Abstract
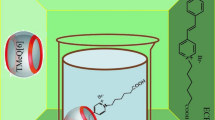
Self-assembled complexes between cage compounds cucurbit[n = 5–8]urils and hexamethylenetetramine were studied by using NMR techniques. Experimental results reveal that hexamethylenetetramine can lid cucurbit [5] uril to forming self-assembled capsules in which nothing is encapsulated yet; the cavity of the cucurbit[7]uril can accommodate a hexamethylenetetramine molecule to form a selfassembled host-guest inclusion. Moreover, both the cavity interaction of the cucurbit[7]uril with hexamethylenetetramine·HCl and the portal interaction of the dipole carbonyl of the cucurbit[7]uril with hexamethylenetetramine·HCl lead to form self-assembled capsules in which the hexamethylenetetramine·HCl are encapsulated in the hexamethylenetetramine·HCl “lidded” cucurbit[7]uril. Although the structures of the portal and cavity to cucurbit[5]uril are similar, there is no obvious interaction between decamethylcucurbit[5]uril and hexamethylenetetramine, and also between cucurbit [6]uril or cucurbit[8]uril and hexamethylenetetramine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Liu, Y., You, C. C., Zhang, H. Y., Supramolecular Chemistry (in Chinese), Tianjin: Nankai University Press, 2001, 385–448.
Winston, O., Marielle, G. K., Angel, E. et al., New-Cucurbit[7]uril: A Very Effective Host for Viologens and Their Cation Radicals, Organic Letters, 2002, 4(10): 1791–1794
Jansen, K., Buschmann, H. J., Zliobaite, E. et al., New-Steric factors influencing the complex formation with cucurbit[6]uril, Thermochimica Acta, 2002, 385(1-2): 177–184.
Freeman, W. A., Mock, W. L., Shih, N. Y., Cucurbituril, J. Am. Chem. Soc., 1981, 103: 7367–7368.
Day, A. I., Arnold, A. P., Method for synthesis cucurbiturils, WO 0068232, 2000, 8.
Kim, J., Jung, I. S., Kim, S. Y. et al., New cucurbituril homologues: Syntheses, isolation, characterization, and X-ray crystal structures of cucurbit[n]uril (n = 5, 7, and 8), J. Am. Chem. Soc., 2000, 122(3): 540–541.
Day, A. I., Blanck, R. J., Arnold, A. P., A cucurbituril-based gyroscane: a new supramolecular form, Angew Chem. Int. Ed, 2002, 41(2): 275–277.
Neugebauer, R., Knoche, W., Host-guest complexes of cucurbituril with 4-amino-4-nitroazobenzene and 4,4′-diaminoazobenzene in acidic aqueous solutions, J. Chem. Soc., Perkin Trans. 2, 1998, 3: 529–534.
Jeon, Y. M., Heo, J., Whang, D. et al., Molecular container assembly capable of controlling binding and release of its guest molecules: reversible encapsulation of organic molecules in sodium ion complexed cucurbituril, J. Am. Chem. Soc, 1996, 118(40): 9790–9791.
Whang, D., Heo, J., Park, J. H. et al., A molecular bowl with metal ion as bottom: reversible inclusion of organic molecules in cesium ion complexed cucurbituril, Angew. Chem. Int. Ed. Engl., 1998, 37(1/2): 78–80.
Blanck, R., Sleeman, Day, A. I., Cucurbit[7]uril and o-carborane self-assemble to form a molecular ball bearing, Nano Lett., 2002, 2(2): 147–149.
Kim, S. Y., Jung, I. S., Kim, K., Macrocycles within macrocycles: cyclen, cyclam, and their transition metal complexes encapsulated in cucurbit[8]uril, Angew Chem. Int. Ed. Engl., 2001, 40(11): 2119–2121.
Yao, X. Q., Shen, Y. Q., Tao, Z. et al., New cage compounds–Cucurbit[n]uril (n = 5,7 and 8,), Journal of Guizhou University (in Chinese), 2003, 20(1): 105–110.
Han, B. H., Liu, Y., Molecular Recognition and Assembly of cucurbituril, Chinese Journal of Inorganic Chemistry, 2003, 23(2): 139–149.
Lorenzo, S., Day, A. I., Craig, D. et al., The first endoannular metal halide-cucurbituril: cis-SnCl4(OH2)2@cucurbit[7]uril, Cryst. Eng. Comm., 2001, 49: 1–7.
Samsonenko, D. G., Virovets, A. V., Lipkowski, J. et al., Newdistortion of the cucurbituril molecule by an Included 4-methylpyridinum cation. Journal of Structural Chemistry (Translation of Zhurnal Strukturnoi Khimii), 2002, 43(4): 664–668.
Samsonenko, D. G., Gerasko, O. A., Lipkowski, J. et al., New synthesis and crystal structure of the nanosized supramolecular SmIII complex with macrocyclic cavitand cucurbituril [Sm(H2O)4]2(C36H36N24O12)3Br6 · 44H2O, Russian Chemical Bulletin (Translation of Izvestiya Akademii Nauk, Seriya Khimicheskaya), 2002, 51(10): 1915–1918.
Zhang, G. L., Xu, Z. Q., Xue, S. F. et al., A New Family of Cage Compounds Cucurbit[n]urils: (II) Influence of Acidity, Alkaline and Alkaline-earth Metal Ions on Solubility of Cucurbit[n = 5—8]urils, Chinese Journal of Inorganic Chemistry (in Chinese), 2003, 19(6): 655–659.
Flinn, A., Hough, G. C., Stoddart, J. F. et al., Decamethylcucurbit[5]uril, Angew. Chem., Int. Ed. Engl., 1992, 31(11): 1475–1477.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Shen, Y., Xue, S., Zhao, Y. et al. NMR study on self-assembled cage complex of hexamethylenetetramine and cucurbit[n]urils. Chin. Sci. Bull. 48, 2694–2697 (2003). https://doi.org/10.1007/BF02901758
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02901758