Abstract
Medicinal herbs have been used for thousands of years to heal burn wounds, skin ulcers, and sores. The yarrow plant (Achillea millefolium) is a well-known medicinal plant. The aim of this study was to evaluate the clinical and microbiological efficacy of the yarrow (alcoholic extract) on the healing of burn wounds in an animal model. Ten male New Zealand white rabbits, 4–6 months old and weighing 2,100 ± 240 g, were used. Experimental burn wounds (20 × 25 mm2) were created on the dorsal region of each animal. In the treatment group, the wounds were washed daily with normal saline followed by 5 ml of alcoholic extracted yarrow applied to each wounds. This procedure was repeated for 21 days. In the control group, the wounds were only washed with the same volume of normal saline over the same period. Clinical, histopathological, and microbiological evaluations of the burn wounds were carried out on days 0, 7, 14, and 21. Compared with the control group, wounds treated with the yarrow extract had a better quality of healing with a reduced microorganism count (p < 0.05). To conclude, topical application of alcoholic yarrow extract, as used in this study, not only improved the quality of healing of burn wounds but also reduced the microbiological burden of such wounds in rabbits.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Burn wounds have always been a major challenge to deal with, having both a high morbidity and mortality. Documents related to the medical care and treatment of the burn wounds can be traced back to ancient times (Lawrence 1994). Traditional remedies for the treatment of the burn wounds include the application of a vast variety of different products, such as natural oils, herb extracts, and home-made balms (Arora and Kaur 1999). Yarrow (Achillea millefolium) is a flowering plant in the family of Asteraceae. It grows widely in different parts of the world and is famous for its versatile medical applications (Moerman 1998). Yarrow is the interest of both traditional and modern medicine, and although it is commonly used across the world, it seems that its first use for medicinal purposes started from Europe where all parts of the plant including leaves, flowers, and stem were valued as having therapeutic qualities (Chevallier 1996; Perry 1980). The scientific name for yarrow is said to being derived from the legendary Greek hero Achilles who was the central character of Homer’s Iliad. According to this epic story, Achilles is said to have ordered his soldiers to carry and use yarrow preparations in the battlefields to treat their wounds (Kreutterbuch 1960). Since then and for the centuries, different types of yarrow have been used for treatment of different diseases, namely respiratory, gastrointestinal, cardiovascular, urogenital, and hepatobiliary conditions (Blumenthal et al. 1998; Bown 1995; Bradley 1992; Hoffmann 1990; Mills 1994). Its vast medicinal properties have drawn the attention and interest of scientists in many biological fields specially biochemists and physiologists (Saeidnia et al. 2005). Despite the fact that there is a plenty of anecdotal evidence regarding the therapeutic effect of this plant on wounds, surprisingly few scientific studies on the degree of efficacy of the plant on wound healing especially burn wounds exist (Nedelec et al. 2000; Richey et al. 1989).
The studies have shown that in patients with the burn wounds, the occurrence of infection is the main cause of the increased mortality rate which can reach as high as 50% (Subrahmanyam et al. 2001). It has been noted that the alcoholic extract of yarrow demonstrates some antibacterial and antifungal properties (Barel et al. 1991; Hausbn et al. 1991; Pattnaik et al. 1997). Thus, we hypothesized that the plant’s extract could have some beneficial effects on preventing infection of burn wounds which would in turn improve the quality of the wound healing. This study was set up to determine the degree of antimicrobial action and clinical efficacy of a topical application of alcoholic extract of yarrow plant in the healing process of experimental burn wounds in rabbits.
Materials and methods
Plant samples
For the purpose of this research, the aerial parts of the plant, including the flower, leaf, and stem, were harvested from its natural habitat around the city of Urmia during the flowering season (between May and July). The identification of collected plants were confirmed scientifically at the research laboratories of the Department of Agriculture of West Azerbaijan province.
Preparation of the extract
The plant extract was prepared using methods described by Aburjai and Hudaib (2006). Following their procedure, 100 g of Yarrow powder was added to 1,000 ml alcohol heated to 80°C and mixed in a laboratory shaker for 24 h. The mixture was filtered through a Büchner funnel using ordinary filter paper. The solution was then passed through a Whatman filter paper grade no. 1. The filtered solution was transferred to a rotary evaporator (Heidolph Laborota 4000 eco-Germany), and up to 80% of the solvent was recovered. The rest of the solvent was removed using a 30°C water bath. The alcoholic extract was filter-sterilized (0.2 μm, Chromafil) and stored at 4°C until it was used.
