Abstract
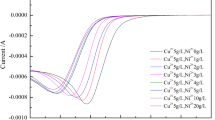
Optimization of reduction potential for electroseparation was studied for the recovery of gold, copper, and lead from acidic solution. A linear sweep voltammetric method enabled us to determine characteristic reduction potentials for each metal and the kinetics of the metal deposition indicated by current-voltage curves. In order to precipitate the metal species sequentially, reduction potentials were examined for the individual and mixed solutions of Au(III), Cu(II), and Pb(II). The three metals were reasonably well isolated from the mixed solutions such as Cu(II)/ Pb(II) and Au(III)/Cu(II)/Pb(II) in the order of the corresponding reduction potentials, in particular, the mass transfer controlled reduction potentials, obtained from linear sweep voltammetry (LSV) measurement.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Armstrong, R. D., Todd, M., Atkinson, J. W. and Scott, K., “Electroseparation of Cobalt and Nickel from a Simulated Wastewater”,J. Appl. Electrochem.,27, 965 (1997).
Armstrong, R. D., Todd, M., Atkinson, J. W. and Scott, K., “Selective Electrodeposition of Metals from Simulated Waste Solutions”,J. Appl. Electrochem.,26, 379 (1996).
Bard, A. J. and Faulkner, L. R, “Electrochemical Methods Fundamentals and Applications”, John Wiley & Sons, Inc., New York (1980).
Bernardes, A., Bohlinger, I., Rodriguez, D., Milbrandt, H. and Wuth, W, “Recycling of Printed Circuit Boards by Smelting with Oxidizing/Reducing Top Blowing Process”, Proceedings of the EPD Congress, Orlando, FL, Feb. 9–13 (1997).
Chang, J. S. and Huang, J. C, “Selective Adsorption/Recovery of Pb, Cu, and Cu with Multiple Fixed Beds Containing Immobilized Bacterial Biomass”,Biotechnol. Prog.,14, 735 (1998).
Dean, J. A., “Langes Handbook of Chemistry, 40th Ed.”, McGraw-Hill, Inc., New York (1992).
Fresner, J. and Schnitzer, H., “Lessons from 13 Waste Minimization Case Studies in Styria”, Proceedings of the 7th ISWA International Congress and Exhibition-Volume 2, Yokohama, Japan, Oct. 27–Nov. 1 (1996).
Fu, J., Nakamura, S. and Akiba, K., “Separation and Recovery of Gold, Platinum and Palladium by a Trioctylamine Liquid Membrane”,Anal Sci., 11,149 (1995).
Juang, R. S., Wang, S. W and Lin, L. C, “Simultaneous Recovery of EDTA and Lead(II) from Their Chelated Solutions Using a Cation Exchange Membrane,J. Membr. Sci.,160, 225 (1999).
Kim, K. and Cho, J. W, “Selective Recovery of Metals from Spent Desulfurization Catalyst”,Korean J. Chem. Eng., 14,162 (1997).
Kim, S. I., Jeung S. Y and Moon, H., “Removal and Recovery of Heavy Metal Ions in Fixed and Semi-Fluidized Beds”,Korean J. Chem. Eng.,15, 637 (1998).
Pesic, B. and Zhou, T., “Recovery and Separation of Nickel and Cobalt from Cobaltite and Siegenite Leach Solutions with a Pyridine-based Extradant Mixture”, Proceedings of the EPD Congress, Orlando, FL, Feb. 9–13(1997).
Pletcher, D. and Walsh, F. C, “Industrial Electrochemistry 2nd Ed.”, CHAMPMAN AND HALL, London (1990).
Raubenheimer, H. G. and Cronje, S., “Gold: Progress in Chemistry, Biochemistry, and Technology”, John Wiely & Sons, Inc., New York (1999).
Ringbom, “Complexation in Analytical Chemistry”, Interscience Publishers (1963).
Scott, K., “Electrochemical Processes for Clean Technology”, The Royal Society of Chemistry, Cambridge (1995).
Snoeyink, V. L. and Jenkins, D., “Water Chemistry”, John Wiley & Sons, Inc., New York (1980).
Voropanova, L. A. and Velichko, L. N., “Recovery of A1(III), Cu(II), and Cr(III) Ions and Dispersed and Emulsified Oils from Industrial Wastewater”,Russian J. Appl. Chem.,72, 469 (1999).
Walsh, F. C. and Gabe, D. R., “Controlled-potential Electrodeposition of Metals at a Rotating Cylinder Electrode”,Surface Technol.,12, 25 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Park, JS., Moon†, SH. Use of cascade reduction potential for selective precipitation of Au, Cu, and Pb in hydrochloric acid solution. Korean J. Chem. Eng. 19, 797–802 (2002). https://doi.org/10.1007/BF02706970
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706970