Abstract
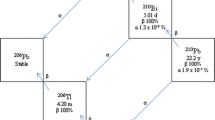
Magnesium isotope separation was investigated by chemical ion exchange with the 1-aza-12-crown-4 bonded Merrifield peptide resin using elution chromatography. The capacity of the novel azacrown ion exchanger was 1.0 meq/g dry resin. The heavier isotopes of magnesium were enriched in the resin phase, while the lighter isotopes were enriched in the solution phase. The single stage separation factor was determined according to the method of Glueckauf from the elution curve and isotopic assays. The separation factors of24Mg2+-25Mg2+,24Mg2+-26Mg2+, and25Mg2+-26Mg2+ isotope pairs were 1.012, 1.024, and 1.011, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Ceq :
-
metal ion concentration of the standard solution [mole/l]
- Cst :
-
metal ion concentration after equilibrium [mole/l]
- Kd :
-
distribution coefficient
- M:
-
mass of ion exchanger [g]
- N:
-
number of theoretical plated in the column
- V:
-
total volume of solution [ml]
- Vmax :
-
peak elution volume [ml]
- α:
-
separation factor [1 + ε]
- Δm:
-
isotope mass difference unit
- ε/Δm:
-
enrichment factor per mass unit
- ε:
-
enrichment factor [α-1]
References
Aaltonen, J., “Separation of Alkaline Earth Metal Isotopes by Ion Exchange”,Suom. Kemi.,B44, 1 (1971).
Aaltonen, J., Heumann, K. G. and Pietila, P., “Isotope Effects of Uranium by Ion Exchange in the System Dowex 50/μ-Hydroxyisobutyrate”,Z.Naturforsch.,29b, 190 (1974).
Bradshaw, J. S., Krakowiak, K. E. and Izatt, R. M., “Aza-Crown Macrocycles”, Interscience Publication, John Wiley & Sons, Inc., New York (1993).
Dormer, K., “Ionenaustauscher”, Walter de Gruyter, Berlin, New York (1970).
Dormer, K., “Synthetic Ion Exchange Resin”, Ch. 1 in Ion Exchanger, Dorfner, K. (Ed.) Walter de Gruyter, Berlin, New York (1991).
Fujine, S., Saito, K. and Shiba, K., “Lithium Isotope Separation by Displacement Chromatography using Cryptand Resin”,J. Nucl. Sci. Technol,20, 439 (1983).
Glueckauf, E., “Theory of Chromatography”,Trans. Faraday Soc,51, 34 (1955).
Glueckauf, E., “Theory of Chromatography”,Trans. Faraday Soc,54, 1203 (1958).
Heumann., K. G. and Lieser, K. H., “Untersuchung von Calciumisotoieeffekten bei heterogenen Austauschgleichgewichten”,Z.Naturforsch.,27b, 126 (1972).
Heumann, K. G. and Schiefer, H. P., “Calcium Isotope Separation on an Exchange Resin Having Cryptand Anchor Groups”,Angrew. Chem.,92, 406 (1980).
Hiraoka, M., “Crown Compounds, Their Characteristics and Applications”, Elsevier Scientific Pub. Comp., Amsterdam-Oxford, New York (1982).
Jepson, B. E. and De Witt, R., “Separation of Calcium Isotopes with Macrocyclic Polyether Complexes”,J. Inorg. Nucl. Chem.,38, 1175 (1976).
Jepson, B. E., Novotny, M. A. and Evans, W. F., “Mound Activities in Chemical and Physical Research: January-December 19907 MLM-3695, EG&G Mound Applied Technologies, Miamisburg, Ohio 45343-3000 (1991).
Kim, D. W, Jeon, B. K., Kim, C. S. and Jeon, Y S., “Enrichment of Magnesium Isotopes by N3O2 Azacrown Ion Exchange Resin”,J. Radioanal. Nucl. Chem.,245, 509 (2000).
Klinskii, G. D., Knyazev, D. A. and Vlasova, G. L., “Chromatographic Separation of Calcium Isotopes”,Russ. J. Phys. Chem.,48, 330 (1974).
Kondoh, A., Oi, T. and Hosoe, M., “Fractionation of Barium Isotopes in Cation-Exchange ChromatographySep. Sci. Technol.,31, 39 (1996).
Konstantinov, B. and Bakulin, P. E. A., “Separation of24Mg2+-26Mg2+ and63Cu2+-65Cu2+ Isotope Pairs in Aqueous Solution of Magnesium Chloride and Copper Chloride”,Russ. J. Phys. Chem.,39, 1 (1965).
Lee, D. A., “Separation of Isotopes of Selected Divalent Elements by Ion Exchange”,J. Inorg. Nucl. Chem.,38, 161 (1976).
Lee, D. H. and Moon, H., “Adsorption Equilibrium of Heavy Metals on Natural Zeolites”,Korean J. Chem. Eng.,18, 247 (2001).
Neubert, A. and Klemm, A., “Isotopentrennung bei Mg, Ca, Sr und Ba durch Ionenwanderung in geschmolzenen Halogeniden”,Z.Naturforsch.,16a, 685 (1961).
Nishizawa, K., Nishida, T., Yamato, T. and Hoseo, M., “Extractive Behavior of Magnesium Chloride and Isotopic Enrichment of Magnesium by Crown Ether”,Sep. Sci. Technol.,31, 643 (1996).
Oi, T., Kawada, K., Hosoe, M. and Kakihana, H., “Fractionation of Lithium Isotopes in Cation Exchange Chromatography”,Sep. Sci. Technol.,26, 1353 (1991).
Oi, T., Ogino, H., Hosoe, M. and Kakihana, H., “Fractionation of Strontium Isotopes in Cation-Exchange Chromatography”,Sep. Sci. Technol.,27, 631 (1992).
Ooi, K., Miyai, Y., Makita, Y. and Kanoh, H., “Fractionation of Lithium Isotopes in Ion Exchange Chromatography with Titanium Phosphate Exchanger”,Sep. Sci. Technol.,34, 1133 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, D.W., Joen, B.K., Kwon, S.H. et al. Separation of magnesium isotopes by 1-aza-12-crown-4 bonded Merrifield peptide resin. Korean J. Chem. Eng. 18, 750–754 (2001). https://doi.org/10.1007/BF02706395
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706395