Animals
Ten male New Zealand white rabbits weighing 2,100 g ± 240 and aged between 4 and 6 months were used in this study. The animals were provided by the Experimental Animal House, Urmia University. They were randomly divided into two equal groups—control and treatment. The experiment was approved by the University Ethics Committee and conducted in accordance with its regulations and guidelines. Animals were kept under standard conditions described for experimental animals and were subjected to a daily 12-h light–12-h dark cycle with light on from 8 am. Temperature was kept at 23°C at all times. They were fed standard commercial rabbit pellets (Dan Pars Co. Tehran), and water was allowed ad libitum.
Experimental burn wounds
Anesthesia was induced by intramuscular injection of 5 mg/kg diazepam (Chemidarou, Iran) and was then maintained by intramuscular injection of 50 mg/kg ketamine hydrochloride (Alfasan, Woerden, Holland). With animals in ventral recumbency, their dorsal hair was clipped and shaved. The area was aseptically prepared and draped surgically. The Hoekstra’s method was used to cause the experimental burn wound (Brans et al. 1994). The square experimental burn wound (20 × 25 mm2) on the dorsum of the each rabbit was caused by a heated metal plate (170°C) applied to the shaven area with a pressure of 300 g for 10 s. In the treatment group, the wounds were washed daily with normal saline followed by an application of 5 ml of alcoholic extract of yarrow. However, in the control group, normal saline was used instead of yarrow extract. All wounds were dressed and bandaged routinely. No antibiotics were used throughout the experiment, and the animals were kept individually.
Clinical assessment
The clinical parameters in this study included gross features of the wounds and the rate of wound contraction in the two groups. The rate of wound contraction was calculated by subtracting the wound area from the area of the previous day and then multiplying it by 100. This was done by documenting the wounds by standard digital photograph on a daily basis and analyzing the data with Sigma Scan Pro 5.0 software (Sigma Scan Pro 5.0 software, SPSS Science, Chicago, IL, USA).
Microbiologic assessment
The microbiologic parameters in this study included determining the number and type of microorganisms colonizing the wounds. Swabs were applied to the wounds’ surface on a daily basis, and microorganisms were cultured in BHI agar. Furthermore, a specific culture media used for biochemical tests was conducted in order to differentiate the isolated microorganisms.
Histopathological assessment
The histopathological assessment of the healing process was performed on the 21st day of the study. Before samples were taken for analysis, animals were subjected to general anesthesia. The samples were collected from the wounds borders as well as from the middle of the wounds area. Samples were processed to paraffin, and sections were cut and stained with a standard hematoxylin/eosin stain.
Statistical methods
Differences between experimental and control groups were analyzed with an unpaired Student’s t test. Differences were considered significant if p < 0.05 (SigmaStat for Windows, version 2.03, Jandel Corporation, San Rafel, CA, USA).
Results
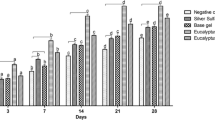
Before applying the yarrow extracts to the wounds, the number of settling bacteria on the surface of the wounds was 4 ± 0.8 × 104/cm2 and 5 ± 0.4 × 104/cm2 in the control and the treatment groups, respectively. The most commonly isolated bacteria from the wounds were Streptococcus pyogenes, Staphylococcus aureus, Escherichia coli, and Candida albicans. Figure 1 shows the total count of the isolated bacteria from the wound surfaces on days 1, 7, 14, and 21. In the treatment group, the total count for isolated bacteria from the wounds’ surface on day 1 was 5.8 ± 0.2 × 104/cm2; however, by day 7, the count had increased to 1.5 ± 0.4 × 105/cm2. On day 14, the total bacterial count in treated animals demonstrated a partial decrease to 9 ± 0.8 × 104/cm2. This decreasing trend in the treatment group continued until day 21 by which point there was a significant reduction in total bacterial count to 1 ± 0.6 × 104/cm2. At this stage, S. aureus was the most dominant isolated microorganism. Compared with the control group, the total count for isolated microorganisms from the wounds’ surface on days 1, 7, 14, and 21 were 4 ± 0.4 × 104/cm2, 3 ± 0.2 × 105/cm2, 4 ± 0.6 × 105/cm2, and 1 ± 0.5 × 105/cm2, respectively. On day 21, in the control group, S. pyogenes was the only isolated microorganism from the wound surfaces.
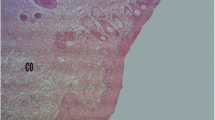
From a clinical perspective, the burn wounds healed in both groups without any apparent complications. The rates of wound contraction in the control and treatment groups have been compared in Table 1. From a histological aspect, on day 21 in the yarrow-treated group, the skin defect filled completely with compact granulation and significant amounts of collagen fibers with the least amount of inflammatory cells (Fig. 3). At the same time, in the control group, the wound cavity was filled with fresh granulation (Fig. 2).
