Marengo 2011).
The unusual behavior of phytoplankton community contradicted the predictions about the climatic change scenario, which is cyanobacterial dominance, especially in shallow lakes (Moss et al. 2011). Instead of an expected increase in eutrophication leading to cyanobacterial blooms (Marengo et al. 2010; Moss et al. 2011; Roland et al. 2012), the lowering water table resulted in less biomass and resources, promoting diatoms and cryptomonads due low light by inorganic particles. This example underscores the importance of the call for more data from tropical systems, as this sole example already refutes the hypothesis that under severe drought, the water will ultimately turn blue-green. The climate pressures on the tropical semi-arid/arid systems are likely strong (e.g., salinization and high water residence times), and the need for more data on these understudied systems is therefore urgent, as suggested by De Senerpont Domis et al. (2013a, b) and Sarmento et al. (2013).
Understanding processes and their relationships with the hydrological regime will enable more accurate predictions on plankton response and possible cyanobacterial blooms in semi-arid reservoirs in regions experiencing more extreme events.
In summary, extreme events such as torrential rains and severe droughts, which are controlled by the hydrological cycle of semiarid regions, determine the availability of resources in the watershed, influencing the changing composition of dominant cyanobacteria species. The inflow of water from the tributaries and overflow reservoir contributed to the lower phytoplankton biomass and modified the populations of cyanobacteria, favoring the dominance of filamentous species with mixing tolerance, such as C. raciborskii. The lower water volume and high residence water in the reservoir were favorable for the growth of colonial cyanobacteria species, such as M. panniformis and S. brasiliensis, which contributed to the higher phytoplankton biomass values. But during a severe drought, the low water level can favor the adaptive phytoplankton species with requirement for low resources by inorganic particles, as diatoms and cryptomonads, an unusual behavior of phytoplankton community contradicted the expectations about the climatic change scenario. This behavior indicates that predictions should take into account the regional climate, lake type (size, depth) and watershed size, beyond expected changes in rainfall and temperature from the IPCC scenarios according to the region.
References
American Public Health Association (APHA) (2012) Standard methods for the examination of water and waste-water, 22nd edn. APHA, AWWA and W.E.F., Washington
Arfi R (2003) The effects of climate and hydrology on the trophic status of Sélingué reservoir, Mali, West Africa. Lakes Reserv Res Manage 8:247–257
Barbosa JEL, Medeiros ESF, Brasil J, Cordeiro RS, Crispim MCB, Silva GHG (2012) Aquatic systems in semi-arid Brazil: limnology and management. Acta Limnol Bras 24:103–118
Barone R, Naselli-Flores L (2003) Distribution and seasonal dynamics of Cryptomonads in Sicilian waterbodies. Hydrobiologia 502:325–329
Berger C, Ba N, Gugger M, Bouvy M, Rusconi F, Couté A, Trousselier M, Bernard C (2006) Seasonal dynamics and toxicity of Cylindrospermopsis raciborskii in Lake Guiers (Senegal, West Africa). FEMS Microbiol Ecol 57:355–366
Beyruth Z (2000) Periodic disturbances, trophic gradient and phytoplankton characteristics related to cyanobacterial growth in Guarapiranga Reservoir, São Paulo State, Brazil. Hydrobiologia 424:51–65
Bezerra AFM (2011) Balanço de massa de fósforo e a eutrofização em reservatórios do semiárido do Rio Grande do Norte – Brasil. Dissertation. Federal University of Rio Grande do Norte
