Abstract
Purpose
To report increased infusion requirements of cis-atracunum during prolonged infusion (six weeks) to provide neuromuscular blockade in a child during prolonged mechanical ventilation. Despite a previous study in adult patients which demonstrated no increase in infusion requirements over five days, we noted a considerable increase over six weeks.
Clinical features
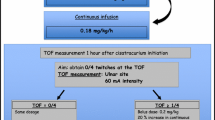
A seven month old infant required prolonged mechanical ventilation and neuromuscular j blockade following an episode of multi-system organ failure from pseudomembranous colitis. The infusion of cisatracunum was adjusted according to the train-of-four response obtained with a peripheral nerve stimulator using standard train-of-four monitoring. Initial infusion requirements which were 2.8 μg · kg−1 min−1 on day # 1 increased to 22.3 μg · kg−1 min−1 on day #40.
Conclusion
Increased infusion requirements were necessary during the prolonged administration of cisatracurium to a critically ill infant. Titration of the dose based on monitoring with a peripheral nerve stimulator is recommended.
Résumé
Objectif
Rapporter l’augmentation des besoins de cis-atracurium pendant une perfusion de longue durée (six semaines) utilisée pour curariser un enfant pendant la ventilation mécanique. Malgré une étude antérieure qui démontrait chez les adultes une augmentation importante des besoins survenant en cinq jours, ici, l’importante augmentation a été constatée en six semaines.
Eléments cliniques
Un enfant de sept mois a eu besoin de ventilation mécanique prolongée avec curarisation à la suite d’une défaillance multiviscérale causée par une colite pseudomembraneuse. La perfusion de cisatracurium était réglée selon la réponse au train-de-quatre monitorée avec un stimulateur nerveux périphérique. Les besoins initiaux de la perfusion qui étaient de 2,8 μg · kg−1 min−1 sont passés à 22 μg · kg−1 min−1 à la quarantième journée.
Conclusion
II a été nécessaire d’augmenter une perfusion de cis-atracurium de longue durée chez un enfant gravement malade. II est recommandé d’ajuster la dose de curare avec un moniteur périphérique pour le bloc neuromusculaire.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sharpe MD. The use of muscle relaxants in the intensive care unit. Can J Anaesth 1992; 39: 949–62.
Magorian T, Wood P, Caldwell J, et al. The pharmacokinetics and neuromuscular effects of rocuronium bromide in patients with liver disease. Anesth Analg 1995; 80: 754–9.
Blutstein LS, Stinson LW Jr, Lennon RL, Quessy SN, Wilson RM. Evaluation of cisatracurium, a new neuro-muscular blocking agent, for tracheal intubation. Can J Anaesth 1996; 43: 925–31.
Prielipp RC, Coursin DB, Scuderi PE, et al. Comparison of the infusion requirements and recovery profiles of vecuronium and cisatracurium 51W89 in intensive care unit patients. Anesth Analg 1995; 81: 3–12.
Tobias JD. Continuous infusion of rocuronium in a paediatric intensive care unit. Can J Anaesth 1996; 43: 353–7.
Tobias JD, Lynch A, McDuffee A, Garrett JS. Pancuronium infusion for neuromuscular block in children in the Pediatric Intensive Care Unit. Anesth Analg 1995; 81: 13–6.
Kushimo TO, Darowski MJ, Morris P, Hollis S, Meakin G. Dose requirements of atracurium in paediatric intensive care patients. Br J Anaesth 1991; 67: 781–3.
Dodson BA, Kelly BJ, Braswell LM, Cohen NH. Changes in acetylcholine receptor number in muscle from critically ill patients receiving muscle relaxants: an investigation of the molecular mechanism of prolonged paralysis. Crit Care Med 1995; 23: 815–21.
Martyn JAJ, White DA, Gronert GA, Joffe RS, Ward JM. Up-and-down regulation of skeletal muscle acetylcholine receptors. Effects on neuromuscular blockers. Anesthesiology 1992; 76: 822–43.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tobias, J.D. Increased cis-atracurium requirements during prolonged administration to a child. Can J Anesth 44, 82–84 (1997). https://doi.org/10.1007/BF03014329
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03014329