Abstract
Purpose
To compare low vs high dose propofol and isoflurane on red cell RBC antioxidant capacity in patients during aortocoronary bypass surgery (ACBP).
Methods
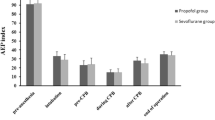
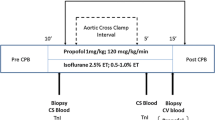
Twenty-one patients, for ACBR were anesthetized with sufentanil 0.5–10 μg·kg−1 and isoflurane 0–2%; ISO = control; n=7), or sufentanil 0.3 μg·kg−1, propofol 1–2.5 μg·kg−1 bolus then 100μg·kg−1 before, and 50 μg·kg−1·min−1 during CPB (LO; n=7), or sufentanil 0.3 μg·kg−1, propofol 2–2.5 μg·kg−1 bolus then 200 μg·kg−1·min−1 (HI; n=7). Venous blood was drawn pre-and post-induction, after 30 min CPB, 5, 10, and 30 min of reperfusion, and 120 min post-CPB to measure red cell antioxidant capacity (malondialdehyde (MDA) production in response to oxidative challenge with t-butyl hydrogen peroxide) and plasma propofol concentration. Preinduction blood samples were analyzed for antioxidant effects of nitrates on red cells. The tBHP concentration response curves for RBC MDA in ISO, LO and HI were determined.
Results
Preoperative nitrate therapy did not effect RBC MDA production. Perioperative RBC MDA production was similar in ISO and LO groups. Sustained intraoperative decrease in RBC MDA was seen with propofol 8.0 ± 2.4 − 11.8 ± 4.5 μg·ml−1 in HI (P < 0.05- 0.0001). MDA production vs log plasma propofol concentration was linear in HI dose.
Conclusions
During CPB, RBC antioxidant capacity is enhanced and maintained with HI dose propofol. Propofol, at this dose, may prove useful in protecting against cardiopulmonary ischemia-reperfusion injury associated with ACBR
Résumé
Objectif
Comparer une faible dose (LO) vs une forte dose (HI) de propofol et d’isoflurane sur la capacité antioxydante des globules rouges (GR) lors d’un pontage aortocoronarien (PAC).
Méthode
Lors d’un PAC, 21 patients ont reçu une anesthésie avec du sufentanil 0,5–10 μg·ml−1 et de l’isoflurane 0–2 %; (ISO = témoin, n = 7) ou du sufentanil 0,3 μg·kg−1 un bolus de propofol 1–2,5 μg·kg−1 suivi d’une perfusion de 100 μg·kg−1·min−1 avant le PAC et de 50 μg·kg−1·min−1 pendant le PAC (LO, n = 7), ou du sufentanil 0,3 μg·kg−1, un bolus de propofol 2–2,5 μg·kg−1 et une perfusion de 200 μg·kg−1·min−1 (HI, n = 7). Le sang veineux a été prélevé avant et après l’induction, 30 min après le PAC, à 5, 10 et 30 min pendant la reperfusion et 120 min après la CEC afin de mesurer la capacité antioxydante des GR (production de dialdéhyde malonique DAM en réponse à la provocation oxydante avec du peroxyde d’hydrogène t-butyl PHtB) et la concentration plasmatique de propofol. Les échantillons de sang prélevés avant l’induction ont été analysés pour vérifier les effets antioxydants des nitrates sur les GR. Les courbes illustrant la réaction des GR au DAM chez les patients des groupes ISO, LO et HI ont été déterminées.
Résultats
La thérapie préopératoire aux nitrates n’a pas changé la capacité antioxydante des GR, donc la production de DAM a été semblable dans les groupes ISO et LO. Une baisse peropératoire de production de DAM a toutefois été observée avec 8,0 ± 2,4 – 11,8 ± 4.5 μg·ml−1 de propofol dans le groupe HI (P < 0,05 - 0,0001). La production de DAM vs le logarithme de la concentration plasmatique de propofol était linéaire dans le groupe HI.
Conclusion
Pendant la CEC, la capacité antioxydante des GR a été améliorée et maintenue par une forte dose de propofol. Administré selon cette dose, le propofol peut se révéler utile pour protéger des lésions cardio-pulmonaires liées à l’ischémie de reperfusion associée au PAC.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mangano DT. Biventricular function after myocardial revascularization in humans: deterioration and recovery patterns during the first 24 hours. Anesthesiology 1985; 62: 571–7.
Breisblatt WM, Stein KL, Wolfe CJ, et al. Acute myocardial dysfunction and recovery: a common occurrence after coronary bypass surgery. J Am Coll Cardiol 1990; 15: 1261–9.
Leung JM, O’Kelly BF, Mangano DT. Relationship of regional wall motion abnormalities to hemodynamic indices of myocardial oxygen supply and demand in patients undergoing CABG surgery. Anesthesiology 1990; 73: 802–14.
Weintraub WS, Jones EL, Craver J, Guyton R, Cohen C. Determinants of prolonged length of hospital stay after coronary bypass surgery. Circulation 1989; 80: 276–84.
