Abstract
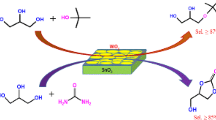
Tungsten oxide-titania catalysts were prepared by drying powdered Ti(OH)4 with ammonium metatungstate aqueous solution, followed by calcining in air at high temperature. Characterization of prepared catalysts was performed by using FTIR, Raman, XPS, XRD, and DSC and by measuring surface area. Upon the addition of tungsten oxide to titania up to 20 wt%, the specific surface area and acidity of catalysts increased in proportion to the tungsten oxide content due to the interaction between tungsten oxide and titania. Since the TiO2 stabilizes the tungsten oxide species, for the samples equal to or less than 20 wt%, tungsten oxide was well dispersed on the surface of titania, but for the samples containing 25 wt% or above 25 wt%, the triclinic phase of WO3 was observed at calcination temperature above 400 °C. The catalytic activities for 2-propanol dehydration and cumene dealkylation were correlated with the acidity of catalysts measured by ammonia chemisorption method.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Adeeva, V., de Haan, J.W., Janchen, J., Lei, G. D., Schunemann, V., van de Ven, L. J. M., Sachtler, W. M. N. and van Santen, R. A., “Acid Sites in Sulfated and Metal-promoted Zirconium Dioxide Catalysts,”J. Catal.,151, 364 (1995).
Alemany, L. J., Berti, F., Busca, G., Ramis, G., Robba, D., Toledo, G. P. and Trombetta, M., “Characterization and Composition of Commercial V2O5-WO3-TiO2 SCR Catalysts,”Appl. Catal. B: Enviromental,10, 299 (1996).
Arata, K., “Solid Superacids,”Adv. Catal.,37, 165 (1990).
Basrur, A. G., Parwardham, S. R. and Vyas, S. N., “Propene Metathesis over Silica-supported Tungsten Oxide Catalyst-catalyst Induction Mechanism,”J. Catal.,127, 86 (1991).
Chan, S. S., Wachs, I. E. and Murrell, L. L., “Relative Raman Crosssections of Tungsten Oxides,”J. Catal.,90, 150 (1984).
Cheung, T. K., d’Itri, J. L., Lange, F. C. and Gates, B. C., “Neopentane Cracking Catalyzed by Iron and Manganese-promoted Sulfated Zirconia,”Catal. Lett.,31, 153 (1995).
DeCanio, S. J., Sohn, J. R., Fritz, P. O. and Lunsford, J. H., “Acid Catalysis by Dealuminated Zeolite-Y,”J. Catal.,101, 132 (1986).
Ebitani, K., Konish, J. and Hattori, H., “Skeletal Isomerization of Hydrocarbons over Zirconium Oxide Promoted by Platinum and SulfateIion,”J. Catal.,130, 257 (1991).
Engweiler, J., Harf, J. and Baiker, A., “WOx/TiO2 Catalysts Prepared by Grafting of Tungsten Alkoxides: Morphological Properties and Catalytic Behavior in the Selective Reduction of NO by NH3,”J. Catal.,159, 259 (1996).
Figueras, F., Coq, B., Walter, C. and Carriat, J. Y., “Hydroconversion of Methylcyclohexane on Bifunctional Sulfated Zirconia-supported Platinum Catalysts,”J. Catal.,169, 103 (1997).
Guitierrez-Alejandre, A., Ramirez, J. and Busca, G., “A Vibrational and Spectroscopic Study of WO3/TiO2-Al2O3 Catalyst Precursors,”Langmuir,14, 630 (1998).
Hino, M. and Arata, K., “Synthesis of Solid Superacid of Tungsten Oxide Supported on Zirconia and its Catalytic Action for Reactions of Butane and Pentane,”J. Chem. Soc., Chem. Commun., 1259 (1988).
Horsley, J. A., Wachs, I. E., Brown, J.M., Via, G. H. and Hardcastle, F. D., “Structure of Surface Tungsten Oxide Species in the WO3/ Al2O3 Supported Oxide System from X-ray Absorption Nearedge Spectroscopy and Raman Spectroscopy,”J. Phys. Chem.,91, 4014 (1987).
Hsu, C. Y., Heimbuch, C. R., Armes, C. T. and Gates B. C., “A Highly Active Solid Superacid Catalyst for n-Butane Isomerization: a Sulfated Oxide Containing Iron, Manganese and Zirconium,”J. Chem. Soc., Chem. Commun., 1645 (1992).
Keogh, R. A., Srinivasan, R. and Davis, B. H., “Pt-SO4 2- -ZrO2 Catalysts the Impact of Water on Their Activity for Hydrocarbon Conversion,”J. Catal.,151, 292 (1995).
Lee, J. K. and Rhee, H. K., “Effect of Metal/Acid Balance in Ptloaded Large Pore Zeolites on the Hydroisomerization of n-Hexane and n-Heptane,”Korean J. Chem. Eng.,14, 451 (1997).
Meijers, S., Gielgens, L. H. and Ponec, V., “IR Spectroscopic Study of the Skeletal Isomerization of Butane by Alumina-supported Tungsten Oxide Catalysts,”J. Catal.,156, 147 (1995).
Satsuma, A., Hattori, A., Mizutani, K., Furuta, A., Niyamoto, A., Hattori, T. and Murakami, Y., “Surface Active Sites of V2O5-WO3 Catalysts,”J. Phys. Chem.,92, 6052 (1988).
Sohn, J. R., Cho, S. G., Pae, Y. I. and Hayashi, S., “Characterization of Vanadium Oxide-zirconia Catalyst,”J. Catal.,159, 170 (1996).
Sohn, J. R., Jang, H. J. and Kim, H.W., “Catalytic Activities and Acid Strengths of NiO-ZrO2 Catalysts Modified with Acids,”Korean J. Chem. Eng.,7, 7 (1990).
Sohn, J. R. and Ozaki, A., “Acidity of Nickel Silicate and its Bearing on the Catalytic Activity for Ethylene Dimerization and Butene Isomeriation,”J. Catal.,61, 29 (1980).
Sohn, J. R. and Park, M. Y., “Characterization of Zirconia-supported Tungsten Oxide Catalyst,”Langmuir,14, 6140 (1998).
Sohn, J. R. and Ryu, S. G., “Surface Characterization of Chromium Oxide-zirconia Catalyst,”Langmuir,9, 126 (1993).
Tanabe, K., Misono, M., Ono, Y. and Hattori, J., “New Solid Acids and Bases,” Elsevier Science, Amsterdam (1989).
Vaudagna, S. R., Conelli, R. A., Canavese, S. A. and Figoli, N. S., “SO4 2--ZrO2 and Pt/SO4 2--ZrO2: Activity and Stability during n-Hexane Isomerization,”J. Catal.,169, 389 (1997).
Vuurman, M. A., Wachs, I. E. and Hirt, A. M., “Structural Determination of Supported V2O5-WO3/TiO2 Catalysts by in situ Raman Spectroscopy and X-ray Photoelectron Spectroscopy,”J. Phys. Chem.,95, 9928 (1991).
Wachs, I. E., Chersich, C. C. and Hardenbergh, J. H., “Reduction of WO3/Al2O3 and Unsupported WO3:A Comparative ESCA Study,”Appl. Catal.,13, 335 (1985).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sohn, J.R., Bae, J.H. Characterization of tungsten oxide supported on TiO2 and activity for acid catalysis. Korean J. Chem. Eng. 17, 86–92 (2000). https://doi.org/10.1007/BF02789259
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02789259