Abstract
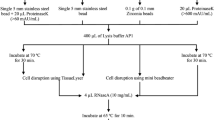
The preparation of good quality genomic DNA from microalgae and plants is often time-consuming because of the need to remove contaminants that may interfere with the downstream enzymatic manipulation of the DNA. Simpler protocols have been reported but these are applicable only to a few species and in many cases are not effective for removing trace contaminants. In this report, we describe a modification of existing protocols that significantly simplified the preparation of genomic DNA from cyanobacteria and plants. A key step in our protocol is the precipitation of DNA in a high concentration of salt (2–2.5 M NaCl) in the presence of isopropanol, immediately following phenol and chloroform extractions. The preparation and enzymatic digestion of the DNA can be performed in a single day. The DNA was easily digested in 2 h at normal restriction enzyme concentrations, and is highly suitable for PCR and Southern hybridization. We successfully used this simplified protocol to prepare genomic DNA from several filamentous cyanobacteria, such asAnabaena sp. PCC 7120,Anabaena siamensis, andSpirulina strains M2 and Kenya. This protocol may also be useful for preparing genomic DNA from other algae and from higher plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA and Struhl K (1998) Current Protocols in Molecular Biology. J. Wiley & Sons, New York, NY.
Ben-Dov E, Zaritsky A, Dahan E, Barak Z, Sinai R, Manasherob R, Khameraev A, Troyetskaya A, Dubitsky A, Berezina N and Margalith Y (1997) Extended screening by PCR for seven cry-group genes from field-collected strains ofBacillus thuringiensis. Appl Environ Microbiol 63: 4883–4890.
Golden J, Carrasco C, Mulligan M, Schneider G and Haselkorn R (1988) Deletion of a 55-kilobase-pair DNA element from the chromosome during heterocyst differentiation ofAnabaena sp. strain PCC 7120. J Bacteriol 170: 5034–5041.
Golden SS, Brussian J and Haselkorn R (1987) Genetic engineering of the cyanobacterial chromosome. Methods Enzymol 153: 215–231.
Joset F (1987) Transformation inSynechocystis PCC 6714 and 6803: Preparation of chromosomal DNA. Methods Enzymol 167: 712–714.
Kallas T, Coursin T and Rippka R (1985) Different organization ofnif genes in nonheterocystous and heterocystous cyanobacteria. Plant Mol Biol 5: 321–329.
Kallas T and Malkin R (1988) Isolation and characterization of genes for cytochromeb6/f complex. Methods Enzymol 167: 779–794.
Philippis RD and Vincenzini M (1998) Exocellular polysaccharides from cyanobacteria and their possible applications. FEMS Microbiol Rev 22: 151–175.
Porebski S, Bailey LG and Baum BR (1997) Modification of a CTAB DNA extraction protocol for plants containing high polysacharide and polyphenol components. Plant Mol Biol Reptr 15: 8–15.
Sambrook J, Fritsch EF and Maniatis T (1989) Molecular Cloning: A Laboratory Manual. 2nd ed., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
Williams JGK (1987) Construction of specific mutations in photosystem II photosynthetic reaction center by genetic engineering methods inSynechocystis 6803. Methods Enzymol 167: 766–778.
Wu XQ, Vennison SJ, Huirong L, Ben-Dov E, Zaritsky A and Boussiba S (1997) Mosquito larvicidal activity ofAnabaena sp. strain PCC 7120 with a combination of delta-endotoxin genes fromBacillus thuringiensis subsp.israelensis. Appl Environ Microbiol 63: 4971–4975.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, X., Zarka, A. & Boussiba, S. A simplified protocol for preparing DNA from filamentous cyanobacteria. Plant Mol Biol Rep 18, 385–392 (2000). https://doi.org/10.1007/BF02825067
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02825067