Abstract
Objective
To clarify the retention mechanism of a PET imaging agent Cu-pyruvaldehyde-bis(N4-methylthiosemicarbazone) (Cu-62-PTSM) in tumor cells, reductive metabolism of non-radioactive Cu-PTSM in five cultured tumor cell lines, a tumor specimen and non-tumor tissuesin vitro was evaluated by electron spin resonance spectrometry (ESR).
Results
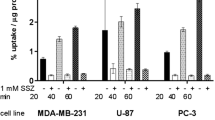
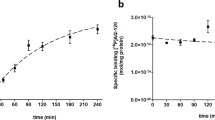
In the brain, mitochondrial electron transport enzyme reduced Cu-PTSM specifically. On the other hand, Cu-PTSM was not reduced in tumor mitochondria. The mitochondrial electron transport enzyme in tumor cells was not damaged, but NADH was considered to be depleted. In compensation for that, the tumor cells acquired complementary reduction activity in the microsome/cytosol. The reduction was enzymatic and NADH-dependent, possibly similar to the activation mechanism of bioreductive anticancer drugs.
Conclusion
Cu-PTSM and its derivatives are considered to be used as a marker for microsome/cytosol redox ability in PET oncology, although the physiological role of the redox enzyme system in tumor cells has not been clarified. The change in electron (NADH) flow in tumor cells might be a mechanism supporting aerobic glycolysis in tumor cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fujibayashi Y, Matsumoto K, Yonekura Y, Konishi J, Yokoyama A. A new zinc-62/copper-62 generator as a copper-62 source for PET radiopharmaceuticals.J Nucl Med 30: 1838–1842, 1989.
Fujibayashi Y, Wada K, Taniuchi H, Yonekura Y, Konishi J, Yokoyama A. Mitochondria-selective reduction of62Cu-pyruvaldehyde-bis(N4-methylthiosemicarbazone) (62Cu-PTSM) in the murine brain; a novel radiopharmaceutical for brain positron emission tomography (PET) imaging.Biol Pharm Bull 16: 146–149, 1993.
Matsumoto K, Fujibayashi Y, Yonekura Y, Wada K, Takemura Y, Konishi J, et al. Application of the new zinc-62/copper-62 generator: an effective labeling method for62Cu-PTSM.Nucl Med Biol 19: 39–44, 1992.
Okazawa H, Yonekura Y, Fujibayashi Y, Nishizawa S, Magata Y, Ishizu K, et al. Clinical application and quantitative evaluation of generator-produced copper-62-PTSM as a brain perfusion tracer for PET.J Nucl Med 35: 1910–1915, 1994.
Wada K, Fujibayashi Y, Taniuchi H, Tajima N, Tamaki N, Konishi J, et al. Effects of ischemia-reperfusion injury on myocardial single pass extraction and retention of Cu-PTSM in perfused rat hearts: comparison with201T1 and14C-iodoantipyrine.Nucl Med Biol 21: 613–617, 1994.
Taniuchi H, Fujibayashi Y, Yonekura Y, Konishi J, Yokoyama A. Hyperfixation of copper-62-PTSM in rat brain after transient global ischemia.J Nucl Med 38: 1130–1134, 1997.
Mathias CJ, Welch MJ, Perry DJ, McGuire AH, Zhu X, Connett JM, et al. Investigation of copper-PTSM as a PET tracer for tumor blood flow.Int J Rad Appl Instrum B 18: 807–811, 1991.
Mathias CJ, Green MA, Morrison WB, Knapp DW. Evaluation of Cu-PTSM as a tracer of tumor perfusion: comparison with labeled microspheres in spontaneous canine neoplasms. Evaluation of Cu-PTSM as a tracer of tumor perfusion: comparison with labeled microspheres in spontaneous canine neoplasms.Nucl Med Biol 21: 83–87, 1994.
Fujibayashi Y, Taniuchi H, Wada K, Yonekura Y, Konishi J, Yokoyama A. Differential mechanism of retention of Cu-pyruvaldehyde-bis(N4-methylthiosemicarbazone) (Cu-PTSM) by brain and tumor: a novel radiopharmaceutical for positron emission tomography imaging.Ann Nucl Med 9: 1–5, 1995.
Shibamoto Y, Takahashi M, Ono K, Komuro C, Abe M. Recovery from potentially lethal damage after X-irradiation in three experimental tumors in mice.Jpn J Cancer Res 76: 278–283, 1985.