Discussion
Currently, there are numerous clinical approaches in the treatment of burn wounds, and antibiotics are common drugs used to prevent and treat wound infections. However, the development of new resistant strains, and the issue of side effect and sensitivity reactions to current antibiotics have always been the matter of some concern. Thus, finding new antimicrobial material with higher efficacy and less side effects has been the centerpiece of any drug research program (Edwards-Jones et al. 2000). The use of botanical and herbal medicine has become widespread during recent years as they offer effective, side-effect free, cheap, and easy alternatives to conventional medicines. The yarrow plant, one of the famous members of the Asteraceae family, has been used frequently as a medicinal herb in western medicine. There are numerous reports documenting its application in treating diseases of different body systems (Dalsenter et al. 2004; Lans et al. 2007; Lemmens et al. 2006; Moore M 1993; Singh and Blumenthal 1998; Taylor and Francis 2001). However, little research has been conducted to evaluate its healing and antibacterial properties in an experimental setup (Nedelec et al. 2000; Richey et al. 1989).
According to the results of this study, wound contraction in all groups (control and experiment) occurred during days 7 through to 21 which indicates that the percentage change in wound area was constant during this period. The current results showed that in terms of the healing process and the rate of wound contraction, there was a statistically significant difference between the control and treatment groups on days 7, 14, and 21 (p < 0.05). Comparing the rate of wound contraction on day 21 between the two groups showed that in the treatment group, the wound edges almost reached the opposite sides. This resulted in a contraction rate of 99.42%, while this value for the control group on the same day was 81.91%, showing an incomplete wound closure (Table 1). These results demonstrate that the healing process and the wound contraction in the experimentally induced burn wounds in rabbits could be accelerated by using an alcoholic extract of yarrow. The results are in agreement with findings from similar studies (Nedelec et al. 2000; Richey et al. 1989).
Wound contraction involves a complex set of extracellular and cellular interactions that results in the closure of an open wound. Although there are several theories as to the exact mechanism, it is believed that movement of proliferating fibroblasts into the newly open wound contributes to the early stages of wound closure (Nedelec et al. 2000). Clearly, wound contraction is a fibroblast-driven event that is acutely sensitive to fluctuations in the fibroblast-dependent cellular and extracellular matrix. Although vital to wound closure, wound contraction and shrinkage of the healed scar can lead to scar contractures, which may cause significant functional and cosmetic morbidity. The histopathological evaluation revealed equal collagen and fibroblast accumulation in both groups, but in the experimental wounds this was more orderly arranged (Figs. 2 and 3).
From a microbiological perspective, the total number of isolated microorganisms from the treatment group on day 21 was 1 ± 0.6 × 104/cm2, which was significantly lower than its equivalent in the control group (1 ± 0.5 × 105/cm2) (p < 0.05). It has been reported that the yarrow plant has a broad-spectrum antibacterial properties against both microorganisms and fungi (Barel et al. 1991). It is believed that special bioactive components and essential oils from the yarrow plant are responsible for its antimicrobial properties (Aburjai and Hudaib 2006). The bioactive component of the yarrow plant is mainly thought to be flavonoide compounds such as rutin, apigenin, luteolin, and acacetin (Aljancic et al. 1999). A naturally occurring terpene alcohol called linalool found in the yarrow plant is also believed to have an inhibitory effect on 17 and 10 microbial and fungal strains, respectively (Pattnaik et al. 1997).
Considering the positive clinical and microbiologic results of this study regarding the application of an alcoholic extract of yarrow on burn wounds in decreasing the microorganism burden and improving of the healing process and comparing them with similar studies, one can consider the yarrow plant’s efficacy in treating wounds. This study is the first to demonstrate this efficacy on experimental burn wounds rather than surgical wounds. Taking into account its desired actions, world-wide availability of the plant, and uncomplicated preparation procedure, the yarrow plant could be considered as an alternative or complementary medicine in handling wounds. Although the majority of studies report no major side effects or poisoning related to the yarrow plant, some reports of an allergic rash and light hypersensitivity with repeated usage at high doses have been documented (Foster S and Duke 1990). Therefore, additional studies and clinical trials are imperative before the plant’s extract or any of its preparations find their way into the mass market.
