Abstract
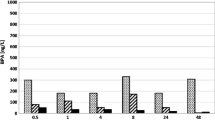
Removal efficiencies of endocrine disrupting chemicals (EDCs), bisphenol A and nonylphenol, during various types of water treatment processes were evaluated extensively using laboratory- and pilot-scale experiments. The specific processes of interest were coagulation/flocculation sedimentation/filtration (conventional water treatment process), powdered activated carbon (PAC), granular activated carbon (GAC), ozonation and chlorination. Batch sorption tests, coagulation tests, and ozone oxidation tests were also performed at higher concentrations with 14 EDCs including bisphenol A. The conventional water treatment process had very low removal efficiencies (0 to 7%) for all the EDCs except DEHP, DBP and DEP that were removed by 53%, 49%, and 46%, respectively. Ozonation at 1 mgO3/ L removed 60% of bisphenol A and 89% of nonylphenol, while chlorination at 1 mg/L removed 58% and 5%, respectively. When ozone and chlorine doses were 4 and 5 mg/L, respectively, both EDCs were not detected. PAC removal efficiencies ranged from 15% to 40% at 3 to 10 mg/L of PAC with a contact time of 15 minutes. In the high concentration batch sorption tests, EDC removal efficiencies by PAC were closely related to octanol-water partition coefficient (Kow). GAC adsorption was very effective water treatment process. The type and service time of GAC did not affect EDC removal efficiencies. The combination of ozonation and GAC in series appears to remove EDCs effectively to safe levels while conventional water treatment could not.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ahel, M., Giger, W. and Koch, M., “Behavior of alkylphenol polyethoxylate surfactants in the aquatic environment — I. Occurrence and transformation in sewage treatment,”Water Res.,28(5), 1131 (1994).
Ahel, M., Molnar, E., Ibric, S. and Giger, W., “Estrogenic metabolites of alkylphenol polyethoxylates in secondary sewage effluents and rivers,”Water Science and Technology,42(7), 15 (2000).
Altschuh, J., Brüggemann, R., Santl, H., Eichinger, G. and Piringer, O. G., “Henry’s law constants for a diverse set of organic chemicals: Experimental determination and comparison of estimation methods,”Chemosphere,39(11), 1871 (1990).
Atlas, E., Sullivan, V. K. and Giam, C. S., “A radiotracer study of airwater exchange of synthetic organic compounds,”Chemosphere,12(9–10), 1259 (1983).
Anderson, H., Siegrist, H. and Ternes, T. A., “Fate of estrogens in a municipal sewage treatment plant,”Environ. Sci.. & Tech.,37 (18), 4021 (2003).
APHA,Standard Methods, 20th ed., p. 4–137, Method 4500-03 B (1998).
Baronti, C., Curini, R., D’Ascenzo, G., Di Corcia, A., Gentili, A. and Samperi, R., “Monitoring natural and synthetic estrogens at activated sludge sewage treatment plants and in a receiving river water,”Environ. Sci. & Tech.,34(24), 5059 (2000).
Boček, P., Lekova, K., Deml, M. and Janák, J., “Separation of some typical krebs cycle acids by high-speed isotachophoresis,”Journal of Chromatography A,117(1), 97 (1979).
Boublik, T., Fried, V. and Hala, E.,The vapor pressures of pure substances selected values of temperature dependence of the vapor pressures of some pure substance in the normal and low presuure region, Elsevier Sci. Publ., Amsterdam, Netherlands 2nd Edition, (17), 241 (1984).
Carballa, M., Omil, F., Lema, J. M., Llompart, M., Garcia-Jares, C., Rodriguez, I., Gomez, M. and Ternes, T., “Behavior of pharmaceuticals, cosmetics and hormones in a sewage treatment plant,”Water Res.,38(12), 2918 (2004).
Chao, J., Lin, C. T. and Chung, T. H., “Vapor pressure of coal chemicals,”Jour. Phys Chem Ref Data,12, 1033 (1983).
