Summary
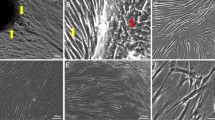
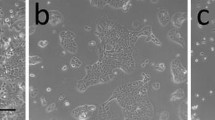
This study examined the effect of 3T3 fibroblasts on the expression of anchorage independence and the degree of cornification in early cultures of three carcinoma-derived epithelial cell lines (R59, R63a, R63b) and in one cell line derived from non-malignant dysplastic epithelium where there was no evidence of invasion (R66a). The epithelial cell lines originated from the palatal (R63a, R66a) and the lingual (R59, R63b) mucosa of rats that had been painted with the carcinogen 4-nitroquinoline N-oxide. In the absence of 3T3 fibroblasts, progressive culture resulted in an increase in the colony forming efficiency (CFE) of R63a, R63b and R59 and a decrease in the percentage of cornified cells in all cell lines. 3T3 fibroblasts caused a decrease in the CFE and the degree of cornification in the 3T3-dependent cell line (R63a), particularly at the lower passages, but these parameters remained essentially unchanged by 3T3 fibroblasts in the 3T3-independent cell lines (R59, R63b). 3T3 fibroblasts did not influence the cornification of R66a and this cell line remained anchorage dependent throughout the study. The results suggest that in malignant cell lines characterised by being independent of 3T3 fibroblasts (R63b, R59) the CFE was inversely correlated to the degree of cornification. However, in the malignant cell line showing a greater dependence on support (R63a) the relationship between CFE and cornification was unclear because these parameters may have been modulated by the presence of 3T3 fibroblasts. The cell line from dysplastic non-invasive tissue (R66a) differed from its malignant counterparts in the fact that CFE and cornification were unaffected by 3T3 fibroblasts despite previous studies showing a dependence on mesenchymal support.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anzano MA, Roberts AB, Sporn MB (1986) Anchorage-independent growth of primary rat embryo cells is induced by platelet-derived growth factor and inhibited by type-beta transforming growth factor. J Cell Physiol 126:312–318
Bouick N, Head M (1985) The majority of independently transformed BHK cell clones share a single functional lesion which determines anchorage independence and influences tumourigenicity. In Vitro 21:463–469
Bouick N, Stoler A, Polverini PJ (1986) Coordinate control of anchorage independence, actin cytoskeleton and angiogenesis by human chromosome 1 in hamster human hybrids. Cancer Res 46:5101–5105
Boukamp P, Rupniak HTR, Fusenig NE (1985) Environmental modulation of the expression of differentiation and malignancy in six human squamous cell carcinoma cell lines. Cancer Res 45:5582–5592
Boyce ST, Ham RG (1983) Calcium-regulated differentiation of normal human epidermal keratinocytes in chemically defined clonal culture and serum-free serial culture. J Invest Dermatol 81:335–405
Brittain MG, Brittain DE, Sarif AM (1982) Enhancement of growth of human colon tumour cell lines by feeder cells. J Natl Cancer Inst 69:767–771
Chen TR (1977) In situ detection of mycoplasma contamination in cell cultures by fluorescent Hoescht 33258 stain. Exp Cell Res 104:225–256
Citron ML, Jaffe ND, Hamburger AW, Lindblad AL, Banda FP, Yenson A, Nathan KA, Cohen MH (1986) Improvement of human tumor cloning assay by suspension of fibroblasts into the bottom layer of agarose. Cancer 57:2357–2362
Cowley G, Gusterson BA, Knight J (1983) Growth in agar and tumour formation in immunologically incompetent mice as criteria for keratinocyte transformation. Cancer Lett 21:95–104
Crane IJ, Luker J, Stone A, Scully C, Prime SS (1986) Characterisation of malignant rat keratinocytes in culture following the induction of oral squamous cell carcinomas in vivo. Carcinogenesis 7:1723–1727
Dover R, Watt FM (1987) Measurement of the rate of epidermal terminal differentiation: expression of involucrin by S-phase keratinocytes in culture and in psoriatic plaques. J Invest Dermatol 89:349–352
Easty DM, Easty GC, Carter RL, Monaghan P, Butler LJ (1981) Ten human carcinoma cell lines derived from squamous carcinomas of the head and neck. Br J Cancer 43:772–785
Foulds L (1964) Tumour progression and neoplastic development. In: Emmelot P, Muhobock O (eds) Cellular control mechanisms and cancer. Elsevier Publishing, Corp Amsterdam, pp 242–259
