Abstract
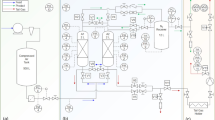
The effects of pressure drop on the dynamics of fixed-beds were theoretically studied. The system used was an H2/CO mixture (70 vol% H2, 30 vol% CO) in zeolite 5A. The pressure drop at the pressurization step affected the breakthrough time at an adsorption step in the PSA process. As a result, the combined effects of pressure drop during adsorption and pressurization steps led to earlier breakthrough compared to the case without a pressure drop. The effect of pressure drop at the adsorption step under the non-isothermal condition was slightly larger than that under the adiabatic condition. In the case of pressurization and blowdown steps with large pressure drop, the flow pattern near the open end during a short period of time had to be explained by the Ergun equation instead of Darcy’s law. However, there was only a slight difference in the results of a multi-bed PSA process depending on whether or not the pressure drops at the pressurization/depressurization steps as well as at the adsorption step were considered.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Buzanowski, M. A., Yang, R. T. and Haas, O. W., “Direct Observation of the Effects of Bed Pressure Drop on Adsorption and Desorption Dynamics“,Chem. Eng. Sci.,44, 2392 (1989).
Doong, S. J. and Yang, R. T., “The Role of Pressure Drop in Pressure Swing Adsorption“,AIChE Symp. Ser.,264, 84, 145 (1988).
Hart, J., Battram, M. J. and Thomas, W. J., “Axial Pressure Gradients During the Pressurization and Depressurization Steps of a PSA Gas Separation Cycle“,Gas Sep. Purif.,4, 97 (1990).
Han, S. and Lee, H., “A Study on Adsorption of H2/CO Mixture at Elevated Pressure”,HWAHAK KONGHAK,33, 720 (1995).
Han, S., Yang, J., Lee, C.-H. and Lee, H., “A Study on Separation of H2/CO Mixture by One-column PSA Process”,HWAHAK KONGHAK,34, 277 (1996).
Kikkinides, E. S. and Yang, R. T., “Effects of Bed Pressure Drop on Isothermal and Adiabatic Adsorber Dynamics”,Chem. Eng. Sci.,48, 1545 (1993).
Kim, W.-G., Yang, J., Han, S., Cho, C, Lee, C.-H. and Lee, H., “Experimental and Theoretical Study on H2/CO2 Separation by a Five-Step One-Column PSA Process”,Korean J. Chem. Eng.,12, 503 (1995).
Lu, Z. P., Loureiro, J., Le Van, D., Rodrigues, A., “Pressurization and Blowdown of Adsorption Beds: II. Effect of the Momentum and Equilibrium Relations on Isothermal Operation”,Chem. Eng. Sci.,48, 1699 (1992a).
Lu, Z. P., Loureiro, J., LeVan, D. and Rodrigues, A., “Dynamics of Pressurization and Blowdown of an Adiabatic Bed: III”,Gas Sep. Purif.,6, 15 (1992b).
Rodrigues, A. E., Loureiro, J. M. and Le Van, M. D., “Simulated Pressurization of Adsorption Beds”,Gas Sep. Purif.,5, 115 (1991).
Ruthven, D. M., Farooq, S. and Knaebel, K. S., “Pressure Swing Adsorption”, VCH, New York, 1994.
Sereno, C. and Rodrigues, A., “Can Steady-state Momentum Equations be Used in Modeling Pressurization of Adsorption Beds?”,Gas Sep. Purif,7, 167 (1993).
Sundaram, N. and Wankat, P. C., “Pressure Drop Effects in the Pressurization and Blowdown Steps of Pressure Swing Adsorption”,Chem. Eng. Sci.,43, 123 (1988).
Yang, R. T., “Gas Separation by Adsorption Process”, Butterworths, Boston, 1987.
Yang, J., Cho, C, Baek, K. H. and Lee, C.-H., “Comparison of One-bed and Two-bed H2 PSA Using Zeolite 5A”,HWAHAK KONGHAK,35, 545 (1997a).
Yang, J., Chang, J.-W. and Lee, C.-H., “Separation of Hydrogen Mixtures by a Two-Bed Pressure Swing Adsorption Process Using Zeolite 5A”,Ind. Eng. Chem. Res.,36, 2789 (1997b).
Yang, J., Han, S., Cho, C, Lee, C. H. and Lee, H., “Bulk Separation of Hydrogen Mixtures by a One-column PSA Process”,Separations Technology,5, 239 (1995).
Zhong, G. M., Meunier, F., Huberson, S. and Chalfen, J. B., “Pressurization of a Single-component Gas in an Adsorption Column”,Chem. Eng. Sci.,47, 543 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, J., Park, MW., Chang, JW. et al. Effects of pressure drop in a PSA process. Korean J. Chem. Eng. 15, 211–216 (1998). https://doi.org/10.1007/BF02707074
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707074