Bezerra AFM, Becker V, Mattos A (2014) Balanço de Massa de Fósforo Total e o Estado Trófico em Reservatórios do Semiárido Brasileiro. Rev Bras Recur Hídr 19:67–76
Bicudo CEM, Ramírez RJJ, Tucci A, Bicudo DC (1999) Dinâmica de populações fitoplanctônicas em ambiente eutrofizado: O Lago das Graças, São Paulo. In: Henry R (ed) Ecologia de reservatórios: estrutura, função e aspectos. FAPESP, Botucatu, pp 451–507
Bittencourt-Oliveira MC, Moura AN, Hereman TC, Dantas EW (2011) Increase in straight and coiled Cylindrospermopsis raciborskii (Cyanobacteria) populations under conditions of thermal de-stratification in a shallow tropical reservoir. J Water Resour Prot 3:245–252
Blomqvist P, Pettersson A, Hyenstrand P (1994) Ammonium–nitrogen: a key regulatory factor causing dominance of non-nitrogen-fixing Cyanobacteria in aquatic systems. Arch Hydrobiol 132:141–164
Bonilla S, Aubriot L, Soares MCS, González-Piana M, Fabre A, Huszar VLM, Lürling M, Antoniades D, Padisák J, Kruk C (2012) What drives the distribution of the bloom-forming cyanobacteria Planktothrix agardhii and Cylindrospermopsis raciborskii? FEMS Microbiol Ecol 79:594–607
Bouvy M, Molica R, Oliveira S, Marinho M, Beker B (1999) Dynamics of a toxic cyanobacterial bloom Cylindrospermopsis raciborskii) in a shallow reservoir in the semi-arid region of Northeast Brazil. Aquat Microb Ecol 20:285–297
Bouvy M, Falcão D, Marinho M, Pagano M, Moura A (2000) Occurrence of Cylindrospermopsis (Cyanobacteria) in 39 Brazilian tropical reservoirs during the 1998 drought. Aquat Microb Ecol 23:13–27
Bouvy M, Nascimento SM, Molica RJR, Ferreira A, Huszar V, Azevedo SMFO (2003) Limnological features in Tapacurá reservoir (Northeast Brazil) during a severe drought. Hydrobiologia 493:115–130
Briand JF, Robillot C, Quiblier-Llobéras C, Humbert JF, Couté A, Bernard C (2002) Environmental context of Cylindrospermopsis raciborskii (Cyanobacteria) blooms in a shallow pond in France. Water Res 36:3183–3192
Burford MA, Mcneale KL, Mckenzie-Smith FJ (2006) The role of nitrogen in promoting the toxic cyanophyte Cylindrospermopsis raciborskii in a subtropical water reservoir. Freshw Biol 51:2143–2153
Calijuri MC, Santos ACA, Jati S (2002) Temporal changes in the phytoplankton community structure in a tropical and eutrophic reservoir (Barra Bonita, S.P.—Brazil). J Plankton Res 24:617–634
Carmichael WW, Azevedo SMFO, An JS, Molica RJR, Jochimsen EM, Lau S, Rinehart KI, Shaw GR, Eaglesham GK (2001) Human fatalities from cyanobacteria: chemical and biological evidence for cyanotoxins. Environ Health Perspect 109:663–668
Carpenter SR, Cole JJ, Kitchell JF, Pace ML (1998) Impact of dissolved organic carbon, phosphorus, and grazing on phytoplankton biomass and production in experimental lakes. Limnol Oceanogr 43:73–80
Carvalho LR, Pipole F, Werner VR, Laughinghouse HD IV, Camargo ACM, Rangel M, Konno K, Sant’Anna CL (2008) A toxic cyanobacterial bloom in an urban coastal lake, Rio Grande do Sul State, Southern Brazil. Braz J Microbiol 39:761–769
Chellappa NT, Borba JM, Rocha O (2008) Phytoplankton community and physica–chemical characteristics of water in the public reservoir of Cruzeta, RN, Brazil. Braz J Biol 68:477–494
Chellappa NT, Chellappa T, Câmara FRA, Rocha O, Chellappa S (2009) Impact of stress and disturbance factors on the phytoplankton communities in Northeastern Brazil reservoir. Limnologica 39:273–282
Cole GA (1994) Textbook of limnology. Waveland Press Inc, Illinois
Costa IAS, Cunha SRS, Panosso RF, Araújo MFF, Melo JL, Eskinazi-Sant’Anna EM (2009) Dinâmica de cianobactérias em reservatórios eutróficos do semi-árido do Rio Grande do Norte. Oecol Bras 13:382–401