Fusenig NE (1986) Mammalian epidermal cells in culture. In: Bereiter-Hahn J, Matoltsy AG, Richards KS (eds) Biology of the integument, 2. Vertebrates. Springer Verlag, Berlin Heidelberg New York, pp 409–442
Green H (1980) The keratinocyte as a differentiated cell type. Harvey Lect 74:101–139
Green H, Rheinwald JG, Sun T-T (1977) Properties of an epithelial cell type in culture: the epidermal keratinocyte and its dependence on products of the fibroblast. Cell Shape and Surface Architecture. A R Liss, New York, pp 493–500
Holbrook KA, Hennings H (1983) Phenotypic expression of epidermal cells in vitro: a review. J Invest Dermatol 81:11s-24s
Kirk D, Szalay MF, Kaighn ME (1981) Modulation of growth of a human prostatic cancer cell line (PC-3) in agar culture by normal human lung fibroblasts. Cancer Res 41:1100–1103
Kuratomi Y, Ono M, Yasutakie C, Mawatari M, Kuwano M (1987) Mouse Balb/c 3T3 cell mutant with low epidermal growth factor activity: induction of stable anchorage-independent growth by transforming growth factor. J Cell Physiol 130:51–57
Laboisse CL, Angeron C, Potet F (1981) Growth and differentiation of human gastrointestinal adenocarcinoma stem cells in soft agarose. Cancer Res 41:310–315
La Rocca PJ, Rheinwald JG (1985) Anchorage-independent growth of normal human mesothelial cells: a sensitive bioassay for EGF which discloses the absence of this factor in foetal calf serum. In Vitro 21:67–72
Luker J, De Gay L, Crane IJ, Stone A, Scully C, Prime SS (1988) The inter-relationship between anchorage independence and tumourigenicity in early cultures of oral keratinocytes. Virchows Arch [Cell Pathol] 54:246–251
Parkinson EK (1985) Defective responses of transformed keratinocytes to terminal differentiation stimuli. Their role in epidermal tumour promotion by phorbol esters and by deep skin wounding. Br J Cancer 53:479–493
Pera MF, Gorman PA (1984) In vitro analysis of multistage carcinogenesis: development of indefinite renewal capacity and reduced growth factor requirements in colony forming keratinocytes precedes malignant transformation. Carcino-genesis 5:671–682
Prime SS, Rosser TJ, Davies LS, Scully C (1987) Loss of epithelial cell surface carbohydrates during experimental oral carcinogenesis in the rat. Br J Cancer 55:633–638
Rheinwald JG (1980) Serial cultivation of normal human epidermal keratinocytes. Meth Cell Biol 21a:229–254
Rheinwald JG, Beckett MA (1980) Defective terminal differentiation in culture as a consistent and selectable character of malignant human keratinocytes. Cell 22:629–632
Rheinwald JG, Beckett MA (1981) Tumourigenic keratinocyte lines requiring anchorage and fibroblast support cultured from human squamous cell carcinomas. Cancer Res 41:1657–1663
Rheinwald JG, Green H (1975) Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinising colonies from single cells. Cell 6:331–334
Rheinwald JG, Green H (1977) Epidermal growth factor and the multiplication of cultured human keratinocytes. Nature 265:421–424
Rice RH, Green H (1977) The cornified envelope of terminally differentiated human epidermal keratinotes consists of a cross-linked protein. Cell 11:417–422
Rupniak HT, Rowlatt C, Lane EB, Steel JG, Trejdosiewiz LK, Laskiewicz B, Povey S, Hill BT (1985) Characteristics of four new human cell lines derived from squamous cell carcinomas of the head and neck. J Natl Cancer Inst 75:621–635
Skehan P, Friedman SJ (1981) The transformed cell. Academic Press, New York, pp 7–65
Speers WC (1982) Conversion of malignant murine embryonal carcinomas to benign teratomas by chemical induction of differentiation in vivo. Cancer Res 42:1843–1849
Sun T-T, Green H (1976) Differentiation of the epidermal keratinocyte in cell culture: formation of the cornified envelope. Cell 9:511–521
Virolainen E, Carey TE, Wicha M, Krause CJ (1983) Factors affecting the growth of head and neck squamous cell carcinoma cell lines. Otolaryngol Head Neck Surg 91:126–135
Wallenius K, Lekholm J (1973) Oral cancer in rats induced by the water soluble carcinogen 4-nitroquinoline N-oxide. Odont Rev 24:39–48
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Luker, J., Crane, I.J., Scully, C. et al. The effect of 3T3 fibroblasts on the expression of anchorage independence and cornification of oral keratinocytes. Virchows Archiv B Cell Pathol 57, 19–26 (1989). https://doi.org/10.1007/BF02899061
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02899061