Abstract
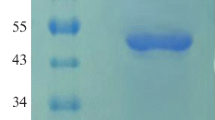
Phytase genephyA2, whose signal peptide encoding sequence and intron sequence had been removed, was modified. The Arg-encoding codons CGG and CAG inphyA2 were mutated into synonymous codon AGA. The modifiedphyA2 was fused behind a-factor signal sequence under the control ofAOX1 promoter in plasmid pPIC9, then introduced into the hostPichia pastoris by electroporation. The results of Southern blotting analysis and Northem blotting analysis demonstrated that thephyA2 gene had integrated into the genome ofP. pastoris and transcribed. The result of SDS-PAGE of the phytase expressed by P.pastoris showed that the modifiedphyA2 had been overexpressed and secreted. The concentration of the phytase expressed by P.pastoris with modifiedphyA2 exceeded 15 000 U/mL, which had a 3 000-fold increase over that of originAspergillus niger 963 and was 37 times higher than that of recombinantP. pastoris with non-modifiedphyA2.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Nelson, T. S., The utilization of phytate phosphorus by poultry—A review,Poult. Sci., 1967, 46: 862.
Sharma, C. B., Goel, M., Irshad, M., Myo-inositol hexaphosphate as a potential inhibitor of a-amylases of different origins,Phytochemistry, 1978, 17: 201.
Nelson, T. S., Shieh, T. R., Wodzinski, R. J. et al., The availability of phytate phosphorus in soya bean meal before and after treatment with a mold phytase,Poult. Sci., 1968, 47: 1842.
Sambrook, J., Fritsch, E. F., Maniatis, T.,Molecular Cloning: A Laboratory Manual, New York: Cold Spring Harbor Laboratory Press, 1989.
Ullah, A. H. J.,Aspergillus ficuum phytase: partial primary structure, substrate selectivity, and kinetic characterization.Preparative Biochemistry, 1988, 18(4): 459.
Sharp, P. M., Tuohy, T. M. F., Mosurski, K. R., Codon usage in yeast: cluster analysis clearly differentiates highly and lowly expression genes,Nucleic. Acids Res., 1986, 14(13): 5125.
Piddington, C. S., Houston, C. S., Paloheimo, M. et al., The cloning and sequencing of the genes encoding phytase (phy) and pH 2.5-optimum acid phosphatase (aph) fromAspergillus niger var.awamori, Gene, 1993, 133: 55.
Van Hartingsveldt, M., Van Zeijl, C. M. J., Harteveld, G. M. et al., Cloning, characterization and overexpression of the phytase-encoding gene (phyA) ofAspergillus niger, Gene, 1993, 127: 87.
Moore, E., Helly, V. R., Conneely, O. M. et al., Molecular cloning, expression and evaluation of phosphohydrolases for phytate-degrading activity,J. Ind. Microbiol., 1995, 14: 396.
Cregg, J. M., Vedvick, T. S., Raschke, W. C., Recent advances in the expression of foreign genes inPichia pastoris, Bio/Technology, 1993, 11: 905.
11 Wagner, E. H., Biochemical conversions by yeast fermentation at high-cell densities, US Patent 44114329, 1983.
Vedvick, T., Buckholz, R. G., Engel, M. et al., High-level secretion of biologically active aprotinin from the yeastPichia pastoris, J. Ind. Microbiol., 1991, 7: 197.
Siegel, R. S., Brierley, R. A., Methylotrophic yeastPichia pastoris prcduced in high-cell-density fermentations with high cell yields as vehicle for recombinant protein production,Biotechnol. Bioeng., 1989, 34:403.
Author information
Authors and Affiliations
Additional information
Project supported by the “863” program, National Science and Technology Commission of China.
Rights and permissions
About this article
Cite this article
Yao, B., Zhang, C., Wang, J. et al. RecombinantPichia pastoris overexpressing bioactive phytase. Sci. China Ser. C.-Life Sci. 41, 330–336 (1998). https://doi.org/10.1007/BF02895110
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02895110