Choi, J. W., Song, H. K., Lee, W., Koo, K. K., Han, C. and Na, B. K., “Reduction of COD and color of acid and reactive dyestuff wastewater using ozone,”Korean J. Chem. Eng.,21, 398 (2004).
Daubert, T. E. and Danner, R. P.,Physical and thermodynamic properties of pure chemicals: data compilation. design inst physical property data, American Institute of Chemical Engineering. Hemisphere Pub. Corp., New York, NY. Cited in HSDB (1989).
Defoe, D. L., Holcombe, G. W., Hammermeister, D. E. and Biesinger, K. E., “Solubility and toxicity of eight phthalate esters to four aquatic organisms,”Environ. Toxicol. Chem.,9, 623 (1990).
De Bruijn, J., Busser, F. and Seinen, W., “Determination of octanol/water partition coefficients for hydrophobic organic chemicals with the “slow-stirring” method,”Environ Toxicol Chem.,8, 499 (1989).
Desbrow, C., Routledge, E. J., Brighty, G. C., Sumpter, J. P. and Waldock, M., “Identification of estrogenic chemicals in STW effluent 1. Chemical fractionation and in vitro biological screening,”Environ. Sci. & Tech.,32(11), 1549 (1998).
Donovan, S., “New method for estimating vapor pressure by the use of gas chromatography,”J. Chromatogr A.,749, 123 (1996).
Dorn, P. B., Chou, C. S. and Gentempo, J. J., “Degradation of bisphenol A in natural waters,”Chemosphere,16(7), 1501 (1987).
Ellington, J. J. and Floyd, T. L.,Measuring octanol/water partition coefficients by the slow-stirring method, U.S. Environmental Protection Agency, Athens, Georgia, in press (1996).
Fujita, M., Ike, M., Mori, K., Kaku, H., Sakaguchi, Y., Asano, M., Maki, H. and Nishihara, T.1, “Behaviour of nonylphenol ethoxylates in sewage treatment plants in Japan — biotransformation and ecotoxicity,”Water Sci. & Tech.,42(7), 23 (2000).
Hansch, C., Leo, A. and Hoekman, D., “Exploring QSAR: hydrophobic, electronic, and steric constants,” ACS Professional Reference Book.Chemosphere,29(6), 1155 (1995).
Hinckley, D. A., Bidleman, T. F., Foreman, W. T. and Tuschall, J. R., “Determination of vapor pressures for nonpolar and semipolar organic compounds from gas chromatographic retention data,”Chem. Eng. Data,35, 232 (1990).
Howard, P. H., Banerjee, S. and Robillard, K. H., “Measurement of water solubilities, octanol/water partition coefficients and vapor pressures of commercial phthalate esters,”Environ. Toxicol. Chem.,4, 653 (1985).
Huber, M. M., Canonica, S., Park, G. Y. and Von, G. U., “Oxidation of pharmaceuticals during ozonation and advanced oxidation processes,”Environ. Sci. & Tech.,37(5), 1016 (2003).
Itoh, S., Ueda, H., Naasaka, T., Nakanishi, G. and Sumitomo, H., “Evaluating variation of estrogenic effect by drinking water chlorination with the MVLN assay,”Water Sci. & Tech.,42(7–8), 61 (2000).
Japan Environmental Protection Agency. Survey of endocrine disrupting chemicals in the environment in 1999–2002.http://w-edcdb. nies.go.jp/HPEF/MEDB_e.html (2002).
Rhim, J. A., “Characteristics of adsorption and biodegradation of dissolved organic carbon in biological activated carbon pilot plant,”Korean J. Chem. Eng.,23, 38 (2006).
Kawaguchi, M., Takahashi, S., Seshimo, F., Sakui, N., Okanouchi, N., Ito, R., Inoue, K., Yoshimura, Y., Izumi, S., Makino, T. and Nakazawa, H., “Determination of 4-tert-octylphenol and 4-nonylphenol in laboratory animal feed sample by stir bar sorptive extraction followed by liquid desorption and column-switching liquid chromatography-mass spectrometry with solid-phase extraction,”Jour. of Chromatography A,1046(1–2), 83 (2004).
