Abstract
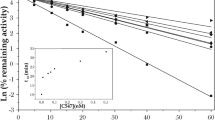
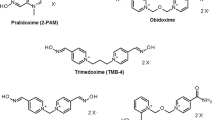
Oximes in combination with atropine, are an integral part of the treatment of acute intoxications with organophosphorus insecticides or with the nerve agents such as tabun, sarin, soman, cyclosarin or VX. Organophosphorus compounds are extremely potent inhibitors of the enzyme acetylcholinesterase (AChE, 3.1.1.7). The pharmacological action of oximes is multiple: they are able to reactivate the inhibited AChE, but they affect acetylcholine release in peripheral and central cholinergic synapses, allosterically modulate the muscarinic receptors in peripheral and central synapses and influence the nicotinic receptor associated ion-channels. In our study, we have determined the acute toxicity of different structures of oximes after intramuscular application in mice. The acute toxicity of oximes is crucial for the assesment of a dose applied as a treatment for organophosphorus intoxications. We have tested 7 oximes of different structures (HS-6, K033, BI-6, MMB-4, K048, HI-6 and obidoxime) and during experiment we have observed the intoxication process including typical signs of intoxication, and times of death. K033 was the most toxic oxime with LD50 only 48 mg/kg, while the least toxic oxime - HI-6 has value of LD50 671 mg/kg. All the oximes tested were of bispyridinium type with different length or shape of the connecting chain and positions of oxime groups at the pyridinium rings. All these structure features play important role in biological activity of these compounds performed by their acute toxicity as well as by their reactivation potency.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- HS-6:
-
[ 1 -(2-hydroxyiminomethylpyridinium)-3-(3-car-bamoylpyridinium)-2-oxa-propane dichloride]
- BI-6:
-
[ 1 -(2-hydroxyiminomethylpyridinium)-4-(4-car-bamoylpyridinium)-but-2-ene dibromide]
- K033:
-
[ 1,2-bis(2-hydroxyiminomethylpyridinium)but-ane dibromide]
- MMB-4:
-
[methoxime; 1,1-bis(4-hydroxyiminomethylpyridinium)-methane dibromide]
- K048:
-
[ 1 -(4-hydroxyiminomethylpyridinium)-4-(4-car-bamoylpyridinium)-butane dibromide]
- HI-6:
-
[ 1 -(2-hydroxyiminomethylpyridinium)-3-(4-car-bamoylpyridinium)-2-oxa-propane dichloride]
References
Balali-Mood, M and M Shariat (1998) Treatment of organophosphate poisoning. Experience of nerve agents and acute pesticide poisoning on the effects of oximes.J. Physiol. Paris 92, 375–378.
Bielavsky J, J Vachek and F Ornst (1972) Preparation and properties of cholinesterase reactivators of bis-quaternary monopyridinaldoxime type.Coll. Czech. Chem. Commun. 37, 1044–1048.
Bielavsky J, J Kassa, I Elsnerova and L Dejmek (1998) Cholinesterase reactivators derived from pyridine-2-carbaldoxime.Coll. Czech. Chem. Commun. 63, 199–204.
Dube SN, D Kumar, J Acharya, DK Dubey, Purnanand and AK Sikder (2000) Antidotal efficacy of pyridinium oximes and cholineacetyltransferase inhibitors against organophosphorus intoxication in rodents.Indian J. Physiol. Pharmacol. 44, 50–56.
Eyer P, A Kawan and B Ladstetter (1987) Formation of cyanide after i.v. administration of the oxime HI 6 to dogs.Arch. Toxicol. 61, 63–69.
Faff J and W Bak (1978) Increased sensitivity to obidoxime induced by fluostigmine in the rat.Toxicol. Appl. Pharmacol. 46, 429–433.
Kassa J (2006) The influence of oxime and anticholinergic drug selection on the potency of antidotal treatment to counteract acute toxic effects of tabun in mice.Neurotoxicity Res. 9, 59–62.
Kuca K and J Patocka (2004) Reactivation of cyclosarin-inhibited rat brain acetylcholinesterase by pyridinium-oximes.J. Enz. Inhib. Med. Chem. 19, 39–43.
Kuca K, J Bielavsky, J Cabal and J Kassa (2003) Synthesis of a new reactivator of tabun-inhibited acetylcholinesterase.Bioorg. Med. Chem. Lett. 13, 3545–3547.
Kuca K, J Cabal, J Patocka and J Kassa (2004a) Synthesis of Bisquaternary Symmetric - X, δ-bis(2-hydroxyiminomethylpyridinium) alkane dibromides and their reactivation of cyclosarininhibited acetylcholinesterase.Lett. Org. Chem. 1, 84–86.
Kuca K, J Patocka, J Cabal and D Jun (2004b) Reactivation of organophosphate-inhibited acethyclhoinersterase by quaternary pyridinium addoximes.Neurotoxicity Res. 6, 565–570.
Lüllmann H, K Mohr and M Wehling (2002) Dose-effect relationships, In:Pharmacology and Toxicology (Grada Publishing: Prague, Czech Republic), pp 30–33.
Marrs TC (1991) Toxicology of oximes used in the treatment of organophosphate poisoning.Adverse Drug React. Acute Pois. Rev. 10, 61–73.
Nocentini S, G Moreno, PX Petit, M Guggiari, C Salet, P Demerseman and G Dodin (1997) Induction of mitochondrial dysfunction and apoptosis in HeLa cells by bis-pyridinium oximes, a newly synthesized family of lipophilic biscations.Biochem. Pharmacol. 53, 1543–52.
Roth Z, M Josifko and V Trcka (1962) Analysis of biological data, In:Statistical Methods in Experimental Medicine (Statni zdravotnicke nakladatelstvi:Prague, Czech Republic) pp 325–335, in Czech.
Scott RJ (1986) Repeated asystole following PAM in organophosphate self-poisoning.Anaesth Intensive Care 14, 458–60.
Sidell FR (1992) Clinical considerations in nerve agent intoxication, In:Chemical Warfare Agents (Somani SM, Eds.)(Academic Press, San Diego), pp 155–194.
Sidell FR (1997) Nerve agents, In:Medical Aspects of Chemical and Biological Warfare (Zajtchuk R and RF Bellamy, Eds.) (Office of The Surgeon General at TMM Publications, Washington, USA), pp 129–179.
Simeon-Rudolf V, E Reiner, M Skrinjaric-Spoljar, B Radic, A Lucic, I Primozic and S Tomic (1998) Quinuclidinium-imidazolium compounds: synthesis, mode of interaction with acetylcholinesterase and effect upon Soman intoxicated mice.Arch. Toxicol. 72, 289–295.
Tallarida Rand R Murray (1987)Manual of Pharmacological Calculation with Computer Programs, 2nd Ed. (Springer-Verlag: New York), pp 145–160.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bartosova, L., Kuca, K., Kunesova, G. et al. The Acute Toxicity of Acetylcholinesterase Reactivators in Mice in Relation to Their Structure. neurotox res 9, 291–296 (2006). https://doi.org/10.1007/BF03033319
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF03033319