Abstract
Purpose
To evaluate the comparative preemptive effects of gabapentin and tramadol on postoperative pain and fentanyl requirement in laparoscopic cholecystectomy.
Methods
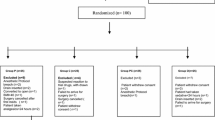
Four hundred fifty-nine ASA I and II patients were randomly assigned to receive 300 mg gabapentin, 100 mg tramadol or placebo in a double-blind manner two hours before laparoscopic cholecystectomy under general anesthesia. Postoperatively, patients’ pain scores were recorded on a visual analogue scale every two hours for the initial 12 hr and thereafter every three hours for the next 12 hr. Patients received fentanyl 2μg·kg−1 intravenously on demand. The total fentanyl consumption for each patient was recorded.
Results
Patients in the gabapentin group had significantly lower pain scores at all time intervals (2.65 ± 3.00, 1.99 ± 1.48, 1.40 ± 0.95, 0.65 ± 0.61) in comparison to tramadol (2.97 ± 2.35, 2.37 ± 1.45, 1.89 ± 1.16, 0.87 ± 0.50) and placebo (5.53 ± 2.22, 3.33 ± 1.37, 2.41 ± 1.19, 1.19 ± 0.56). Significantly less fentanyl was consumed in the gabapentin group (221.16 ± 52.39 μg) than in the tramadol (269.60 ± 44.17 μg) and placebo groups (355.86 ± 42.04 μg;P < 0.05). Sedation (33.98%), nausea/retching/vomiting (24.8%) were the commonest side effects in the gabapentin group whereas respiratory depression (3.9%) was the commonest in the tramadol group and vertigo (7.8%) in the placebo group.
Conclusion
Preemptive use of gabapentin significantly decreases postoperative pain and rescue analgesic requirement in laparoscopic cholecystectomy.
Résumé
Objectif
Évaluer et comparer les effets préventifs de la gabapentine et du tramadol sur la douleur postopératoire et les besoins de fentanyl lors d’une cholécystectomie laparoscopique.
Méthode
Quatre cent cinquante-neuf patients d’état physique ASA I et II ont été répartis au hasard et ont reçu 300 mg de gabapentine, 100 mg de tramadol ou un placebo, en double aveugle, deux heures avant la cholécystectomie laparoscopique sous anesthésie générale. Après l’opération, les scores de douleur ont été notés sur l’échelle visuelle analogique toutes les deux heures pendant les 12 premières heures et toutes les trois heures pendant les 12 h suivantes. Les patients ont reçu 2 μg·kg−1 de fentanyl intraveineux sur demande et la consommation totale a été notée pour chacun.
Résultats
Les patients du groupe gabapentine ont présenté des scores de douleur significativement plus bas pour tous les intervalles de mesures (2,65 ± 3,00; 1,99 ± 1,48; 1,40 ± 0,95; 0,65 ± 0,61) que ceux du groupe tramadol (2,97 ± 2,35; 2,37 ± 1,45; 1,89 ± 1,16; 0,87 ± 0,50) ou placebo (5,53 ± 2,22; 3,33 ± 1,37; 2,41 ± 1,19; 1,19 ± 0,56). La demande de fentanyl a été significativement plus basse avec la gabapentine (221,16 ± 52,39 μg) qu’avec le tramadol (269,60 ± 44,17 μg) ou le placebo (355,86 ± 42,04 μg; P < 0,05). La sédation (33,98 %), les nausées/haut-lec∁ur/vomissements (24,8 %) ont été les effets négatifs les plus fréquents avec la gabapentine tandis que la dépression respiratoire (3,9 %) a été plus fréquente avec le tramadol et le vertige (7,8 %) avec le placebo.
Conclusion
L’usage préventif de gabapentine diminue significativement la douleur postopératoire et la demande d’analgésique de secours lors de la cholécystectomie laparoscopique.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wu CT, Yu JC, Yeh CC, et al. Preincisional dextromethorphan treatment decreases postoperative pain and opioid requirement after laparoscopic cholecystectomy. Anesth Analg 1999; 88: 1331–4.