Komori, K., Miyamoto, N., Tanaka, H., Sato, C., Hayashi, K. and Horiuchi, S., Reduction of the estrogen-like substances and estrogen-like activity in the river water treatment facilities. Global threats to large lakes: managing in an environment of instability and unpredictability, 81 (2003).
Korea Ministry of Environment (2004). Industrial wastewater: generation and treatment in 2003.http://www.me.go.kr, 61–78.
Karickhoff, S. W., Brown, D. S. and Scott, T. A., “Sorption of hydrophobic pollutants on natural sediments,”Water Res.,13, 241 (1979).
Kuramitz, H., Saitoh, J. and Hattori, T., “Electrochemical removal of pnonylphenol from dilute solutions using a carbon fiber anode,”Water Res.,36 (13), (2002).
Lee, J., Park, H. and Yoon, J., “Ozonation characteristics of bisphenol A in water,”Environ. Technol.,24(2), 241 (2003).
Maquire, R. J., “Review of the persistence of nonylphenol and nonylphenol ethoxylates in aquatic environments,”Water Quality Res. J. of Canada,34(1), 37 (1999).
Meylan, W. M. and Howard, P. H., “Bond contribution method for estimating Henry’s law constants,”Environ. Toxicol. Chem.,10, 1283 (1991).
Meylan, W. M. and Howard, P. H., “Atom/fragment contribution method for estimating octanol-water partition coefficients,”Jour. of Pharm. Sci.,84, 83 (1995).
Meylan, W. M. and Howard, P. H., “Improved method for estimating water solubility from octanol/water partition coefficient,”Environ. Toxicol. Chem.,15, 100 (1996).
Myrdal, P., Ward, G. H., Dannenfelser, R. M., Mishra, D. and Yalkowsky, S. M., AQUAFAC 1: “Aqueous functional group activity coefficients; application to hydrocarbons,”Chemosphere,24(8), 1047 (1992).
Nasu, M., Goto, M., Kato, H., Oshima, Y. and Tanaka, H., “Study on endocrine disrupting chemicals in wastewater treatment plants,”Water Sci. & Tech.,43(2), 101 (2001).
Neely, W. B. and Blau, G. E.,Environmental exposure from chemicals, vol. 1. Boca Raton, FL: CRC Press, Inc. (1985).
Paterson, S., Mackay, D., Tam, D. and Shiu, W. Y., “Uptake of organic chemicals by plants: a review of processes, correlations and models,”Chemosphere,21, 297 (1990).
Perry, R. H. and Green, D. W.,Perry’s chemical engineers’ handbook, 6th Ed., New York, NY, McGraw-Hill (1984).
Rodriguez-Mozaz, S., López de Alda, M. J. and Barceló, D., “Monitoring of estrogens, pesticides and bisphenol A in natural waters and drinking water treatment plants by solid-phase extraction-liquid chromatography-mass spectrometry,”Jour. of Chromatography A,1045(1–2), 85 (2004).
Roefer, P., Snyder, S., Zegers, R. E., Rexing, D. J. and Fronk, J. L., “Endocrine-disrupting chemicals in a source water,”Jour. of AWWA,92(8), 52 (2000).
Routledge, E. J., Sheahan, D., Desbrow, C., Brighty, G. C., Waldock, M. and Sumpter, J. P., “Identification of estrogenic chemicals in STW effluent,” 2. In vivo responses in trout and roach.Environ. Sci. & Tech.,32, 559 (1998).
Rudel, R. A., Melly, S. J., Geno, P. W., Sun, G. and Brody, J. G., “Identification of alkylphenols and other estrogenic phenolic compounds in wastewater,” septage, and groundwater on Cape Cod, Massachusetts.Environ. Sci. & Tech.,32(7), 861 (1998).
Ryoko, K. N., Yoshiyasu, T. and Ryushi, N., “Identification of estrogenic activity of chlorinated bisphenol A using a GFP expression system,”Environ. Toxicology & Pharmacology,12(1), 27 (2002).
