Abstract
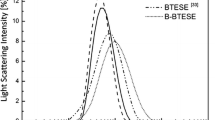
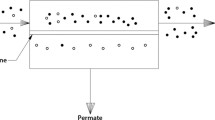
Microporous silica membranes for hydrogen separation were prepared on a γ-alumina coated α-alumina tube by sol-gel method. The reactants of sol-gel chemistry were tetraethoxysilane (TEOS) and methacryloxypropyl-trimethoxysilane (MOTMS). The silane coupling agent, MOTMS, was added as a template in order to control the pore structure to the silicon alkoxide, TEOS. In particular, the microporous membranes were prepared by changing the molar ratio of MOTMS with respect to other substances, and their pore characteristics were analyzed. Then, the effects of thermal treatment on the micropore structure of the resulting silica membranes were investigated. The pore size of the silica membrane prepared after calcination at 400–700 ‡C was in the range of 0.6–0.7 nm. In addition, permeation rates through the membranes were measured in the range of 100–300 dgC using H2, CO2, N2, CH4, C2H6, C3H6 and SF6. The membrane calcined at 600 ‡C showed a H2 permeance of 2×10-7-7×10-7 molm-2s-1Pa-1 at permeation temperature 300 ‡C, and the separation factors for equimolar gas mixtures were 11 and 36 for a H2/CO2 mixture and 54 and 132 for a H2/CH4 mixture at permeation temperatures of 100 ‡C and 300 ‡C, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Asaeda, M. and Kitao, S., “Inorganic Membranes”, Proc. 2nd Intern. Conf. on Inorganic Membranes, 295 (1991).
Bertoluzza, A. Fangnano, C., Morelli, M. A., “Raman and Infrared Spectra on Silica Gel Evoluting Toward Glass”,J. Non-Cryst. Solids,48, 117 (1982).
Brinker, C. J., Sehgal, R., Hietala, S. L., Deshpande, R., Smith, D. M., Loy, D. and Ashley, C. S., “Sol-gel Strategies for Controlled Porosity Inorganic Materials, Alex Stern Honorary Symposium on Membrane-Based Gas Separations”,J. Membr. Sci.,94 85 (1994).
Burggraaf, A. J. and Cot, L., “Fundamentals of Inorganic Membrane Science and Technology”, Elsevier, Amsterdam (1996).
Cho, Y. K., Han, K. W. and Lee, K. H., “Separation of CO2 by Modified γ-A12O3 Membranes at High Temperature”,J. Membr. Sci.,104, 219 (1995).
de Vos, R. M. and Verweij, H., “Improved Performance of Silica Membranes for Gas Separation”,J. Membr. Sci.,143, 37 (1998).
de Vos, R. M., Maier, W. F. and Verweij, H., “Hydrophobie Silica Membranes for Gas Separation”,J. Membr. Sci.,158, 277 (1999).
Jung, K. Y., So, J. H., Park, S. B. and Yang, S. M., “Hydrogen Separation from the H2/N2 Mixture by using a Single and Multi Stage Inorganic Membrane”,Korean J. Chem. Eng.,16, 193 (1999).
Kim, H. S., Han, J. W., Chun, K. Y., Shul, Y. G. and Joe, Y. I., “Synthesis of Organic-inorganic Composite Membrane by Sol-gel Process”,Korean J. Chem. Eng.,12, 405 (1995).
Kim, Y. S., Kusakabe, K., Morooka, S. and Yang, S. M., “Characterization of Silica Membranes formed by a Sol-gel Technique with Organic Templating Reagents”,J. Membr. Sci., submitted.
Kusakabe, K., Sakamoto, S., Saie, T. and Morooka, S., “Pore Structure of Silica Membranes formed by a Sol-gel Technique using Tetraethoxysilane and Alkyltriethosysilanes”,Sep. & Purif. Tech.,16, 139 (1999).
Labot, A., Julbe, A., Guizard, C. and Cot, L., “Silica Membranes by the Sol-gel Process”,J. Membr. Sci.,44, 289 (1989).
