Abstract
Purpose
To identify risk factors for post-carotid endarterectomy (CEA) hematoma formation and establish the incidence of this complication at The Ottawa Hospital — Civic Campus (TOH-CC).
Methods
A chart review of all patients who underwent CEA at TOH-CC from January 1, 1996 to December 31,1997 was completed. Identified cases of post-CEA wound hematoma were entered into a case-control study using age and sex-matched controls from within the cohort. These matched pairs were assessed for 31 potential risk factors including demographic details, co-existing medical conditions, preoperative medications, intraoperative management, and postoperative parameters. Risk factors associated with post-CEA hematoma withP < 0.05 were entered into a backward step-wise logistic regression model for multivariate analysis.
Results
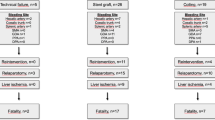
Charts from 249 patients were reviewed and 29 cases of post-carotid endarterectomy hematoma were identified (12% incidence). Six of the initial 31 potential risk factors emerged as univariate predictors of post-CEA hematoma formation (P < 0.05): general anesthesia, carotid shunt placement, intraoperative hypotension, nonreversal of heparin, neurosurgery service, and preoperative aspirin use. Following logistic regression only nonreversal of heparin, intraoperative hypotension, and carotid shunt placement were identified as multivariate predictors of post-CEA hematoma formation. More time was spent in critical care settings (ICU/PACU) (P < 0.01) and there was increased perioperative mortality (P = 0.04) within the hematoma group.
Conclusions
Post-CEA hematoma formation is associated with increased morbidity and mortality. Non-reversal of heparin, intraoperative hypotension, and carotid shunt placement are multi-variate predictors of post-CEA hematoma formation.
Résumé
Objectif
Identifier les facteurs de risques de formation d’un hématome post-endartériectomie carotidienne (EAC) et établir son incidence à The Ottawa Hospital — Civic Campus (TOH-CC).
Méthode
On a révisé les dossiers de tous les patients qui ont subi une EAC à TOH-CC du 1 janvier 1996 au 31 décembre 1997. Les cas d’hématome post-EAC ont été identifiés et inscrits à une étude cas-témoins appariés selon l’âge et le sexe à partir des sujets de la cohorte. Ces paires de sujets ont été évalués selon 31 facteurs de risque potentiels incluant les données démographiques, la coexistence de problèmes médicaux, la médication préopératoire, la prise en charge peropératoire et les paramètres postopératoires. Les facteurs de risque associés à l’hématome post-EAC avec P < 0,05 ont été introduits dans un modèle rétrospectif de régression logistique multifactorielle en paliers.
Résultats
Les dossiers de 249 patients ont été examinés et 29 cas d’hématome post-endartériectomie carotidienne ont été identifiés (incidence de 12 %). Six des 31 facteurs de risque potentiels désignés au début sont apparus comme des prédicteurs unifactoriels de la formation d’hématome post-EAC (P < 0,05): l’anesthésie générale, la mise en place d’un shunt carotidien, l’hypotension peropératoire, le non-renversement de l’héparine, le service de neurochirurgie et l’utilisation d’aspirine préopératoire. À la suite de la régression logistique, seules la non-inversion de l’héparine, l’hypotension peropératoire et la mise en place du shunt carotidien sont apparus comme des prédicteurs multifactoriels. Le temps passé à l’unité des soins intensifs (USI/salle de réveil) a été plus long (P < 0,01) pour le groupe présentant un hématome et la mortalité périopératoire était plus élevée (P = 0,04).
Conclusion
La formation d’un hématome post-EAC est associée à une morbidité et à une mortalité croissantes. Le non-renversement de l’héparine, l’hypotension peropératoire et la mise en place d’un shunt carotidien sont des prédicteurs multifactoriels de la formation d’un hématome post-EAC.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Shah CP. Public Health and Preventative Medicine in Canada, 3rd ed. Toronto: University of Toronto Press, 1994: 108–10.
Kelley RE. Stroke prevention and intervention. New options for improved outcomes. Postgrad Med 1998; 103: 43–61.
North American Symptomatic Carotid Endarterectomy Trial Collaborators. Beneficial effect of carotid endarterectomy in symptomatic patients with highgrade carotid stenosis. N Engl J Med 1991; 325: 445–53.
European Carotid Surgery Trialists’ Collaborative Group. MRC European Carotid Surgery Trial: interim results for symptomatic patients with severe (70–99%) or with mild (0–29%) carotid stenosis. Lancet 1991; 337: 1235–43.
Hsia DC, Moscoe LM, Krushat WM. Epidemiology of carotid endarterectomy among medicare beneficiaries. 1985–1996 Update. Stroke 1998; 29: 346–50.
Moore WS, Barnett HJM, Beebe HG, et al. Guidelines for carotid endarterectomy. A multidisciplinary consensus statement from the ad hoc committee, American Heart Association. Stroke 1995; 26: 188–201.
O’Sullivan JC, Wells DG, Wells GR. Difficult airway management with neck swelling after carotid endarterectomy. Anesth Intensive Care 1986; 14: 460–4.
Kunkel JM, Gomez ER, Spebar MJ, Delgado RJ, Jarstfer BS, Collins GJ Jr. Wound hematomas after carotid endarterectomy. Am J Surg 1984; 148: 844–7.
