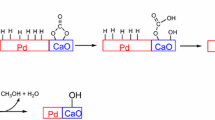
Abstract
The main goal of this contribution was to study the probability of chain growth of coke on metal sites and on support sites for hexane dehydrogenation. The coke structure of the catalysts examined by IR was found to have the aromatic structure. Soxhlet extraction coupled with GC-14B (DB1 column) analysis was mainly employed for coke composition analysis and determination of the probability of chain growth (alpha value). It was found that the soluble coke was mainly composed of C8–C12 on both sites. Interestingly, the probabilities of chain growth on both sites were identical. However, the extracted coke on the metal site was more easily removable and had lower carbon numbers than that on the support site. Moreover, the addition of promoter, especially of K promoter, was sensitive to inhibit the probability of chain growth, resulting in the reduction of the amount of coke.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barbier, I, “Coking of Reforming Catalysts,”Catalyst Deactivation, 1 (1987).
Biswas, J., Bickle, G. M. and Gray, P. G., “The Role of Deposited Poisons and Crystallite Surface in the Activity and Selectivity of Reforming Catalysts,”Catal. Rev.-Sci. Eng.,30,161 (1988).
Biswas, J., Gray, P. G. and Do, D. D., “The Reforming Lineout Phenomenon and its Fundamental Importance to Catalyst Deactivation,”Applied Catalysis,32, 249 (1987).
Bond, G. C, “Heterogeneous Catalysis: Principles and Application,” Clarendon Press: Oxford (1987).
Figoli, N. S., Beltramini, J. N., Martinelli, E. E., Dad, M. R. and Parera, J. M., “Operational Conditions and Coke Formation on Pt-Al2O3 Reforming Catalyst,”Applied Catalysis,5,19 (1983).
Flory, P. I, “Principle of Polymer Chemistry,” Cornell University Press, New York, 317 (1953).
Forzatti, P. and Lietti, L., “Catalyst Deactivation”Catalysis Today,52, 165 (1999).
Froment, G. F., “Coke Formation in Catalytic Process: Kinetics and Catalyst Deactivation”Catalyst deactivation, 53 (1997).
Froment, G. F., “The Modeling of Catalyst Deactivation by Coke Formation,”Catalyst deactivation, 53 (1991).
Gerard, P. V D. L. and Beenackers, A. A. C. M., “Kinetics and Selectivity of the Fischer-Tropsch Synthesis: A Literature Review,”Catal. Rev.-Sci. Eng.,41, 255 (1999).
Hamada, H., “Cooperative of Catalytic Species for the Selective Reduction of Nitrogen Monoxide with Hydrocarbons,”Catalysis Surveys from Japan,1,53 (1997).
Inaba, M., Kintaichi, Y and Hamada, H., “Cooperative Effect of Platinum and Alumina for the Selective Reduction of Nitrogen Monoxide with Propane,”Catalysis Letters,36, 223 (1996).
Lamy-Pitara, E., Ouazzani-Benhima, H., Barbier, J., Cahoreau, M. and Caisso, X, “Platinum Catalyst Modified by Tin”Applied Catalysis A: General,81, 47 (1992).
Lietz, G., Völter, J., Dobrovolszky, M. and Paal, Z., “Initial Changes of the Catalytic Properties of Platinum Containing Catalysts: I. Transformations of Monoand Bimetallic Pt/Al2O3 Catalysts by Carbonaceous Deposits,”Applied Catalysis,13,77 (1984).
Lox, E. S. and Froment, G. F., “Kinetics of the Fischer-Tropsch Reaction on a Precipitated Promoted Iron Catalyst: 2. Kinetic Modeling,”Ind. Eng. Chem. Res.,32, 71 (1993).
Pieck, C. L., Verderone, R J., Jablonski, E. L. and Parera, J. M., “Burning of Coke on Pt-Re/Al2O3 Catalyst Activation Energy and Oxygen Reaction Order,”Applied Catalysis,55,1 (1989).
Radwan, A. M., Kyotani, T. and Tomita, A, “Characterization of Coke Deposited from Cracking of Benzene over USY Zeolite Catalyst,”Applied Catalysis A: General,192, 43 (2000).
Reyniers, G. and Froment, G. F., “Coke Formation in the Thermal Cracking of Hydrocarbons: 4. Modeling of Coke Formation in Naphtha Cracking,”Ind. Eng. Chem. Res.,33, 2548 (1994).
Rostrup-Nielsen, J. R., “Catalytic Steam Reforming,” Berlin: Buchbinderei Bruno Helm (1984).
Rostrup-Nielsen, J. R., “Industrial Relevance of Coking,”Catalysis Today,37,225 (1997).
Sunee, S., Piyasan, P. and Tharathon, M., “Deactivation of the Metal and Acidic Functions for Pt, Pt-Sn and Pt-Sn-K using Physically Mixed Catalysts,”Korean J. Chem. Eng,17, 548 (2000).
Sunee, S., Piyasan, P. and Tharathon, M., “Cooperative Effect of Platinum and Alumina on Catalyst Deactivation for Dehydrogenation Reaction,”Reaction Kinetics and Catalysis Letters,72, 125 (2001).
Trimm, D. L., “Catalyst Design for Reduced Coking,”Applied Catalysis,5, 263 (1983).
Trimm, D. L., “Coke Formation and Minimisation during Steam Reforming Reactions,”Catalysis Today,37,233 (1997).
Van Trimpont, P. A., Marin, G. B. and Froment, G. F., “Kinetics of the Reforming of C7, Hydrocarbons on a Commercial PtRe/Al2O3 Catalyst,”Applied Catalysis,24, 53 (1986).
Vértes, Cs., Tálas, E., Czakó-Nagy, I., Ryczkowski, J., Göbölös, S., Vétes, A. and Margitfalvi, J., “Mössbauer Spectroscopy Studies of Sn-Pt/Al2O3 Catalysts Prepared by Controlled Surface Reactions”Applied Catalysis,68,149 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srihiranpullop, S., Praserthdam, P. Probability of chain growth in coke formation on metals and on supports during catalytic reforming over Pt, Pt-Sn and Pt-Sn-K catalysts mixed physically with Al2O3 . Korean J. Chem. Eng. 20, 1017–1022 (2003). https://doi.org/10.1007/BF02706930
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706930