Abstract
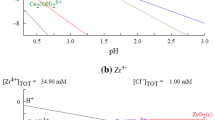
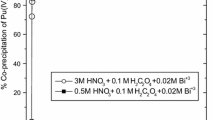
The partitioning of americium and neodymium by precipitation with oxalic acid was investigated in the simulated radwaste, which was composed of 10 elements of alkali, akaline earth, and transition metals in nitric acid solution. The effect of concentrations of oxalic acid and nitric acid in the simulated waste on the precipitation yield and purity of Nd and Am was examined in this study. As a result, the precipitated fraction of each element was increased with increasing concentration of oxalic acid and decreasing concentration of nitric acid. At an initial concentration of 0.5 M nitric acid and 0.5 M oxalic acid, both Am3 and Nd3+ were precipitated over 99% and other elements almost remained in the simulated solution. It was also found that Am was completely coprecipitated with Nd-oxalate precipitates, and Zr4- caused the coprecipitation of Cs2, Sr2+, and Pd2+.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Burney, G. A. and Porter, J.A., “Solubilities of Pu(III), Am(III), and Cm(III) Qxalates”,Inorg. Nucl. Chem. Letters,3, 79 (1967).
Crouthamel, C.E. and Martin, D.S. Jr., “The Solubility of Ytterbium Oxalate and Complex Ion Formation in Oxalate Solutions”,J. Am. Chem. Soc.,72, 1382 (1950).
Crouthamel, C. E. and Martin, D. S. Jr., “Solubility of the Rare Earth Oxalates and Complex Ion Formation in Oxalate Solution (II), Neodymium(III) and Cerium(III)”,J. Am. Chem. Soc.,73, 569 (1951).
Forsberg, C. W., “Separation of Americium, Curium, and Trivalent Lanthanides from High-Level Wastes by Oxalate Precipitation: Experiments with Synthetic Waste Solutions”,Nuclear Technology,49, 243(1980).
IAEA Technical Reports Series No. 214,“Evaluation of Actinide Partitioning and Transmutation”, IAEA, Vienna (1982).
IAEA Technical Reports Series No. 137,“Chemical Precipitation Process for the Treatment of Aqueous Radioactive Waste”, IAEA, Vienna (1992).
Kim, E. H., et al., in preparation.
Kobayashi, T., Morita, Y. and Kubota, M.,“Development of Partitioning Method : Method of Precipitation Transuranium Elements with Oxalic acid”, JAERI-M88-026 (1988).
Kobayashi, T., Shirahashi, K. and Kubota, M.,“Development of Partitioning Method : Precipitation Behavior of Other Elements in Separating the Transuranic Elements Group with Oxalic Acid”, JAERI-M89-168 (1989).
Kubota, M. and Fukase, T., “Formation of Precipitate in High-Level Liquid Waste from Nuclear Fuel Reprocessing”,J. Nuclear Sci. and Tech.,17(10), 783(1980).
Miyashio, H. and Kubota, M.,“Development of Partitioning Method : Fundamental Study on Oxalate Precipitation Method”, JAERI-M86-0144 (1986).
Medeiaos, L. O., “Cation-Exchange Properties of Zirconyl Oxalate Precipitates”,J. Inorg. Nucl. Chem.,28, 599(1966).
Posey, J. C.,“Process for the Recovery of Cm-244 from Nuclear Waste”, ORNL-5687 (1980).
Rankin, D. T., Burney, G. A., Smith, P. K. and Sission, R. D. Jr., “Precipitation and Characterization of Oxalate-Based237NpO2 Powder”,Ceramic Bulletin,56(5), 478(1977).
Shirahashi, K. and Kubota, M., “Precipitation Behavior of Transuranium Elements during Denitration of High-Level Radioactive Liquid Waste by Formic Acid”,J. Nuclear Sci. and Tech.,29(6), 61 (1992).
Skalberg, M. and Liljenzin, J.-O., “Partitioning and Transmutation: the State of the Art”,Nuclear Eng. Int.,Feb., 30(1990).
Tedder, D. W. and Blomeke, J. O.,“Actinide Partitioning and Transmutation Program Progress Report for Period April 1 to June 30, 1977”, ORNL/TM-6056 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, EH., Shin, YJ., Kim, WH. et al. Partitioning of neodymium and americium in the liquid radioactive waste by oxalate precipitation. Korean J. Chem. Eng. 12, 557–562 (1995). https://doi.org/10.1007/BF02705859
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705859