Carmichael FJ, McGuire GP, Wong DT, Crofts S, Sharma S, Montanera W. Computed tomographic analysis of airway dimensions after carotid endarterectomy. Anesth Analg 1996; 83: 12–7.
Welling RE, Ramadas HS, Gansmeller KJ. Cervical wound hematoma after carotid endarterectomy. Ann Vasc Surg 1989; 3: 229–31.
Corson JD, Chang BB, Shah DM, Leather RP, DeLeo LM, Karmody AM. The influence of anesthetic choice on carotid endarterectomy outcome. Arch Surg 1987; 122: 807–12.
Nunn DB. Carotid endarterectomy: an analysis of 234 operative cases. Ann Surg 1975; 182: 733–8.
Sundt TM Jr, Sandok BA, Whisnant JP. Carotid endarterectomy. Complications and preoperative assessment of risk. Mayo Clin Proc 1975; 50: 301–06.
Zarins CK. Carotid endarterectomy: the gold standard. J Endovasc Surg 1996; 3: 10–5.
Wong DHW. Perioperative stroke. Part I: General surgery, carotid artery disease, and carotid endarterectomy. Can J Anaesth 1991; 38: 347–73.
Schiro J, Mertz GH, Cannon JA, Cintora I. Routine use of a shunt for carotid endarterectomy. Am J Surg 1981; 142: 735–8.
Brott T, Thalinger K. The practice of carotid endarterectomy in a large metropolitan area. Stroke 1984; 15: 950–5.
Nunn DB. Carotid endarterectomy in patients with territorial transient ischémic attacks. J Vasc Surg 1988; 8: 447–52.
Muuronen A. Outcome of surgical treatment of 110 patients with transient ischemic attack. Stroke 1984; 15: 959–64.
Gumerlock MK, Neuwelt EA. Carotid endarterectomy: to shunt or not to shunt. Stroke 1988; 19: 1485–90.
Peitzman AB, Webster MW, Loubeau J-M, Grundy BL, Bahnson HT. Carotid endarterectomy under regional (conductive) anesthesia. Ann Surg 1982; 196: 59–64.
Lee KS, Davis CH Jr, McWhorter JM. Low morbidity and mortality of carotid endarterectomy performed with regional anesthesia. J Neurosurg 1988; 69: 483–7.
Davies MJ, Mooney PH, Scott DA, Silbert BS, Cook RJ. Neurologic changes during carotid endarterectomy under cervical block predict a high risk of postoperative stroke. Anesthesiology 1993; 78: 829–33.
Spetzler RF, Martin N, Hadley MN, Thompson RA, Wilkinson E, Raudzens PA. Microsurgical endarterectomy under barbiturate protection: a prospective study. J Neurosurg 1986; 65: 63–73.
Cirone R, Sullivan P, Posner M, Cole CW, Lindsay P. Regional vs general anaesthesia for carotid endarterectomy surgery. Can J Anaesth 1994; 41: A37-A.
Clark JC, Stanley TH. Anesthesia for vascular surgery.In: Miller RD (Ed.). Anesthesia, 4th ed. New York: Churchill Livingstone Inc., 1994: 1864–9.
Riles TS, Imparato AM, Jacobowitz GR, et al. The cause of perioperative stroke after carotid endarterectomy. J Vasc Surg 1994; 19: 206–16.
Krul JMJ, van Gijn J, Ackerstaff RGA, Eikelboom BC, Theodorides T, Vermeulen FEE. Site and pathogenesis of infarcts associated with carotid endarterectomy. Stroke 1989; 20: 324–8.
Steed DL, Peitzman AB, Grundy BL, Webster MW. Causes of stroke in carotid endarterectomy. Surgery 1982; 92: 634–41.
Mauney MC, Buchanan SA, Lawrence WA, et al. Stroke rate is markedly reduced after carotid endarterectomy by avoidance of protamine. J Vasc Surg 1995; 22: 264–70.
Wells J, Nicosia S, Wale C, Smith LJ, Buchanan MR. Thrombin generation in patients undergoing carotid endarterectomy: implications in acute vessel wall closure and antithrombic therapy. Thromb Res 1994; 75: 419–26.
Chandler WF, Ercius MS, Ford JW, LaBond V, Burkel WE. The effect of heparin reversal after carotid endarterectomy in the dog. J Neurosurg 1982; 56: 97–102.
Ercius MS, Chandler WF, Ford JW, Burkel WE. Early versus delayed heparin reversal after carotid endarterectomy in the dog. J Neurosurg 1983; 58: 708–13.
Spallone A, Sasaki T, Kassell NF, Tamashita M. Effect of antithrombotic agents after carotid endarterectomy in rats. Neurosurgery 1985; 16: 780–5.
Treiman RL, Cossman DV, Foran RF, Levin PM, Cohen JL, Wagner WH. The influence of neutralizing heparin after carotid endarterectomy on postoperative stroke and wound hematoma. J Vasc Surg 1990; 12: 440–6.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Self, D.D., Bryson, G.L. & Sullivan, P.J. Risk factors for postcarotid endarterectomy hematoma formation. Can J Anesth 46, 635–640 (1999). https://doi.org/10.1007/BF03013950
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03013950