Abstract
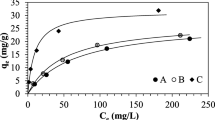
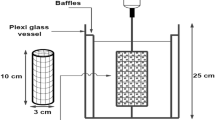
Simultaneous removal of cyanide and copper ions from electroplating wastewater was studied in a liquid-solid semi-fluidized ion exchanger bed. The diameter and the height of column are 20 mm and 600 mm, respectively. Strong-base anion exchange resin particles (Dowex 1X8-50) were contacted with synthetic solutions containing copper and cyanide ions. Cyanide and copper ions in the solution were analyzed by a cyanide electrode and ICP (inductively coupled plasma), respectively. The ion exchange equilibrium data of Cu+ removed as cyanide-copper complexes on Dowex 1X8-50 at 25 ‡C can be fitted with the Langmuir equation. Early leakage of cyanide from experimental loading profile data results in not only lower selectivity of free cyanide but also different selectivities of CN-Cu complexes due to the size and the structure of complexes. The optimum molar ratio (Q) between cyanide and copper ions is about 3 to obtain a reasonable removal rate of cyanide in this experiment.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Avery, N. L. and Fries, W., “Selective Removal of Cyanide from Industrial Waste Effluent with Ion-Exchange Resins,”Ind. Eng. Chem., Prod, Res. Dev.,14(2), 102 (1975).
Fagan, P., Paull, B., Haddad, P. R., Dume, R. and Kamar, H., “Ion Chromatographic Analysis of Cyanate in Gold Processing Samples Containing Large Concentrations of Copper (I) and other Metallo-cyanide Complexes,”J. of Chromatography A,770,175 (1997).
Goncalves, M. M. M., Pinto, A. F. and Granato, M., “Biodegradation of Free Cyanide, Thio Cyanate and Metal Complexed Cyanides in Solutions with Different Compositions,”Environmental Technology,19(2), 133 (1998).
Goto, M. and Goto, S., “Removal and Recovery of Heavy Metal by Ion Exchange Fiber,”J. Chem. Eng. Japan,20(5), 417 (1987).
Gupta, A., “Recovery of Metal-cyanide Complexes from Electoplating Wastewaters by Ion Exchange,” Ph. D. thesis (1985).
Hsu, T. L., Tran, T. and Young, D., “Modeling of the Chemical Speciation of Cyanide Species-Application to Effluent Treatment,” Ausimm Extractive Metal Con., 133 (1991).
Jae, W. M., Hong, Z. D. and Kim, M. S., “The Effective Removal Method of Copper and Cyanide in Waste Water of Metal Plating Factories,”Bulletin of Environmental Science,9 (1988).
Jennifer, B.B., Ryan, R. L. and Paziranden, M., “Comparison of Ion Exchange Resins and Biosorbents for the Removal of Heavy Metals from Plating Factory Wastewater,”Environmental Science Technology,31, 2910 (1997).
Lee, J. S., Deorkar, N. V. and Tavlarides, L. L., “Adsorption of Copper Cyanide on Chemically Active Adsorbents,”Ind. Eng. Chem, Res.,37,2812 (1998).
Lee, K. and Hong, J., “Separation and Recovery of Lead by Cation Exchange Process Combined with Precipitation,”AIChE J.,41 (12), 2653 (1995).
Short, A. E., Haselmann, S. F. and Semmens, M. J., “The GM-IX Process-A Pilot Study for Recovering Zinc Cyanides,”J. of Environmental Science & Health, Part A,32,1 (1997).
Tchobanoglous, G. and Burton, F L., “Wastewater Engineering,” 3rd Ed., Metcalf & Eddy, Inc. (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, SJ., Hwang, KR., Cho, SY. et al. Simultaneous removal of cyanide and copper ions in a semi-fluidized ion exchanger bed. Korean J. Chem. Eng. 16, 664–669 (1999). https://doi.org/10.1007/BF02708149
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02708149