Shiu, W. Y., William, D., Frank, A. P., Gobas, P. C., Anders, A. and Donald, M., “Physical-chemical properties of chlorinated dibenzopdioxins,”Environ. Sci. & Tech.,22, 651 (1988).
Shiu, W. Y., Ma, K. C., Varhaní ková, D. and Mackay, D., “Chlorophenols and alkylphenols: A review and correlation of environmentally relevant properties and fate in an evaluative environment,”Chemosphere,29(6), 1155 (1994).
Snyder, S. A., Westerhoff, P. and Yoon, Y., “Pharmaceuticals, personal care products, and Endocrine disruptors in water: Implications for the water industry,”Environ. Eng. Sci.,20(5), 449 (2003).
Staples, C. A., Dorn, P. B., Klecka, G. M., Branson, D. R., O’Block, S. T. and Harris, L. R., “A review of the environmental fate, effects and exposures of bisphenol A,”Chemosphere,36, 2149 (1998).
Staples, C. A., Markham, D. and McNett, D., Analysis of surface water receiving streams for bisphenol a: manufacturing sites, final report to bisphenol A task group, The Society of the Plastics Industry, Inc., Washington, D.C., May 14 (1997).
Svenson, A., Allard, A. S. and Ek, M., “Removal of estrogenicity in Swedish municipal sewage treatment plants,”Water Res.,37(18), 4433 (2003).
Talley, P. K., Sangster, J., Bale, C. W. and Pelton, A. D., “Prediction of vapor-liquid and liquid-liquid equilibria and thermodynamic properties of multicomponent organic systems from optimized binary data using the Kohler method,”Fluid Phase Equilibria,85, 101 (1993).
Tanghe, T. and Verstraete, W., “Adsorption of nonylphenol onto granular activated carbon,”Water, Air and Soil Pollution,131(1–4), 61 (2001).
Ternes, T. A., Meisenheimer, M., McDowell, D., Sacher, F., Brauch, H. J., Haist-Gulde, B., Preuss, G., Wilme, U. and Zulei-Seibert, N., “Removal of pharmaceuticals during drinking water treatment,”Environ. Sci. & Tech.,36(17), 3855 (2002).
van Vlaardingen, P. L.A., Posthumus, R. and Traas, Environmental risk limits for alkylphenols and alkylphenol ethoxylates, RIVM report 601501019/2003, National Institute of Public Health and the Environment, PO Box 1, 3720 BA Bilthoven, The Netherlands (2003).
Verstraeten, I. M., Heberer, T., Vogel, J. R., Speth, T., Zuehlke, S. and Duennbier, U., “Occurrence of endocrine-disrupting and other wastewater compounds during water treatment with case studies from Lincoln, Nebraska and Berlin, Germany,”Pract. Periodical Hazard., Toxic, Radioact. Waste Manage,79(4), 253 (2003).
Yamamoto, T., Yasuhara, A., Shiraishi, H. and Nakasugi, O., “Bisphenol A in hazardous waste landfill leachate,”Chemosphere,42(4), 415 (2000).
Yaws, C. L., “Water solubility and Henry’s law constant: Part 3 Aromatics,”Waste Management,14(3–4), 337 (1994).
Ying, G. G., Kookana, R. S. and Dillon, P., “Sorption and degradation of selected five endocrine disrupting chemicals in aquifer material,”Water Res.,37(15), 3785 (2003).
Zacharewski, T., Identification and assessment of endocrine disruptors: limitation of in vivo and in vitro assays.Environmental Health Perspectives 106 (Supplement 2), 577 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, K.J., Kim, S.G., Kim, C.W. et al. Removal efficiencies of endocrine disrupting chemicals by coagulation/flocculation, ozonation, powdered/granular activated carbon adsorption, and chlorination. Korean J. Chem. Eng. 23, 399–408 (2006). https://doi.org/10.1007/BF02706741
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706741