Crossetti LO, Bicudo CEM (2008) Phytoplankton as a monitoring to in a tropical urban shallow reservoir (Garças pond): the assemblage index application. Hydrobiologia 610:161–173
Dantas EW, Moura AN, Bittencourt-Oliveira MC (2011) Cyanobacterial blooms in stratified and destratified eutrophic reservoirs in semi-arid region of Brazil. An Acad Bras Cienc 83:1327–1338
Dantas EW, Bittencourt-Oliveira MC, Moura AN (2012) Dynamics of phytoplankton associations in three reservoirs in northeastern Brazil assessed using Reynolds’ theory. Limnologica 42:72–80
De Senerpont Domis LN, Elser JJ, Gsell AS, Huszar VLM, Ibelings BW, Jeppesen E, Kosten S, Mooij WM, Roland F, Sommer U, Van Donk E, Winder M, Lürling M (2013a) Plankton dynamics under different climatic conditions in space and time. Freshw Biol 58:463–482
De Senerpont Domis LN, Elser JJ, Gsell AS, Huszar VLM, Ibelings BW, Jeppesen E, Kosten S, Mooij WM, Roland F, Sommer U, Van Donk E, Winder M, Lürling M (2013b) Plankton dynamics under different climate conditions in tropical freshwater systems (a reply to the comment by Sarmento, Amado and Descy). Freshw Biol 58:2211–2213
Diniz R (2005) Ritmos nictemerais e distribuição espaço-temporal de variáveis limnológicas e sanitárias em dois açudes do trópico semi-árido (PB). Ph.D. Thesis. Universidade Federal de Campina Grande
Empresa Brasileira de Pesquisa Agropecuária – EMBRAPA (1971) Levantamento Exploratório – Reconhecimento de solos do Estado do Rio Grande do Norte. http://www.uep.cnps.embrapa.br
Fernandes VO, Cavati B, Oliveira LB, Souza BD (2009) Ecologia de cianobactérias: fatores promotores e consequências das florações. Oecol Bras 13:247–258
Fonseca BM, Bicudo CEM (2008) Phytoplankton seasonal variation in a shallow stratified eutrophic reservoir (Garças Pond, Brazil). Hydrobiologia 600:267–282
Freire RHF, Calijuri MC, Santaella ST (2009) Longitudinal patterns and variations in water quality in a reservoir in the semiarid region of NE Brazil: responses to hydrological and climatic changes. Acta Limnol Bras 21:251–262
Freitas FRS, Righetto AM, Attayde JL (2011) Cargas de fósforo total e material em suspensão em um reservatório do semi-árido brasileiro. Oecol Aust 15:655–665
Haande S, Rohrlack T, Semyalo RP, Brettum P, Edvardsen B, Lyche-Solheim A, Sorensen K, Larsson P (2011) Phytoplankton dynamics and cyanobacterial dominance in Murchison Bay of Lake Victoria (Uganda) in relation to environmental conditions. Limnologica 41:20–29
Hillebrand H, Durselen CD, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35:403–424
Hoeger SJ, Shaw G, Hitzfeld BC, Dietrich DR (2004) Occurrence and elimination of cyanobacterial toxins in two Australian drinking water treatment plants. Toxicon 43:639–649
Huszar VLM, Silva LH (1999) Estrutura da comunidade fitoplanctônica no Brasil: cinco décadas de estudos. Limnotemas 2:1–21
Huszar VLM, Silva LHS, Marinho MM, Domingos P, Sant’Anna CL (2000) Cyanoprokariota assemblages in eight productive tropical Brazilian waters. Hydrobiologia 424:67–77
IPCC (Intergovernmental Panel on Climate Change) (2007) Climate change 2007: impacts, adaptation and vulnerability. Contribution of working group II to the fourth assessment report of the intergovernmental panel on climate change. Parry ML, Canziani OF, Palutikof, JP, van der Linden, PJ, Hanson CE (eds). Cambridge University Press, Cambridge. 976 pp
IPCC (Intergovernmental Panel on Climate Change) (2013) Climate change 2013: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Stocker, TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Cambridge University Press, Cambridge. 1535 pp
Jensen P, Jeppesen E, Olrik K, Kristensen P (1994) Impact of nutrients and physical factors on the shift from cyanobacterial to chlorophyte dominance in shallow Danish lakes. Can J Fish Aquat Sci 51:97–109
