Abstract
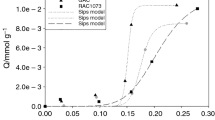
Equilibrium data for the adsorption of hydrogen, carbon dioxide, and binary mixture of both gases on activated carbon were determined experimentally. Pure component isotherms were presented along with pressures up to 30 atm at 301 K, 323 K and 348 K. Also, the binary equilibria were obtained at various temperatures same above for pressure of 1.5, 10 and 20 atm, respectively. For the pure component system, Freundlich isotherm was shown to be fitted best to the experimental results. However, in the binary system, the ideal adsorption solution (IAS) theory gave good representation of the binary experimental data in high pressure range.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- a, al :
-
parameters for adsorption isotherm
- b, bi :
-
parameters for adsorption isotherm
- k, ki :
-
parameters for adsorption isotherm
- m:
-
parameter for adsorption isotherm
- N:
-
numbers of component
- Nd :
-
numbers of data point
- n:
-
parameter for adsorption isotherm
- P:
-
total pressure
- pi :
-
partial pressure of component i
- q:
-
amount of adsorbed
- q cal :
-
calculated value of adsorbed amount
- q exp :
-
experimental value of adsorbed amount
- Γ ii,Γij :
-
parameters for statistical thermodynamic model
- i, j:
-
of component i, j
- AD:
-
average percent deviation
References
Langmuir, I.:J. Amer. Chem. Soc,40, 1361 (1918).
Redlich, O. and Peterson, D. L.:J. Phys. Chem.,63, 1024(1959).
Radke, C. J. and Prausnitz, J. M.:AIChE J.,18, 761 (1972).
Toth, J.:J. Colloid Interf. Sci.,79, 85 (1981).
Toth, J.:J. Colloid Interf. Sci.,94, 573(1983).
Koresh, J. and Soffer, A.:J. Colloid Interf. Sci.,92, 517 (1983).
Sips, R.:J. Chem. Phys., 16, 490 (1948).
Ruthven, D.M.: Principles of Adsorption and Adsorption Process, New York, Wiley, 1984.
Butler, J.A. V. and Ockerent, C:J. Phys. Chem.,34, 2841 (1930).
Yon, C. M. and Turnock, P. H..:AIChE Symp. Ser.,67, 3(1971).
Ruthven, D. M. and Wong, F.:Ind. Eng. Chem. Fundam.,24, 27(1985).
Myers, A. L. and Prausnitz, J. M.:AIChE J.,11, 121 (1965).
Suwanayuen, S. and Danner, R. P.:AIChE J.,26, 68 (1980).
Sheindorf, C. H., Rebhum, M. and Scheituch, M.:J. Colloid Interf. Sci.,79, 136 (1981).
Fritz, W. and Schlundar, E.U.:Chem. Eng. Sci.,36, 136(1981).
Hwang, K. S.: M.S. Thesis, KAIST, 1990.
Menon, P. G.:Chem. Rev.,68, 277(1968).
Ozawa, S., Wakasuki, Y. and Ozino, Y.:J. Colloid Interf. Sci.,79, 399(1981).
Kim, H.J., Moon, H. and Park, H. C.:Korean J. Chem. Eng.,2, 181(1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hwang, K.S., Gong, S.Y. & Lee, W.K. Adsorption equilibria for hydrogen and carbon dioxide on activated carbon at high pressure up to 30 atm. Korean J. Chem. Eng. 8, 148–155 (1991). https://doi.org/10.1007/BF02706676
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706676