Abstract
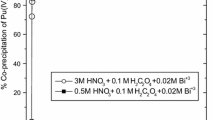
This work has been performed as a part of the partitioning of minor actinides. Minor actinides can be recovered from high-level wastes as oxalate precipitates, but they tend to be co-precipitated together with lanthanide oxalates. This requires another partitioning step for mutual separation of actinide and lanthanide groups. Accordingly, the objective of this study was to decompose and dissolve oxalate precipitates into a dilute nitric acid solution by using a photochemical reaction. In order to do this, oxalic acid and neodymium oxalate precipitate were used in this study. Neodymium oxalate was chosen as a stand-in element representing americium, curium and lanthanides. As a result, decomposition characteristics of oxalic acid were first investigated and then on the basis of these results, the decomposition of neodymium oxalate precipitates was evaluated. From results using oxalic acid, the oxalate decomposition appeared to take place due to the reaction between the oxalate ion and hydroxyl radical generated from the nitric acid by photo-radiation. And the oxalate decomposition rate was measured in the experiments for various nitric acid contents when a mercury lamp (λ=254 nm) was used as a light source. The maximum decomposition rate was obtained when the nitric acid concentration was around 0.5 M, while the decomposition rate was reduced with an increase in the nitric acid concentration at more than 0.5 M. The photo-decomposition rate of neodymium oxalate precipitates was found to be 0.0034 M/h at the condition of 0.5 M HNO3.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Daniels, M., Meyers, R. V. and Belardo, E. V., “Photochemistry of the Aqueous Nitrate System I. Excitation in the 300-μm Band,”J. Phys. Chem.,72(2), 389 (1968).
Forsberg, C. W., “Separation of Americium. Curium, and Trivalent Lanthanides from High-Level Wastes by Oxalate Precipitation: Experiments with Synthetic Waste Solutions,”Nucl. Tech.,49, 243 (1980).
IAEA Technical Reports Series No. 214, “Evaluation of Actinide Partitioning and Transmutation,” IAEA, Vienna (1982).
Jenkins, I. L., Moore, F. H. and Waterman, M. J., “X-ray Powder Crystallographic Data on Plutonium and Other Oxalates-1,”J. Inorg. Nucl. Chem.,27, 77 (1965).
Kim, E. H., “Application of New Oxalate Precipitation Process to Partitioning of Long-Lived Nuclides from High Level Liquid Waste,” Submitted toNucl. Tech. (1998b).
Kim, E. H., Chung, D. Y., Kim, W. H., Shin, Y. J., Lee, E. H., Yoo, J. H. and Choi, C. S., “Neptunium Oxalate Precipitation from the Simulated Radioactive Liquid Waste,”J. Nucl. Sci. and Tech.,34, 283 (1997).
Kim, E. H., “Dissolution of Oxalate Precipitate and Destruction of Oxalate Ion by Hydrogen Peroxide in Nitric Acid Solution,” Submitted toJ. Nucl. Sci. and Tech. (1998a).
Kim, E. H., Kim, Y. H., Chung, D. Y., Yoo, J. H. and Choi, C. S., “Effect of H2O2 on Photodestruction of Oxalic Acid,”HWAHAK KONGHAK,35, 440 (1997). Kim, E. H., Shin, Y. J., Kim, W. H., Chung, D. Y., Kim, S. S.,
Yoo, J. H. and Choi, C. S., “Partitioning of Neodymium and Americium in the Liquid Radioactive Waste by Oxalate Precipitation,”Korean J. Chem. Eng.,12, 557 (1995).
Rankin, D. T., Burney, G. A., Smith, P. K. and Sisson, R. D. Jr., “Preparation and Characterization of Oxalate-Based237NpO2 Powder,”Ceramic Bulletin,56, 478 (1977).
Sarakha, M., Boule, P. and Lenoir, D., “Phototransformation of 2-Phenylphenol Induced in Aqueous Solution by Excitation of Nitrate Ions,”J. Photochem. Photobiol. A: Chem.,75, 61 (1993).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, EH., Chung, DY., Kwon, SW. et al. Photochemical decomposition of oxalate precipitates in nitric acid medium. Korean J. Chem. Eng. 16, 351–356 (1999). https://doi.org/10.1007/BF02707124
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707124