Abstract
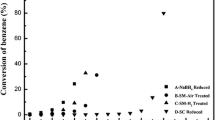
Pt catalysts supported on silicoaluminophosphate molecular sieves were synthesized by adding platinum salts directly to the gel mixture of SAPO. The synthesized products were marked as PtSAPO-5, in order to distinguish with the SAPO-5 supported platinum catalyst prepared by impregnation, which was marked as Pt/SAPO-5. The as-synthesized and calcined PtSAPO-5 samples were characterized by XRD,29Si MAS NMR,129XeNMR and NH3-TPD, and their catalytic activities were evaluated by the hydroisomerization of n-hexane. The results revealed that in PtSAPO-5, the platinum could catalyze the removal of the templates entrapped in the as-synthesized samples. The presence of Pt in the gel mixture can increase silicon content in the framework of SAPO-5, and the acid sites. In the activated PtSAPO-5 catalyst, Pt was highly dispersed in the channel of SAPO-5 molecular sieve. Pt exists in the form of big particles outside of the channel of Pt/SAPO-5 prepared by impregnating method, and impregnation with H2PtCl6 improved the strength of acid sites. PtSAPO-5 has a higher activity and selectivity than Pt/SAPO-5 in the hydroisomerization of n-hexane. This is because the former has medium acid sites and high dispersion of Pt, while the channel of Pt/SAPO-5 is somewhat retarded by the big platinum particles, and the acid sites are stronger than the former.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Blackwell, C. S. and Patton, R. L., “Solid State NMR of Silicoaluminophosphate Molecular Sieves and Aluminophosphate Materials”,J. Phys. Chem.,92, 3095 (1988).
Campelo, J. M., Lafont, F. and Marinas, J. M., “Hydroisomerization and Hydrocracking of n-Hexane on Pt/SAPO-5 and Pt/SAPO-11 Catalysts”,Zeolites,15, 97 (1995).
Das, J., Satyanaryana, C. V. V., Chakrabarty, D. K., Piramanayagam, S. N. and Shrigle, S. N., “Substitution of Al in the AlPO4-5 and AIPO4-H Framework by Si and Fe: A Study of Moessbauer, Magi-angle Spinning Nuclear Magnetic Resonance and Electron Paramagnetic Resonance Spectroscopes and Chemical Probes”,J. Chem. Soc. Faraday Trans.,88, 3255 (1992).
Du, Y.-r. and Yang, R.-h., “Characterization of Catalyst Structure and the Molecules Adsorbed on It Surface by Solid State NMR Spectroscopy”,China J. Petrochemical Engineering,20, 718 (1991).
Lok, B.M., Messina, C.A., Patton, R.L., Richard, T.G., Cannan, T. R. and Flanigen, E. M., “Silicoaluminophosphate Molecular Sieves: Another New Class of Microporous Crystalline Inorganic Solids”,J. Am. Chem. Soc.,106, 6092 (1984).
Ojo, A. F., Dwyer, J., Dewing, J., O’Malley, P. J. and Nabhan, A., “Synthesis and Properties of SAPO-5 Molecular Sieves, Silicon Incorporation into the Framework”,J. Chem. Soc. Faraday Trans.,88, 105 (1992).
Wang, R., Lin, C.F., Ho, Y.S., Leu, L.J. and Chao, K.J., “Silicon Species in a SAPO-5 Molecular Sieve”,Applied Catalysis,72, 39 (1991).
Xiao, T.-c., An, L.-d. and Wang, H.-I., “Characterization and Catalytic Performance of PdSAPO-5 Molecular Sieve”,Stud. Surf. Sci. Catal.,97, 351 (1995).
Xiao, T.-c., “Synthesis, Characterization and Catalytic Performance of SAPO-5 and Its Supported Palladium Catalysts, Ph. D Thesis, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences, 1993.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xiao, TC., Wang, HT., Lu, YL. et al. Nature and catalytic performance of PtSAPO-5 molecular sieve catalyst synthesized by one step. Korean J. Chem. Eng. 15, 505–509 (1998). https://doi.org/10.1007/BF02707100
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707100