Abstract.
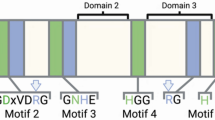
Protein-tyrosine dephosphorylation is a major mechanism in cellular regulation. A large number of protein-tyrosine phosphatases is known from Eukarya, and more recently bacterial homologues have also been identified. By employing conserved sequence patterns from both eukaryotic and bacterial protein-tyrosine phosphatases, we have identified three homologous sequences in two of the four complete archaeal genomes. Two hypothetical open reading frames in the genome of Methanococcus jannaschii (MJ0215 and MJECL20) and one in the genome of Pyrococcus horikoshii (PH1732) clearly bear all the conserved residues of this family. No homologues were found in the genomes of Archaeoglobus fulgidus and Methanobacterium thermoautotrophicum. This is the first report of protein-tyrosine phosphatase sequences in Archaea.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 29 April 1998 / Accepted: 27 November 1998
Rights and permissions
About this article
Cite this article
Stravopodis, D., Kyrpides, N. Identification of Protein-Tyrosine Phosphatases in Archaea. J Mol Evol 48, 625–627 (1999). https://doi.org/10.1007/PL00013155
Issue Date:
DOI: https://doi.org/10.1007/PL00013155