Menkel DT, Petering DH. Initial reaction of 3-ethoxy-2-oxobytyraldehyde bis(thiosemicarbazonato)coper(II) with Ehrlich ascites tumor cells.Cancer Res 38: 117–123, 1978.
Voss DO, Camplleo AP, Bacila M. The respiratory chain and the oxidative phosphorylation of rat brain mitochondria.Biochem Biophys Res Commun 4: 48–51, 1961.
Pennington RJ. Biochemistry of dystrophic muscle; mitochondrial succinate-tetrazolium reductase and adenosine triphosphatase.Biochem J 80: 649–654, 1961.
Omura T, Takasue S. A new method for simultaneous purification of cytochrome b5 and NADPH-cytochrome c reductase from rat liver microsomes.J Biochem 67: 249–257, 1970.
Kornberg A. Lactate dehydrogenase of muscle. In:Medhods in Enzymology, vol. 1, pp. 441–443, 1955.
Warren LE, Horner SM, Hatfield WE. Chemistry of alpha-diketone-bis(thiosemicarbazone)copper(II) complexes.J Amer Chem Soc 94: 6392–6396, 1972.
Dietzen DJ, Davis EJ. Oxidation of pyruvate, malate, citrate, and cytosolic reducing equivalents by AS-30D hepatoma mitochondria.Arch Biochem Biophys 305: 91–102, 1993.
Pedersen PL. Tumor mitochondria and the bioenergetics of cancer cells.Prog Exp Tumor Rev 22: 190–274, 1978.
Eigenbrodt E, Fister P, Reinacher M. New perspectives on carbohydrate metabolism in tumor cells. In:Regulation of Carbohydrate Metabolism, vol. 2. CRC Press: Oxford, pp. 141–179, 1986.
Chance H, Williamson JR, Jamieson D, Schoener H. Properties and kinetics of reduced pyridine nucleotide fluorescence of the isolated andin vivo rat heart.Biochem Zeit 341: 357–377, 1965.
Kogure K, Busto R, Schwartzman RJ, Scheinberg P. The dissociation of cerebral blood flow, metabolism and function in the early stages of developing cerebral infarction.Annals Neurol 8: 278–290, 1979.
Waki A, Fujibayashi Y, Yonekura Y, Sadato N, Ishii Y, Yokoyama A. Reassessment of FDG uptake in tumor cells: high FDG uptake as a reflection of oxygen-independent glycolysis dominant energy production.Nucl Med Biol 24: 665–670, 1997.
Shawartz JP, Passonneau JV, Johnson GS, Pastan I. The effect of growth conditions on NAD+ and NADH concentrations and the NAD+: NADH ratio in normal and transformed fibroblasts.J Biol Chem 249: 4138–4143, 1974.
Darville MI, Antoine IV, Mertens-Strijthagen JR, Dupriez VJ, Roussenu GG. An E2F-dependent late-serum-response promoter in a gene that controls glycolysis.Oncogene 11: 1509–1517, 1995.
Fitzsimmons SA, Workman P, Grever M, Pauli K, Camalier R, Lewis AD. Reductase enzyme expression across the National Cancer Institute Tumor Cell Line Panel: correlation with sensitivity to mitomycin C and EO9.J Natl Cancer Inst 88: 259–269, 1996.
Walton MI, Workman P. Enzymology of the reductive bioactivation of SR4233.Biochem Phamacol 39: 1735–1742, 1990.
Workman P, Stratford IJ. The experimental development of bioreductive drugs and their role in cancer therapy.Cancer Metast Rev 12: 73–82, 1993.
Petering DH. Carcinostatic copper complexes. In:Metal Ions in Biological Systems, vol. 11. Marcel Dekker, Inc.: New York, pp. 198–229, 1980.
Kilkuskie RE, Macdonald TL, Hecht SM. Bleomycin may be activated for DNA cleavage by NADPH-cytochrome P-450 reductase.Biochem 23: 6165–6171, 1984.
Biaglow JE, Cerniglia G, Tuttle S, Bakanauskas V, Stevens C, McKenna G. Effect of oncogene transformation of rat embryo cells on cellular oxygen consumption and glycolysis.Biochem Biophys Res Commun 235: 739–742, 1997.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shibuya, K., Fujibayashi, Y., Yoshimi, E. et al. Cytosolic/microsomal redox pathway: a reductive retention mechanism of a PET-oncology tracer, Cu-pyruvaldehyde-bis(N 4-methylthiosemicarbazone) (Cu-PTSM). Ann Nucl Med 13, 287–292 (1999). https://doi.org/10.1007/BF03164865
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03164865