Abstract
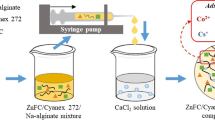
Lead, copper, and cadmium were adsorbed onto calcium alginate beads containing the cell suspension discarded from a brewery. In the cell suspension, there were many cells under lysis. The cell-suspension immobilized beads were prepared by adding 0.6% (w/v) sodium alginate into the cell suspension from the brewery and then making the cell suspension fall dropwise into the swirling 1% (w/v) calcium alginate solution. The dry weight of insoluble solid in the cell suspension was 96 g dry weight/l and the dry density of the bead containing cell suspension was 140 g dry weight/l of the bead. The specific metal uptake of the cell-suspension immobilized bead was 23.7 mg Pb2+, 14.3 mg Cu2+, and 13.4 mg Cd2+/g bead dry weight, respectively. The cell-suspension immobilized beads retained the initial metal-uptake capacity after 20 repeated batches of adsorption and desorption, but the fraction of metal desorbed from the beads by 1 M HCl solution was only 70% of the adsorbed metal. The beads, which had been contained for 14 successive days in the 0.5% (w/v) CaCl2 solution at 4 °C just after 20 cycles of adsorption/desorption, retained the initial metal-uptake capacity after 30 repeated cycles, and more than 90% of the copper and cadmium adsorbed on the beads was desorbed by the 1 M HCl solution.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahn, K. H. and Suh, K. H., “Pb Biosorption bySaccharomyces cerevisiae,”Korean J. Biotechnol. Bioeng.,11, 173 (1996).
Buchholz, K., “Characterization of Immobilized Biocatalysts. In: Dechema Monographs, vol. 84,” Verlag Chemie, Weinheim (1979).
Chen, J., Tendeyong, F. and Yiacoumi, S., “Equilibrium and Kinetic Studies of Copper Ion Uptake by Calcium Alginate,”Environ. Sci. Technol.,31, 1433 (1997).
Choi, S. and Nho, Y. C., “Adsorption of Pb2+, Cu2+ and Co2+ by Polypropylene Fabric and Polyethylene Hollow Fiber Modified by Radiation-Induced Graft Copolymerization,”Korean J. Chem. Eng.,16, 241 (1999).
Harris, P. O. and Ramelow, G. S., “Binding of Metal Ions by Particulate Biomass Derived fromChlorella Vulgaris andScenedesmus Quedricanda,”Env. Sci. Tech.,24, 220 (1990).
Holan, Z. R., Volesky, B. and Prasetyo, I., “Biosorption of Cadmium by Biomass of Marine Algae,”Biotechnol. Bioeng.,41, 819 (1993).
Jin, Y. B. and Park, J. K., “Recovery of Lead using EncapsulatedS. cerevisia,”HWAHAK KONGHAK,36, 229 (1998).
Kim, H. T. and Lee, K., “Application of Insoluble Cellulose Xanthate for the Removal of Heavy Metals from Aqueous Solution,”Korean J. Chem. Eng.,16, 298 (1999).
Kim, J. S., Chah, S. and Yi, J., “Preparation of Modified Silica for Heavy Metal Removal,”Korean J. Chem. Eng.,17, 118 (2000).
Kratochvil, D., Volesky, B. and Demopoulos, G., “Optimizing Cu Removal/Recovery in a Biosorption Column,”Wat. Res.,31, 2327 (1997).
Kuyucak, N. and Volesky, B., “Accumulation of Gold by Algal Biosorbent,”Biorecovery,1, 189 (1989).
Lee, H. S., “Biosorption of Cr, Cu and Al by Sargassum Biomass,”Biotechnol. Bioprocess Eng.,2,126 (1997).
Lee, H. S. and Suh, J. H., “Continuous Biosorption of Heavy Metal Ions by Co-loadedLaminaria japonica in Fixed Bed Column,”Korean J. Chem. Eng.,17, 477 (2000).
Matheickal, J. T., “Biosorption of Heavy Metals from Wastewater Using Macro AlgaeDurvillaea Potatorum andEcklonia Radiata,” Ph.D. thesis, Griffith University, Australia (1998).
Mowll, J. L. and Gadd, G. M., “Zinc Uptake and Toxicity in the YeastsSporobolomyces roseus andSaccharomyces cerevisiae,”J. Gen. Microbiol.,129, 3421 (1983).
Park, J. K., Jin, Y. B. and Park, H. W., “The Recovery of Heavy Metals Using Encapsulated Microbial Cells,”Biotechnol. Bioprocess Eng.,2, 132 (1997).
Park, J. K., Jin, Y. B. and Chang, H. N., “Reusable Biosorbents in Capsules fromZoogloea ramigera Cells for Cadmium Removal,”Biotechnol. Bioeng.,63, 116 (1999).
Park, J. K. and Lee, K. D., “Production of L-Phenylacetylcarbinol (L-PAC) by EncapsulatedSaccharomyces cerevisiae Cells,”Korean J. Chem. Eng.,18, 363 (2001).
Park, J. K., Kim, W. S. and Chang, H. N., “Specific Cd2+uptake of Encapsulated Aureobasidium Pullulans Biosorbents,”Biotechnol. Letts.,23, 1391 (2001).
Schiewer, J. and Volesky, B., “Modeling of Proton-metal Ion Exchange in Biosorption,”Env.. Sci. Tech.,29, 3049 (1995).
Suh, J. H., Kim, D. S., Yun, J. W. and Song, S. K., “Process of Pb2+ Accumulation inSaccharomyces cerevisiae,”Biotechnol. Letts.,20, 153 (1998).
Stoll, A. and Duncan, J. R., “Comparison of the Heavy Metal Sorptive Properties of Three Types of Immobilized, Non-viableSaccharomyces cerevisiae Biomass,”Process Biochem.,32, 467 (1997).
Strandberg, G. W., Shumate, S. E. and Parrot, J. R., “Microbial Cells as Biosorbents for Heavy Metals: Accumulation of Uranium bySaccharomyces cerevisiae andPseudomonas aeruginosa,”Appl. Environ. Microbiol.,41, 237 (1981).
Volesky, B., “Biosorption and Biosorbent,” Biosorption of Heavy Metals, Volesky, B., ed., CRC Press, New York (1990).
Volesky, B., May, H. and Holan, Z. R., “Cadmium Biosorption bySaccharomyces cerevisiae,”Biotechnol. Bioeng.,41, 826 (1993).
Volesky, B. and May-Phillips, H. A., “Biosorption of Heavy Metals bySaccharomyces cerevisiae,”Appl. Microbiol. Biotechnol.,42, 797 (1995).
Wilhelmi, B. S. and Duncan, J. R., “Reusability of ImmobilizedSaccharomyces cerevisiae with Successive Copper Adsorption-Desorption Cycles,”Biotechnol. Letts.,18, 531 (1996).
Yu, Q. and Kaewsarn, P., “A Model for pH Dependent Equilibrium of Heavy Metal Biosorption,”Korean J. Chem. Eng.,16, 753 (1999).
Zhou, J. L. and Kiff, R. J., “The Uptake of Copper from Aqueous Solution by Immobilized Fungal Biomass,”J. Chem. Tech, Biotech.,52, 317 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, J.K., Choi, S.B. Metal recovery using immobilized cell suspension from a brewery. Korean J. Chem. Eng. 19, 68–74 (2002). https://doi.org/10.1007/BF02706876
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706876