Lund JWG, Kipling C, Lecren ED (1958) The inverted microscope method of estimating algal number and the statistical basis of estimating by counting. Hydrobiologia 11:143–170
Klaveness D (1988) Ecoloy of Cryptomonadida: a first review. In: Sandgren CD (ed) Growth and reproductive strategies of freshwater phytoplankton. Cambridge University Press, Cambridge, pp 105–133
Marengo JA (2011) Variabilidade e Mudanças Climáticas no Semiárido. In: Medeiros SS, Gheyi HR, Galvão CO, Paz VP (eds) Recursos Hídricos em regiões áridas e semiáridas. INSA, Campina Grande, pp 383–416
Marengo JA, Ambrizzi T, Rocha RP, Alves LM, Cuadra SV, Valverde MC, Torres RR, Santos DC, Ferraz SET (2010) Future change of climate in South America in the late twenty-first century: intercomparison of scenarios from three regional climate models. Clim Dyn 35:1089–1113
Mccune B, Mefford MJ (2011) PC-ORD. Multivariate analysis of ecological data. version 6.0. MjM Software Design, Oregon
Mischke U (2003) Cyanobacteria associations in shallow polytrophic lakes: influence of environmental factors. Acta Oecol 24:S11–S23
Moss B, Kosten S, Meerhoff M, Battarbee RW, Jeppesen E, Mazzeo N, Havens K, Lacerot G, Liu Z, De Meester L, Paerl H, Scheffer M (2011) Allied attack: climate change and eutrophication Inland Waters. Inland Waters 1:101–105
Murphy J, Riley JP (1962) A modified single-solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31
Naselli-Flores L (2000) Phytoplankton assemblages in 21 reservoirs: relationship between species composition and environmental factors. Hydrobiologia 424:1–11
Naselli-Flores L (2003) Man-made lakes in Mediterranean semi-arid climate: the strange case of Dr. Deep Lake and Mr. Shallow Lake. Hydrobiologia 506(509):13–21
Naselli-Flores L, Barone R (2005) Water-level fluctuations in Mediterranean reservoirs: setting a dewatering threshold as a management tool to improve water quality. Hydrobiologia 548:85–99
O’Neil JM, Davis TW, Burford MA, Gobler CJ (2012) The rise of harmful cyanobacteria blooms: the potential roles of eutrophication and climate change. Harmful Algae 14:313–334
Padisák J (1997) Cylindrospermopsis raciborskii (Woloszynnska) Seenayya et Subba Raju, an expanding, highly adaptive cyanobacterium: worldwide distribution and review of its ecology. Arch Hydrobiol 107:563–593
Padisák J, Reynolds CS (1998) Selection of phytoplankton associations in lake Balaton, Hungary, in response to eutrophication and restoration measures, with special reference to the cyanoprokariotes. Hydrobiologia 384:41–53
Paerl HW (2009) Controlling eutrophication along the freshwater–marine continuum: dual nutrient (N and P) reductions are essential. Estuar Coast 32:593–601
Paerl HW, Huisman J (2009) Climate change: a catalyst for global expansion of harmful cyanobacterial bloom Environ Microbiol Rep 1:27–37
Paerl HW, Paul V (2011) Climate change: links to global expansion of harmful cyanobacteria. Water Res 46:1349–136
Paerl HW, Xu H, McCarthy MJ, Zhu G, Qin B, Li Y, Gardner WS (2011) Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): the need for a dual nutrient (N & P) management strategy. Water Res 45:1973–1983
Reynolds CS (1987) Cyanobacterial water-blooms. In: Callow J (ed) Advances in botanical research. Academic Press, London, pp 67–143
Reynolds CS (1993) Scales of disturbance and their importance in plankton ecology. Hydrobiologia 249:157–171
Reynolds CS (2006) The ecology of phytoplankton (ecology, biodiversity and conservation). Cambridge University Press, Cambridge