Woolf CJ, Chong MS. Preemptive analgesia-treating postoperative pain by preventing the establishment of central sensitization. Anesth Analg 1993; 77: 362–79.
Chiaretti A, Viola L, Pietrini D, et al. Preemptive analgesia with tramadol and fentanyl in pediatric neurosurgery. Childs Nerv Syst 2000; 16: 93–9.
Naguib M, Seraj M, Attia M, Samarkandi AH, Seet M, Jaroudi R. Perioperative antinociceptive effects of tramadol. A prospective, randomized, double-blind comparison with morphine. Can J Anaesth 1998; 45: 1168–75.
Fassoulaki A, Patris K, Sarantopoulos C, Hogan Q. The analgesic effect of gabapentin and mexilentine after breast surgery for cancer. Anesth Analg 2002; 95: 985–91.
Pandey CK, Bose N, Garg G, et al. Gabapentin for the treatment of pain in Guillain-Barré syndrome: a double-blinded, placebo-controlled, crossover study. Anesth Analg 2002; 95: 1719–23.
Ramsay MA, Savege TM, Simpson BR, Goodwin R. Controlled sedation with alphaxalone-alphadolone. BMJ 1974; 2: 656–9.
Goa KL, Sorkin EM. Gabapentin. A review of its pharmacological properties and clinical potential in epilepsy. Drugs 1993; 46: 409–27.
Taylor CP, Gee NS, Su TZ, et al. A summary of mechanistic hypotheses of gabapentin pharmacology. Epilepsy Res 1998; 29: 233–49.
Gee NS, Brown JP, Dissanayake VU, Offord J, Thurlow R, Woodruff GN. The novel anticonvulsant drug, gabapentin (neurontin), binds to the α2δsubunit of a calcium channel. J Biol Chem 1996; 271: 5768–76.
Lee CR, McTavish D, Sorkin EM. Tramadol. A preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in acute and chronic pain states. Drugs 1993; 46: 313–40.
Dirks J, Fredensborg BB, Christensen D, Fomsgaard JS, Flyger H, Dahl JB. A randomized study of the effects of single-dose gabapentin versus placebo on postoperative pain and morphine consumption after mastectomy. Anesthesiology 2002; 97: 560–4.
Eckhardt K, Ammon S, Hofmann U, Riebe A, Gugeler N, Mikus G. Gabapentin enhances the analgesic effect of morphine in healthy volunteers. Anesth Analg 2000; 91: 185–91.
Werner MU, Perkins FM, Holte K, Pedersen JL, Kehlet H. Effects of gabapentin in acute inflammatory pain in humans. Reg Anesth Pain Med 2001; 26: 322–8.
Mao J, Chen LL. Gabapentin in pain management. Anesth Analg 2000; 91: 680–7.
Abdi S, Lee DH, Chung JM. The anti-allodynic effects of amitriptyline, gabapentin, and lidocaine in a rat model of neuropathic pain. Anesth Analg 1998; 87: 1360–6.
Jun JH, Yaksh TL. The effect of intrathecal gabapentin and 3-isobutyl γ-aminobutyric acid on the hyperalgesia observed after thermal injury in the rat. Anesth Analg 1998; 86: 348–54.
Field MJ, Holloman EF, McCleary S, Hughes J, Singh L. Evaluation of gabapentin and S-(+)-3-isobutylgaba in a rat model of postoperative pain. J Pharmacol Exp Ther 1997; 282: 1242–6.
Dirks J, Petersen KL, Rowbotham MC, Dahl JB. Gabapentin suppresses cutaneous hyperalgesia following heat-capsaicin sensitization. Anesthesiology 2002; 97: 102–7.
Nicholson B. Gabapentin use in neuropathic pain syndromes. Acta Neurol Scand 2000; 101: 359–71.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pandey, C.K., Priye, S., Singh, S. et al. Preemptive use of gabapentin significantly decreases postoperative pain and rescue analgesic requirements in laparoscopic cholecystectomy. Can J Anesth 51, 358–363 (2004). https://doi.org/10.1007/BF03018240
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03018240