Abstract
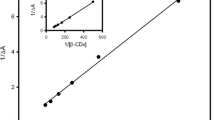
Inclusion complex of Orange II with β-Cyclodextrin (β-CD) and the anti-photolysis effect under UV-light were investigated. The molar ratio of inclusion complex of β-Cyclodextrin and Orange II is 1∶1. The formation constantK = 1.236×103 L/mol was determined by the UV and Fluorescence spectra respectively, which was quite in accordance with the calculation with a modified Benesi-Hildbrand equation. The inclusion complex was characterized by the IR spectra and the molar ratio of inclusion complex is 1∶1 too. The formation constantK = 1.266×103 L/mol was determined by1H NMR analysis and was nearly the same by UV and fluorescence spectra. The photocatalytic decolorization rate of Orange II solutions containing β-CD and TiO2 was smaller by 51.9% than that of the Orange II solutions only containing TiO2, while in the case of direct photolysis of Orange II solutions, β-CD can lower the photolysis rate by 48.1% under UV-light. This result indicates β-CD can inhibit the photolysis and photocatalytic decolorization of Orange II under UV-light. The β-CD inclusion complex was found to be persistent to UV-light photolysis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Matsubara K, Abe K, Irie T. Improvement of Nasal Bioavailability of Luteinizing Hormone-Releasing Hormone Agonist, Buserelin, by Cyclodextrins Derivatives in Rats.J. Pharm Sci, 1995,84(11): 1295–1300.
Kamiya M, Nakamura K, Sasaki C. Inclusion Effects of Cyclodextrins on Photodegradation Rates of Parathion and Paraoxon in Aquatic Medium.Chemosphere, 1994,28(11): 1961–1966.
Ishiwata S, Kamiya M. Cyclodextrin Inclusion: Catalytic Effects on the Degradation of Organophosphorus Pesticides in Neutral Aqueous Solution.Chemosphere, 1999,39(10): 1595–1600.
Scalia S, Villani S, Scatturin A,et al. Complexation of the Sunscreen Agent, Butyl-Methoxydibenzoylmethane, with Hydroxypropyl-β-Cyclodextrin.International Journal of Pharmaceutics, 1998,175: 205–213.
Iijima T, Karube Y. The Interaction of Acid Azo Dyes with Chemically Modified β-cyclodextrins.Dyes and Pigments, 1998,36(4): 305–311.
Kalyanasunderam K.Photochemistry in Microheterogeneous Systems. London: Academic Press, 1987.
Roberts E L, Chou P T, Alexander T A,et al. Effects of Organized Media on the Excited-State Intramolecular Proton Transfer of 10-Hydroxybenzo[h] Quinoline.J Phys Chem, 1995,99(15): 5431–5437.
Sanchez F G, Lopez M H, Marquez Gomez J C. Fluorimetric Determination of Scandium Using the Cyclodextrin-1,4-Dihydroxyanthraquinone Inclusion Complex.Analyst, 1987,112(7): 1037–1040.
Takuma T, Deguchi T, Sanemasa I. Association of Halobenzenes with Cyclodextrins in Aqueous Medium.Bull Chem Soc Jpn, 1990,63(4): 1246–1248.
Chen Liang, Chao Jian-bin, Gu Jun. One- and Two-dimensional NMR Study on Inclusion Compound of Procaine Hydrochloride with β-Cyclodextrin.Chinese Journal of Magnetic Resonance, 2000,17(3): 233–239 (Ch)
Author information
Authors and Affiliations
Corresponding authors
Additional information
Foundation item: Supported by the National Natural Science Foundation of China (20177017)
Biography: Lu Ping (1958), male. Associate professor, research direction: environmental photochemistry.
Rights and permissions
About this article
Cite this article
Ping, L., Bin, L., Zhe, Z. et al. Formation of the inclusion complex of Orange II with β-Cyclodextrin and its photostability. Wuhan Univ. J. Nat. Sci. 8, 1147–1152 (2003). https://doi.org/10.1007/BF02903689
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02903689