Abstract
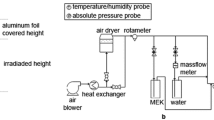
The effects of superficial gas velocity (Ug), wavelength and intensity of ultraviolet (UV) light, oxygen and H2O concentration on the photocatalytic degradation of TCE (Trichloroethylene) over TiO2/SiO2 catalyst have been determined in an annulus fluidized bed photoreactor. The key factor in determining the performance of the annulus fluidized bed photoreactor is found to be an optimum superficial gas velocity (Ug) that provides the optimum UV lighttransmit through the proper size of bubbles in the photoreactor. The degradation efficiency of TCE increases with light intensity but decreases with wavelength of the UV light and H2O concentration in the fluidized bed of TiO2/silica-gel photocatalyst. The optimum concentration of O2 for TCE degradation is found to be approximately 10%. The annulus fluidized bed photoreactor is an effective tool for high TCE degradation with efficient utilization of photon energy.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Coates, N.H. and Rice, R. L., “Sulfur Dioxide Reduction by Combustion of Coal in Fluidized Bed of Limestone,”AIChE Symp. Ser.,70, 124 (1974).
Izumi, I., Dunn, W.W., Wilbourn, K.O., Fan, F. F. and Bard, A. J., “Heterogeneous Photocatalytic Oxidation of Hydrocarbons on Platinized TiO2 Powder,”J. Phys. Chem.,84, 3207 (1980).
Kim, S. B. and Hong, S. C., “Kinetic Study for Photocatalytic Degradation of Volatile Organic Compounds in Air Using Thin Film TiO2 Photocatalyst,”Appl. Catal. B: Environ.,35, 305 (2002).
Lim, T.H., Jeong, S. M., Kim, S.D. and Gyenis, J., “Photocatalytic Decomposition of NO by TiO2 Particles,”J. Photochem. Photobiol. A: Chem.,134, 209 (2000a).
Lim, T.H., Jeong, S. M., Kim, S.D. and Gyenis, J., “Degradation Characteristics of NO by Photocatalysis with TiO2 and CuO/TiO2,”React. Kinet. Catal. Lett.,71, 223 (2000b).
Matthews, R.W. and McEvoy, S. R., “A Comparison of 254 nm and 350 nm Excitation of TiO2 in Simple Photocatalytic Reactors,”J. Photochem. Photobiol. A: Chem.,66, 255 (1992).
Nimlos, M. R., Jacoby, W. A., Blake, D.M. and Milne, T. A., “Direct Mass Spectrometric Studies of the Destruction of Hazardous Wastes: Gas-phase Photocatalytic Oxidation of TCE over TiO2,”Environ. Sci. Technol.,27, 732 (1993).
Ollis, D. F., Pelizzetti, E. and Serpone, N., “Photocatalyzed Destruction of Water Contaminants,”Environ. Sci. Technol.,25, 1522 (1993).
Peral, J. and Ollis, D. F., “Heterogeneous Photocatalytic Oxidation of Gas-phase Organics for Air Purification: Acetone, 1-butanol, Butylaldehyde, Formaldehyde, and m-xylene Oxidation,”J. Catal.,136, 554 (1992).
Tanguat, J. F., Suib, S. L. and Coughlin, R.W., “Dichloromethane Photodegradation using TiO2 Catalyst,”J. Catal.,117, 335 (1989).
Wang, K. H., Tsai, H.H. and Hsieh, Y. H., “The Kinetics of Photocatalytic Degradation of TCE in Gas Phase over TiO2 Supported on Glass Bead,”Appl. Catal.B: Environ.,17, 313 (1998).
Wang, K.H., Tsai, H.H. and Hsieh, Y.H., “A Study of Photocatalytic Degradation of TCE in Vapor Phase over TiO2 Photocatalyst,”Chemosphere,36, 2763 (1998).
Yamazaki-Nishida, S., Nagano, K. J., Phillips, L. A., Cerverna-March, S. and Anderson, M. A., “Photocatalytic Degradation of TCE in the Gas-phase using TiO2 Pellets,”J. Photochem. Photobiol. A: Chem.,70, 95, (1993).
Yue, P. L. and Khan, F., “Photocatalytic Ammonia Synthesis in a Fluidized Bed Reactor,”Chem. Eng. Sci.,41, 171 (1983).
Zhang, J., Ayusawa, T., Minagawa, M., Matsuoka, M. and Anpo, M., “Investigation of TiO2 Photocatalysts for the Decomposition of NO in the Flow System,”J. Catal.,198, 1, (2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is dedicated to Professor Dong Sup Doh on the occasion of his retirement from Korea University.
Rights and permissions
About this article
Cite this article
Lim, TH., Kim, SD. Photocatalytic degradation of trichloroethylene over TiO2/SiO2 in an annulus fluidized bed reactor. Korean J. Chem. Eng. 19, 1072–1077 (2002). https://doi.org/10.1007/BF02707235
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707235