Turnbull KW, Miyagashima RT, Gerein AN. Pulmonary complications and cardiopulmonary bypass: a clinical study in adults. Can Anaesth Soc J 1974; 21: 181–94.
Forman MB, Virmani R, Puett DW. Mechanisms and therapy of myocardial reperfusion injury. Circulation 1990; 81: IV69–78.
McCord JM. Oxygen-derived radicals: a link between reperfusion injury and inflammation. FASAB J 1987; 46: 2402–6.
Marijic J, Stowe DF, Turner LA, Kampine JP, Bosnjak ZJ. Differential protective effects of halothane and isoflurane against hypoxic and reoxygenation injury in the isolated guinea pig heart. Anesthesiology 1990; 73: 976–83.
Shayevitz JR, Johnson KJ, Knight PR. Halothane-oxidant interactions in theex vivo perfused rabbit lung. Fluid conductance and eicosanoid production. Anesthesiology 1993; 79: 129–38.
Murphy PG, Myers DS, Davies MJ, Webster NR, Jones JG. The antioxidant potential of propofol (2,6-diisopropylphenol). Br J Anaesth 1992; 68: 613–8.
Green TR, Bennet SR, Nelson VM. Specificity and properties of propofol as an antioxidant free radical scavenger. Toxicol Appl Pharmacol 1994; 129: 163–9.
Kokita N, Hara A. Propofol attenuates hydrogen peroxide-induced mechanical and metabolic derangements in the isolated rat heart. Anesthesiology 1996; 84: 117–27.
Kahraman S, Demiryürek AT. Propofol as a peroxynitrite scavenger. Anesth Analg 1997; 84: 1127–30.
Myles PS, Buckland MR, Weeks AM, et al. Hemodynamic effects, myocardial ischemia, and timing of tracheal extubation with propofol-based anesthesia for cardiac surgery. Anesth Analg 1997; 84: 12–9.
Ansley DM, Lee J-U, Godin DV, Garnett ME, Qayumi AK. Propofol enhances red cell antioxidant capacity in swine and humans. Can J Anaesth 1998; 45: 233–9.
Gilbert HS, Stump DD, Roth EF Jr. A method to correct for errors caused by generation of interfering compounds during erythrocyte lipid peroxidation. Ann Biochem 1984; 137: 282–6.
Shayevitz JR, Varani J, Ward PA, Knight PR. Halothane and isoflurane increase pulmonary artery endothelial cell sensitivity to oxidant-mediated injury. Anesthesiology 1991; 74: 1067–77.
Lal M, Schöneich C, Mnig J, Asmus K-D. Rate constants for the reactions of halogenated organic radicals. Int J Radiat Biol 1988; 54: 773–85.
Ansley DM. Ischaemia-reperfusion: time to increase our awareness during anaesthesia (Editorial). Can J Anaesth 1998; 45: 195–8.
Nielsen VG, Baird MS, McAdams ML, Freeman BA. Desflurane increases pulmonary alveolar-capillary membrane permeability after aortic occlusion-reperfusion in rabbits. Evidence of oxidant-mediated lung injury. Anesthesiology 1998; 88: 1524–34.
Webster NR. Opioids and the immune system (Editorial). Br J Anaesth 1998; 81: 835–6.
Qayumi AK, Godin DV, Jamieson WRE, Ko KM, Poostizadeh A. Correlation of red cell antioxidant status and heart-lung function in swine pretreated with allopurinol (a model of heart-lung transplantation). Transplantation 1993; 56: 37–43.
Coghlin JG, Flitter WD, Clutton SM, et al. lipid peroxidation and changes in vitamin E levels during coronary artery bypass grafting. J Thorac Cardiovasc Surg 1993; 106: 268–74.
Kim K-B, Chung HH, Kim MS, Rho JR. Changes in the antioxidative defensive system during open heart operations in humans. Ann Thorac Surg 1994; 58: 170–5.
Wohaieb SA, Godin DV. Starvation-related alterations in free radical tissue defence mechanisms in rats. Diabetes 1987; 36: 169–73.
Godin DV, Wohaieb SA, Garnett ME, Goumeniouk AD. Antioxidant enzyme alterations in experimental and clinical diabetes. Mol Cell Biochem 1988; 84: 223–31.
Mora, CT, Dudek C, Torjman MC, White PF. The effects of anesthetic technique on the hemodynamic response and recovery profile in coronary revascularization patients. Anesth Analg 1995; 81: 900–10.
Too KT, Tang ST, Lee J, et al. Intracoronary propofol attenuates myocardial but not coronary endothelial dysfunction after brief ischaemia and reperfusion in dogs. Br J Anaesth 1999; 82: 90–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ansley, D.M., Sun, J., Visser, W.A. et al. High dose propofol enhances red cell antioxidant capacity during CPB in humans. Can J Anesth 46, 641–648 (1999). https://doi.org/10.1007/BF03013951
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03013951