References
Aburjai T, Hudaib M (2006) Antiplatelet, antibacterial and antifungal activities of Achillea falcata extracts and evaluation of volatile oil composition. Pharm Manag 2:191–198
Aljancic I, Vajs V, Menkovic N (1999) Flavones and sesquiterpene lactones from Achillea millefolium: antimicrobial activity. J Nat Prod 62:909–911
Arora D, Kaur J (1999) Antimicrobial activity of spices. Int J Antimicrob Agents 12:257–262
Barel S, Segal R, Yashphe J (1991) The antimicrobial activity of the essential oil from Achillea fragrantissima. J Ethnopharmacol 33:187–191
Blumenthal M, Busse M, Goldberg A (1998) Therapeutic guide to herbal medicines. The American Botanical Council, Austin, pp 65–66
Bown D (1995) Encyclopaedia of herbs and their uses. Dorling Kindersley, London, pp 101–105
Bradley P (1992) British herbal compendium. British Herbal Medicine Association, Dorset, pp 227–229
Brans A, Dutrieux D, Hoekstra MJ (1994) Histopathological evaluation of scalds and contact burns in the pig model. Burns 20:548–551
Chevallier A (1996) The encyclopedia of medicinal plants. Dorling Kindersley, London, pp 102–105
Dalsenter PR, Cavalcanti AM, Andrade AJ, Araujo SL, Marques MC (2004) Reproductive evaluation of aqueous crude extract of Achillea millefolium L. (Asteraceae) in Wistar rats. Reprod Toxicol 18:819–823
Edwards-Jones V, Dawson MM, Childs CA (2000) Survey into TSS in UK Burns Units. Burns 26:323–333
Foster S, Duke JA (1990) A field guide to medicinal plants Eastern and central north of America. Houghton Mifflin, Chicago, pp 225–227
Hausbn BM, Bheuer J, Weglewski J, Rucker G (1991) α-Peroxyachifolid and other new sensitizing sesquiterpene lactones from yarrow (Achillea millefolium L., Compositae). Contact Dermat 24:274–280
Hoffmann D (1990) The new holistic herbal. Element, Dorset, pp 94–98
Kreutterbuch P (1960) Dioscorides. Verlag Konrad Kölbl, München, pp 87–88
Lans C, Turner N, Khan T, Brauer G, Boepple W (2007) Ethnoveterinary medicines used for ruminants in British Columbia. J Ethnobiol Ethnomedicine 3:11
Lawrence JC (1994) Burn bacteriology during the last 50 years. Burns 18:23–29
Lemmens GR, Marchart E, Rawnduzi P, Engel N, Benedek B, Kopp B (2006) Investigation of the spasmolytic activity of the flavonoid fraction of Achillea millefolium on isolated guinea-pig ilea. Arzneimittelforschung 56:582–588
Mills S (1994) The complete guide to modern herbalism. Thorsons, Great Britain, pp 55–59
Moerman D (1998) Native American ethnobotany. Timber, Oregon, pp 90–98
Moore M (1993) Yarrow as a natural remedy. In: Medicinal plants of the Mountain West. Red Crane books, Santa Fe, New Mexico, pp 272–275
Nedelec B, Ghahary A, Scott PG, Tredget EE (2000) Control of wound contraction: basic and clinical features. Hand Clin 16:289–302
Pattnaik S, Subramanyam VR, Bapaji M (1997) Antibacterial and antifungal activity of ten essential oils in vitro. Microbios 89:39–46
Perry LM (1980) Medicinal plants of East and Southeast Asia. The MIT Press, Cambridge, pp 110–113
Richey KJ, Engrav LH, Pavlin EG, Murray MJ, Gottlieb JR, Walkinshaw MD (1989) Topical growth factors and wound contraction in the rat, I: literature review and definition of the rat model. Ann Plast Surg 23:159–165
Saeidnia S, Gohari AR, Yassa N (2005) Composition of the volatile oil of Achillea Conferta from Iran. Daru 13:34–36
Singh YN, Blumenthal M (1998) Kava culture, then & now. Herbs Health 2:56–60
Subrahmanyam M, Sahapure AG, Nagane NS, Bhagwat VR, Ganu JV (2001) Effects of topical application of honey on burn wound healing. Ann Burns Fire Disasters 16(3):15–16
Taylor A, Francis M (2001) Final report on the safety assessment of yarrow (Achillea Millefolium) extract. Int J Toxicol 20:79–84
Acknowledgment
This study was supported financially by the Research Council of Veterinary College, Urmia University, for which the authors are most grateful.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jalali, F.S.S., Tajik, H. & Hadian, M. Efficacy of topical application of alcoholic extract of yarrow in the healing process of experimental burn wounds in rabbit. Comp Clin Pathol 21, 177–181 (2012). https://doi.org/10.1007/s00580-010-1081-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-010-1081-7