Roland F, Huszar VLM, Farjalla VF, Enrich-Prast A, Amado AM, Ometto JPHB (2012) Climate change in Brazil: perspective on the biogeochemistry of inland waters. Braz J Biol 72:709–722
Romo S, Soria J, Fernández F, Ouahid Y, Barón-Solá A (2013) Water residence time and the dynamics of toxic cyanobacteria. Freshw Biol 58:513–522
Sarmento H, Amado AM, Descy JP (2013) Climate change in tropical freshwaters (comment on the paper ‘Plankton dynamics under different climatic conditions in space and time’ by De Senerpont Domis et al. 2013). Freshw Biol 58:2208–2210
Schindler DW (2012) The dilemma of controlling cultural eutrophication of lakes. Proc R Soc 279:4322–4333
Shannon CE, Weaver W (1963) The mathematical theory of communication. Illinois University Press, Urbana
Shapiro J (1990) Currents beliefs regarding dominance by blue-greens: the case of the importance of CO2 and pH. Verh Internat Verein Limnol 24:38–54
Smith V (1986) Light and nutrient effects on the relative biomass of blue-green algae in lake phytoplankton. Can J Fish Aquat Sci 43:148–153
Soares MCS, Rocha MIA, Marinho MM, Azevedo SMFO, Branco CWC, Huszar VLM (2009) Changes in species composition during annual cyanobacterial dominance in a tropical reservoir: physical factors, nutrients and grazing effects. Aquat Microb Ecol 57:137–149
Soares MCS, Marinho MM, Azevedo SMFO, Branco CWC, Huszar VLM (2012) Eutrophication and retention time affecting spatial heterogeneity in a tropical reservoir. Limnologica 42:197–203
Soares MCS, Huszar VLM, Miranda MN, Mello MM, Roland F, Lürling M (2013) Cyanobacterial dominance in Brazil: distribution and environmental preferences. Hydrobiologia 717:1–12
Sommer U (1988) Phytoplankton succession in microcosm experiments under simultaneous grazing pressure and resource limitation. Limnol Oceanogr 33:1037–1054
Uhelinger V (1964) Étude statistique des méthodes de dénobrement planctonique. Arch Sci 17:121–123
Utermöhl H (1958) Zur vervollkommung der quantitativen phytoplankton - methodik. Mitt Int Ver Theor Angew Limnol 9:1–38
Valderrama JC (1981) The simultaneous analysis of total and phosphorus in natural waters. Mar Chem 10:109–122
Wetzel RG, Likens GE (2000) Limnological analyses, 3rd edn. Springer, New York
Xiao L-J, Wang T, Hu R, Han B-P, Wang S, Qian X, Padisák J (2011) Succession of phytoplankton functional groups regulated by monsoonal hydrology in a large canyon-shaped reservoir. Water Res 45:5099–5109
Acknowledgments
We are thankful to FINEP (Financiadora de Estudos e Projetos) Process No. 52009 and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) Process No. 483950/2011-4, for financial support. We are grateful to Anderson Felipe de Medeiros Bezerra, José Neuciano Pinheiro de Oliveira, Laíssa de Macêdo Torres, Mariana R.A. da Costa, Jurandir de Mendonça Junior, Maria da Conceição de Souza and Thársia da Silva Pinto for technical support (at field work and laboratory analyses). We also thank V. Becker Post-Doc fellowship (Process No. 4288/14-9) that was funded by the Federal Government of Brazil, Ministry of Education, through CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Ministério da Educação). This study was conducted under the CAPES/NUFFIC project “Cyanobacterial Blooms in a Changing World” in collaboration between Wageningen University (The Netherlands) and Federal University of Rio Grande do Norte.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Bas W. Ibelings.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Medeiros, L.C., Mattos, A., Lürling, M. et al. Is the future blue-green or brown? The effects of extreme events on phytoplankton dynamics in a semi-arid man-made lake. Aquat Ecol 49, 293–307 (2015). https://doi.org/10.1007/s10452-015-9524-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-015-9524-5