Abstract
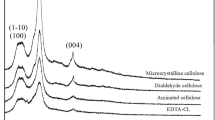
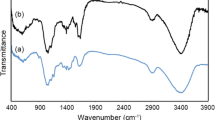
Insoluble cellulose xanthate (ICX), O-alkyl dithiocarbonate of cellulose, was synthesized and used for the removal of heavy metal ions from aqueous solutions. ICX possessed carbon disulfide (CS2) as a functional group, which was obtained by esterification of the hydroxyl group on cellulose polymer matrix with CS2. CS2 could form complexes with divalent transition metal ions, and the resulting ICX-metal complex was water-insoluble and settled easily. The optimum composition for ICX synthesis was cellulose/NaOH/CS2=1: 2: 2.3 in mole base, which showed the highest removal capacity. The selectivity and binding capacity of ICX for heavy metals were in the order of Pb2+> Cd2+> Ni2+ and Cd2+> Pb2+> Ni2+, respectively. More than 90 % of the initial amount of heavy metals was removed within 30 min. The optimum pH was neutral or slightly alkaline, and more than 40 % of initial heavy metals was removed even in the acidic range of pH 2 to 3.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bio-Rad Chemical Division, “Guide to Ion Exchange,” Catalog Number 140-9997, Richmond, California (1997).
Campanella, L., Cardarelli, E., Ferri, T. and Petronio, B.M., “Mercury Removal from Petrochemical Wastes,”Wat. Res.,20 (1), 63 (1986).
Helfferich, F., “Ion Exchange,”McGraw-Hill, New York (1962).
Jawed, M. and Tare, V., “Application of Starch Xanthates for Cadmium Removal: A Comparative Evaluation,”J. Appl. Polym. Sci.,42, 317 (1991).
Kim, H. and Lee, K., “Application of Cellulose Xanthate for the Removal of Nickel Ion from Aqueous Solution,”J. Kor. Soc. Environ. Eng.,20(2), 247 (1998).
Kim, J. W., Lalvani, S. B. and Akash, B. A., “Coliquefaction of Coal and Black Liquor,”Korean J. Chem. Eng.,12, 582 (1995).
Marani, D., Mezzana, M., Passino, R. and Tiravanti, G., “Starch Xanthate and Polycations in Heavy Metal Removal from Wastewaters,”Environ. Technol. Lett.,1, 141 (1980).
Matis, K. A. and Mavros, P., “Recovery of Metals by Ion Flotation from Dilute Aqueous Solutions,”Sep. Purif. Meth.,20(1), 1 (1991).
Mazzi, G., Marani, D., Majone, M. and Coretti, M. R., “Optimization of Mercury Removal from Chloralkali Industrial Wastewater by Starch Xanthate,”Environ. Technol. Lett.,6, 369 (1985).
Morrison, G. H. and Freiser, H., “Solvent Extraction in Analytical Chemistry,” John Wiley & Sons, New York (1957).
Noyes, R., “Unit Operations in Environmental Engineering,” Noyes, NJ (1994).
Peters, R. W., Ku, Y. and Bhattacharyya, D., “Evaluation of Recent Treatment Techniques for Removal of Heavy Metals from Industrial Wastewaters,”AIChE Symp. Ser.,81(243), 165 (1985).
Shreve, R. N. and Brink, J. A., “Chemical Process Industries,” 4th Ed., McGraw-Hill, NY (1977).
Swanson, C. L., Naffziger, T. R., Russel, C. R., Hofreiter, B. T. and Rist, C. E., “Xanthation of Starch by a Continuous Process,”I & EC Prod. Res. Dev.,3(1), 22 (1961).
Swanson, C. L., Wing, R. E., Doane, W. M. and Russel, C. R., “Mercury Removal from Waste Water with Starch Xanthates-Cationic Polymer Complex,”Environ. Sci. Technol.,7(7), 614 (1973).
Tare, V. and Chaudhari, S., “Evaluation of Soluble and Insoluble Xanthate Process for the Removal of Heavy Metals from Wastewater,”Wat. Res.,21(9), 1109 (1987).
Wing, R. E. and Doane, W. M., US Patent, No. 4,083,783 (1978).
Wing, R. E., Doane, W. M. and Russel, C. R., “Insoluble Starch Xanthates: Use in Heavy Metal Removal,”J. Appl. Polym. Sci.,19, 847 (1975).
Wing, R. E., “Dissolved Heavy-Metal Removal by Insoluble Starch Xanthate,”Environ. Progr.,2(4), 269 (1983).
Zeng, H., Li, W. and Li, Z., “Studies on a New Water-Soluble Sulfur-Containing Cellulose Derivative,”J. Appl. Polym. Sci.,54, 1989 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, HT., Lee, K. Application of insoluble cellulose xanthate for the removal of heavy metals from aqueous solution. Korean J. Chem. Eng. 16, 298–302 (1999). https://doi.org/10.1007/BF02707116
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707116