Abstract
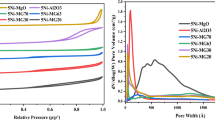
Partial oxidation of methane (POM) was systematically investigated in a fixed bed reactor over 12 wt% Ni catalysts supported on α-A12O3, γ-A12O3 and θ-A12O3 which were prepared at different conditions. Results indicate that the catalytic activity toward POM strongly depends on the BET surface area of the support. All the Ni/ θ-Al2O3 catalysts showed high activity toward POM due to the less formation of inactive NiAl2O4 species, the existence of NiO, species and stable θ-Al2O3 phase. Although Ni/γ-Al2O3 showed the highest activity toward POM, long-time stability cannot be expected as a result of the deterioration of the support at higher temperature, which is revealed from BET results. From the reaction and characterization results, it is inferred that the optimal conditions for the preparation of θ-Al2O3 are 1,173 K and 12 h.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ashcroft, A. T, Cheetham, A. K., Green, M. L. H. and Vernon, P. D.F, “Partial Oxidation of Methane to Synthesis Gas Using Carbon Dioxide,Nature,352, 225 (1991).
Au, C. T., Wang, H. Y. and Wan, H. L, “Mechanistic Studies of CH4/O2 Conversion over SiO2-Supported Nickel and Copper Catalysts”,J. Catal.,158, 343 (1996).
Chang, J.-S., Park, S.-E., Roh, H.-S. and Park, Y.-K., “Thermogravimetric Study on Reactivity of Carbon Dioxide and Methane over Supported Nickel Reforming Catalysts”,Bull. Korean Chem. Soc,19, 809 (1998).
Choudhary, V. R., Rajput, A. M. and Probhaker, B., “Nonequilibrium Oxidative Conversion of Methane to CO and H2 with High Selectivity and Productivity over Ni/Al2O3 at Low Temperatures”,J. Catal.,139, 326(1993).
Diskin, A. M., Cunningham, R H. and Qrmerod, R. M., “The Oxidative Chemistry of Methane over Supported Nickel Catalysts”,Catal. Today,46, 147 (1998).
Dong, W.-S, Roh, H.-S., Liu, Z.-W, Jun, K.-W and Park, S.-E., “Hydrogen Production from Methane Reforming Reactions over NiO/ MgO Catalyst”,Bull. Korean Chem. Soc,22, 1323 (2001).
Hickman, D. A. and Schmidt, L. D., “Production of Syngas by Direct Catalytic Oxidation of Methane”,Science,259, 343 (1993).
Hu, Y. H. and Ruckenstein, E., “Transient Kinetic Studies of Partial Oxidation of CH4”,J. Catal,158, 260 (1996).
Jung, K., Coh, B.-Y and Lee, H.-I, “Ni/Al2O3 Catalyst Prepared by Liquid Phase Oxidation for Carbon Dioxide Reforming of Methane”,Bull. Korean Chem. Soc,20, 89 (1999).
Looij, F. V. and Geus, J. W, “Nature of the Active Phase of aNickel Catalyst during the Partial Oxidation of Methane to Synthesis Gas”,J. Catal,168, 154 (1997).
Lu, Y, Liu, Y. and Shen, S., “Design of Stable Ni Catalysts for Partial Oxidation of Methane to Synthesis Gas”,J. Catal,177, 386 (1998a).
Lu, Y, Xue, J., Yu, C, Liu, Y and Shen, S., “Mechanistic Investigations on the Partial Oxidation of Methane to Synthesis Gas over a Nickelon-Alumina Catalyst”,Appl. Catal. A,174, 121 (1998b).
Miao, Q., Xiong, G., Sheng, S., Xu, L., Cui, W and Guo, X., “Partial Oxidation of Methane to Syngas over Nickel-Based Catalysts Modified by Alkali Metal Oxide and Rare Earth Metal Oxide”,Appl. Catal. A,154, 17 (1997).
Nakagawa, K., Anzai, K., Matsui, N., Ikenaga, N., Suzuke, T., Teng, Y, Kobayashi, T. and Haruta, M., “Effect of Support on the Conversion of Methane to Synthesis Gas over Supported Iridium Catalysts”,Catal. Lett,51, 163 (1998).
Nam, S.-W, Yoon, S.-P, Ha, H.-Y, Hong, S.-A and Maganyuk, A.-P, “Methane Steam Reforming in a Pd-Ru Membrane Reactor”,Korean J. Chem. Eng.,17, 288 (2000).
Peña, M. A, Gómez, J. P. and Fierro, J. L. G., “New Catalytic Routes for Syngas and Hydrogen Production”,Appl. Catal A,144, 7 (1996).
Roh, H.-S, Dong, W.-S., Jun, K.-W. and Park, S.-E, “Partial Oxidation of Methane over Ni Catalysts Supported on Ce-ZrO2 Mixed Oxide”,Chem. Lett, 88 (2001a).
Roh, H.-S, Jun, K.-W, Dong, W.-S, Park, S.-E. and Baek Y.-S, “Highly Stable Ni Catalyst Supported on Ce-ZrO2 for Oxy-Steam Reforming of Methane,Catal. Lett,74, 31 (2001b).
Roh, H.-S, Jun, K.-W, Dong, W.-S, Park, S.-E. and Joe, Y-I, “Partial Oxidation of Methane over Ni/θ-Al2O3 Catalysts”, Chem. Lett., 666 (2001c).
Roh, H.-S, “Catalytic Behavior of Supported Nickel Catalysts for Methane Reforming Reactions, Ph. D. Dissertation, Yonsei University (2001 d).
Ruckenstein, E. and Hu, Y H, “Methane Partial Oxidation over NiO/ MgO Solid Solution Catalysts”,Appl. Catal. A,183, 85 (1999b).
Takeguchi, T, Furukawa, S. and Inoue, M, “Hydrogen Spillover from NiO to the Large Surface Area CeO2-ZrO2 Solid Solutions and Activity of the NiO/CeO2-ZrO2 Catalysts for Partial Oxidation of Methane”,J. Catal,202, 14 (2001).
Tang, S, Lin, J. and Tan, K. L, “Partial Oxidation of Methane to Syngas over Ni/MgO, Ni/CaO andNi/CeO2”,Catal. Lett.,51, 169 (1998).
Tsang, S. C, Claridge, J. B. and Green, M. L. H, “Recent Advances in the Conversion of Methane to Synthesis Gas”,Catal. Today,23, 3 (1995).
Wang, H. Y and Ruckenstein, E, “Partial Oxidation of Methane to Synthesis Gas over MgO- and SiO2-Supported Rhodium Catalysts”,J. Catal,186, 181 (1999a).
Zhu, J, Zhang, D. and King, K. D, “Reforming of CH4 by Partial Oxidation: Thermodynamic and Kinetic Analyses”,Fuel,80, 899 (2001).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, ZW., Jun, KW., Roh, HS. et al. Partial oxidation of methane over nickel catalysts supported on various aluminas. Korean J. Chem. Eng. 19, 735–741 (2002). https://doi.org/10.1007/BF02706961
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706961