Lee, K. H., Cho, Y. K. and Han, K. W., “Manufacture of Alumina Composite Membranes for CO2 Separation”,HWAHAK KONGHAK,33, 570 (1995c).
Lee, S. J., Cho, I. H., Kim, K. Y., Yang, S. M. and Park, S. B., “Preparation and Characterization of Pd/Al2O3 Composite Membrane Supported on Porous Alumina Tube by Sol-gel Method”,HWAHAK KONGHAK,33, 29 (1995a).
Lee, S. J., Yang, S. M. and Park, S. B., “Synthesis of Palladium Impregnated Alumina Membrane for Hydrogen Separation”,J. Membr. Sci.,96, 223 (1994).
Lee, S. Y., Lee, S. J., Kwon, S. J., Yang, S. M. and Park, S. B., “Preparation of Sol-gel Driven Alumina Membrane Modified by Soaking and Vapor Deposition Method”,J. Membr. Sci.,108, 97 (1995b).
Raman, N. K. and Brinker, C. J., “Organic Template Approach to Molecular Sieving Silica Membranes”,J. Membr. Sci.,105, 273 (1995).
Roh, H. S., Chang, J. S. and Park, S. E., “Synthesis of Mesoporous Silica in Acidic Condition by Solvent Evaporation Method”,Korean J. Chem. Eng.,16, 331 (1999).
Sarocco, G., Versteeg, G. F. and van Swaaij, W. P. M., “Current Hurdles to the Success of High Temperature Membrane Reactors”,J. Membr. Sci.,95, 105 (1994).
So, J. H., Yang, S. M. and Park, S. B., “Preparation of Silica-alumina Composite Membranes for Hydrogen Separation by Multi-step Modification”,J. Membr. Sci.,147, 147 (1998).
So, J. H., Yoon, K. Y., Yang, S. M. and Park, S. B., “Preparations of Metal Impregnated Porous Inorganic Membrane for Hydrogen Separation by Multi-step Pore Modification”,Korean J. Chem. Eng.,16(2), 193 (1999).
Tsapatsis, M. and Gavalas, G., “Structure and Aging Characteristics of H2-permselective SiO2-Vycor Membranes”,J. Membr. Sci.,87, 281 (1994).
Tsapatsis, M., Kim, S. J., Nam, S. W. and Gavalas, G., “Synthesis of Hydrogen Permselective SiO2, TiO2, A12O3 and B2O3 Membranes from Chloride Precursors”,Ind. Chem. Res.,30, 2152 (1991).
Uhlhorm, R. J. R., Keizer, K. and Burrggaaf, A. J., “Gas and Surface Diffusion in Modified γ-alumina Systems”J. Membr. Sci.,46, 225 (1989).
Yamasaki, A. and Inoue, H., “Surface Diffusion of Organic Vapor Mixtures Through Porous Glass”,J. Membr. Sci.,59, 233 (1991).
Yan, S., Maeda, H., Kusakabe, K., Morooka, S. and Akiyama, Y., “Hydrogen-selective SiO2 Membrane formed in Pores of Alumina Support Tube by Chemical Vapor Deposition with Tetraethylorthosilicate”,Ind. Eng. Chem. Res.,33, 2696 (1994).
Yoldas, B. E., “Alumina Sol Preparation from Alkoxide”,J. Membr. Sci.,10, 1956 (1975).
Yoldas, B. E., “Hydrolysis of Aluminium Alkoxides and Bayerite Conversion”,J. appl. Chem. Biotechnol.,23, 803 (1973).
Zaman, J. and Chakma, A., “Inorganic Membrane Reactors”,J. Membr. Sci.,92, 1 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, YS., Kusakabe, K., Morooka, S. et al. Preparation of microporous silica membranes for gas separation. Korean J. Chem. Eng. 18, 106–112 (2001). https://doi.org/10.1007/BF02707206
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707206